Optimized Synthesis of Pemetrexed Disodium Intermediates for Commercial Scale Production
Optimized Synthesis of Pemetrexed Disodium Intermediates for Commercial Scale Production
The pharmaceutical landscape for oncology treatments continues to evolve, with Pemetrexed disodium remaining a cornerstone therapy for malignant pleural mesothelioma and non-small cell lung cancer. As demand for this multi-target antifolate agent grows, the efficiency and purity of its supply chain become critical bottlenecks for global manufacturers. Patent CN110305135B introduces a transformative approach to synthesizing key intermediates, specifically addressing the longstanding challenge of impurity control in the condensation of the pyrrolo-pyrimidine core with glutamic acid derivatives. This technical insight report analyzes the strategic advantages of this protected intermediate strategy, offering R&D directors and procurement leaders a clear pathway to cost reduction in API manufacturing while ensuring the highest standards of product quality.
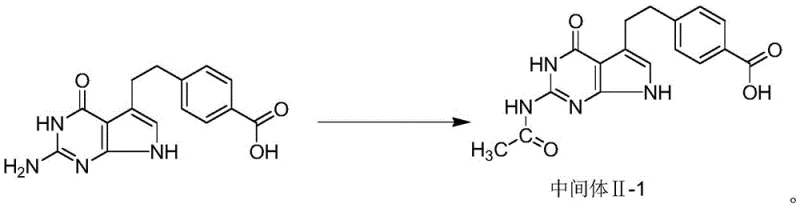
The core innovation lies in the preparation of Intermediate II-1, 4-[2-(2-acetamido-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidine-5-yl)ethyl]benzoic acid. By shifting the synthetic sequence to protect the reactive amino group prior to coupling, the process fundamentally alters the impurity profile of the reaction. This modification is not merely a incremental tweak but a structural reimagining of the synthesis route that eliminates the formation of the notorious Impurity V, a byproduct that has historically plagued production yields and necessitated costly, repetitive purification cycles. For a reliable pemetrexed intermediate supplier, mastering this protection-deprotection sequence is essential for delivering consistent, high-purity materials to downstream drug product manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Pemetrexed disodium typically involve the direct condensation of 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidine-5-yl)ethyl]benzoic acid with L-glutamic acid diethyl ester. While conceptually straightforward, this direct approach suffers from significant chemical liabilities due to the presence of multiple reactive sites on the starting material. The primary amino group on the pyrimidine ring is highly nucleophilic and prone to side reactions, leading to the generation of complex byproducts, most notably Impurity V. In standard operations, the content of this specific impurity can reach levels as high as 5-10%, which is unacceptable for pharmaceutical grade materials.
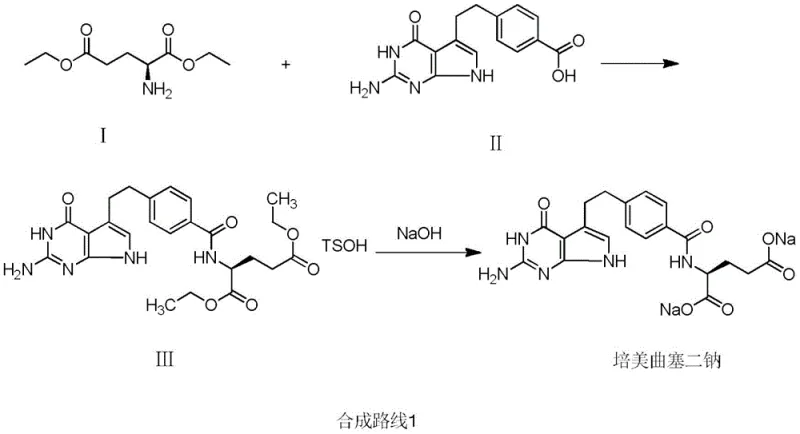
To meet stringent regulatory purity specifications, manufacturers employing the conventional route are forced to implement rigorous purification protocols. This often entails repeating recrystallization processes two to three times, which drastically erodes the overall process yield and inflates production costs through excessive solvent consumption and extended equipment occupancy. Furthermore, the free amino group in the starting material is susceptible to oxidative degradation, leading to discoloration and the formation of additional unidentified impurities that complicate the purification landscape. These factors combine to create a fragile supply chain where small variations in reaction conditions can lead to batch failures, posing a significant risk to commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The patented methodology circumvents these issues by introducing a proactive protection step. The synthesis begins with the acylation of the starting benzoic acid derivative using acetic anhydride in an acetic acid medium. This reaction selectively converts the primary amino group into an acetamido group, yielding Intermediate II-1. This structural modification effectively masks the reactive nitrogen, rendering it inert during the subsequent coupling reaction with the glutamic acid derivative. By preventing the amino group from participating in side reactions, the formation of Impurity V is virtually eliminated at the source.
The benefits of this approach extend beyond simple impurity suppression. The protected intermediate exhibits superior stability, resisting the oxidative discoloration that characterizes the unprotected amine. Consequently, the downstream condensation reaction proceeds with much higher fidelity, producing the coupled intermediate (Intermediate III-1) with significantly improved purity profiles. The patent data indicates that this optimization improves the overall yield by nearly 40%, a massive gain in the context of multi-step API synthesis. This efficiency gain translates directly into reduced raw material waste and lower operational expenditures, making the process highly attractive for high-purity pharmaceutical intermediates production on an industrial scale.
Mechanistic Insights into Acylation Protection Strategy
The success of this synthetic route hinges on the precise control of the acylation mechanism. The reaction utilizes acetic anhydride as the acylating agent in a glacial acetic acid solvent system. Under mild heating conditions of 40-50°C, the nucleophilic attack of the primary amine on the carbonyl carbon of the acetic anhydride proceeds rapidly. The choice of acetic acid as both solvent and a component of the reaction medium helps to solubilize the polar starting material while maintaining a pH environment that favors the formation of the mono-acetylated product without promoting hydrolysis of the sensitive pyrrolo-pyrimidine ring. The resulting acetamido group provides sufficient steric bulk and electronic deactivation to prevent the nitrogen from acting as a nucleophile during the subsequent activation with N,N-carbonyldiimidazole (CDI).
Furthermore, the protection strategy simplifies the final deprotection and hydrolysis steps. In the final stage of the synthesis, the intermediate III-1 is treated with sodium hydroxide. This basic environment serves a dual purpose: it hydrolyzes the ethyl esters of the glutamic acid moiety to form the carboxylate salts, and simultaneously cleaves the acetamido protecting group to regenerate the free amine required for the biological activity of Pemetrexed. The fact that deprotection and salt formation can be achieved in a single pot or sequential steps without requiring harsh acidic conditions or specialized reagents underscores the elegance of this route. This streamlined mechanism ensures that the final product, Pemetrexed disodium, achieves purity levels exceeding 99.85%, meeting the rigorous demands of global pharmacopeias.
How to Synthesize Pemetrexed Disodium Efficiently
The implementation of this optimized route requires careful attention to stoichiometry and temperature control, particularly during the acylation and condensation phases. The process is designed to be robust, utilizing common industrial solvents and reagents that are readily available in the global supply chain. The following sections outline the critical operational parameters derived from the patent examples, providing a framework for process engineers to adapt this chemistry for larger reactor volumes. For detailed standard operating procedures and safety data, please refer to the specific technical guidelines provided below.
- Prepare Intermediate II-1 by reacting 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidine-5-yl)ethyl]benzoic acid with acetic anhydride in acetic acid at 40-50°C.
- Condense Intermediate II-1 with L-glutamic acid diethyl ester using CDI activation, followed by tosylation to form Intermediate III-1.
- Hydrolyze the ester and amide groups of Intermediate III-1 using sodium hydroxide, followed by pH adjustment and salt formation to obtain Pemetrexed disodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this protected intermediate strategy offers tangible economic and logistical benefits that go beyond simple yield metrics. The elimination of repetitive recrystallization steps significantly reduces the consumption of organic solvents such as ethanol and DMF, which are major cost drivers in API manufacturing. Additionally, the reduction in processing time—by removing the need for multiple purification cycles—increases the throughput of existing manufacturing facilities, allowing for greater production capacity without capital investment in new equipment. This efficiency is crucial for maintaining competitive pricing in the generic oncology market.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the drastic reduction in material loss associated with purification. In conventional routes, recovering product from mother liquors after multiple crystallizations is inefficient and often results in significant yield loss. By achieving high purity directly from the reaction, the new method minimizes waste disposal costs and maximizes the utilization of expensive starting materials. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower overall carbon footprint and operational cost structure.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the variability of complex chemical syntheses. The robustness of the acylation protection step ensures consistent batch-to-batch quality, reducing the risk of out-of-specification results that can delay shipments. The use of stable intermediates also allows for potential stockpiling strategies, where Intermediate II-1 can be produced and stored, decoupling the upstream synthesis from the final coupling step. This flexibility enables suppliers to respond more agilely to fluctuations in market demand for Pemetrexed disodium.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the simplicity of this route mitigates those risks. The reactions proceed at moderate temperatures (30-80°C range), avoiding the hazards associated with cryogenic conditions or high-pressure systems. Moreover, the reduction in solvent usage and waste generation aligns with increasingly strict environmental regulations. A cleaner process with fewer purification steps simplifies wastewater treatment and solvent recovery, ensuring long-term compliance and sustainability for manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Pemetrexed disodium intermediates. These answers are derived from the specific technical disclosures in patent CN110305135B and represent the current state-of-the-art in process chemistry for this molecule. Understanding these details is vital for technical teams evaluating potential suppliers or optimizing their own internal production capabilities.
Q: How does the new synthesis route reduce Impurity V?
A: The novel route introduces an acetyl protection group on the amino moiety of the starting material (Intermediate II-1) prior to condensation. This steric and electronic blocking prevents the nucleophilic attack that leads to the formation of Impurity V, reducing its content from 5-10% to negligible levels.
Q: What are the yield improvements in this patented method?
A: By eliminating the need for multiple recrystallizations to remove Impurity V and preventing oxidative discoloration, the overall yield of the intermediate is improved by nearly 40% compared to conventional direct condensation methods.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes mild reaction conditions (40-50°C) and common solvents like acetic acid and DMF. The elimination of complex purification steps makes it highly scalable and cost-effective for commercial API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the protected intermediate route are fully realized in our manufacturing plants. We operate under stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Pemetrexed intermediate meets the exacting standards required for oncology drug formulation.
We invite global pharmaceutical partners to collaborate with us on optimizing their supply chains for Pemetrexed disodium. Whether you are looking to secure a long-term supply of high-quality intermediates or need assistance in adapting this synthesis route for your specific facility, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
