Advanced Green Synthesis of Oxadiazole Derivatives for Commercial Scale Production
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for constructing heterocyclic scaffolds, particularly the versatile oxadiazole ring system. Patent CN108997252B introduces a groundbreaking green synthesis method that fundamentally shifts the paradigm from hazardous organic solvents to benign aqueous media. This innovation addresses critical pain points in the manufacturing of oxadiazole derivatives, which serve as pivotal intermediates for AT1 receptor antagonists and COX inhibitors. By leveraging a one-pot cyclization strategy using N-hydroxybenzamidine and carbonylation reagents in water, this technology offers a compelling value proposition for reliable oxadiazole derivative suppliers aiming to enhance their portfolio with safer, high-yield processes. The transition to water not only mitigates environmental risks but also streamlines the isolation procedure, presenting a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxadiazole core has been plagued by significant safety hazards and operational complexities that hinder large-scale commercial adoption. Traditional synthetic routes frequently rely on potassium cyanate as a key reagent; however, this compound poses a severe safety risk because contact with acidic conditions generates extremely toxic cyanide gas, creating dangerous working environments and requiring expensive containment infrastructure. Another common pathway utilizes diketene under the influence of sodium hydride, but diketene is a potent lachrymator and sodium hydride is pyrophoric, both of which classify as dangerous chemical raw materials that increase production danger and complicate industrial safety protocols. Furthermore, these conventional methods often necessitate the use of volatile organic solvents, leading to high VOC emissions and costly solvent recovery systems that erode profit margins. The multi-step nature of some older protocols also results in lower overall yields and increased waste generation, making them increasingly untenable in a regulatory landscape that demands greener chemistry solutions.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a direct, one-step cyclization in water, effectively simplifying what was previously a multi-step esterification and cyclization sequence into a single efficient operation. By employing N-hydroxybenzamidine or its substituted derivatives alongside safe carbonylation reagents like chloroformates, the reaction proceeds smoothly under mild alkaline conditions without generating toxic byproducts. The use of water as the sole solvent eliminates the fire hazards associated with organic solvents and drastically reduces the environmental footprint of the manufacturing process. This method demonstrates wide substrate applicability, accommodating various electron-donating and electron-withdrawing groups on the benzene ring without compromising yield or purity. The simplicity of the workup, involving merely acidification and filtration, represents a major leap forward in process efficiency, allowing for rapid turnaround times and reduced labor costs.
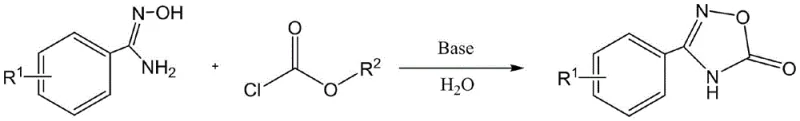
Mechanistic Insights into Base-Catalyzed Aqueous Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway where the nucleophilic nitrogen of the N-hydroxybenzamidine attacks the electrophilic carbonyl carbon of the chloroformate reagent. In the presence of an alkaline reagent, such as DMAP or inorganic bases like sodium carbonate, the deprotonation of the hydroxylamine moiety enhances its nucleophilicity, facilitating the initial acylation step to form an unstable intermediate. This intermediate subsequently undergoes an intramolecular cyclization, driven by the thermodynamic stability of the aromatic oxadiazole ring and the expulsion of the alcohol leaving group from the chloroformate. The aqueous environment plays a crucial role not just as a solvent but potentially in stabilizing transition states through hydrogen bonding networks, which helps to lower the activation energy barrier for the cyclization. The reaction temperature of 100°C provides sufficient thermal energy to overcome kinetic barriers while maintaining the system under reflux, ensuring consistent reaction rates across different batch sizes. This mechanism avoids the formation of harsh side products, leading to a cleaner reaction profile that is easier to purify.
From an impurity control perspective, the specificity of this reaction mechanism ensures that the primary impurities are limited to unreacted starting materials or hydrolysis products of the chloroformate, both of which are easily removed during the acidification and washing steps. The absence of heavy metal catalysts or toxic cyanide sources means that the final product is free from difficult-to-remove inorganic contaminants, which is a critical requirement for high-purity pharmaceutical intermediates. The pH control during the workup phase, specifically adjusting to pH 3-4, is strategically designed to protonate the product and induce precipitation while keeping soluble impurities in the aqueous phase. This precise control over the physicochemical properties of the reaction mixture allows for the production of materials with stringent purity specifications without the need for column chromatography. Consequently, the impurity profile is significantly cleaner compared to traditional methods, reducing the burden on quality control laboratories and accelerating the release of batches for downstream coupling reactions.
How to Synthesize Oxadiazole Derivatives Efficiently
The operational protocol for this synthesis is remarkably straightforward, designed to be easily implemented in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. The process begins with the suspension of the N-hydroxybenzamidine substrate in water, followed by the addition of the chosen base, creating a homogeneous or semi-homogeneous mixture ready for the key transformation. The carbonylation reagent is then added dropwise to manage the exotherm and ensure controlled reaction kinetics, preventing local hot spots that could lead to decomposition. Following the reflux period, the reaction mixture is cooled, and the product is isolated through a simple acid-base extraction logic that leverages the solubility differences between the product and the aqueous medium. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving N-hydroxybenzamidine or its substituted derivative in water, then add an alkaline reagent such as DMAP or sodium carbonate.
- Dropwise add a carbonylation reagent like phenyl chloroformate while stirring, and heat the mixture to reflux at 100°C for 1 to 5 hours to complete the cyclization.
- After cooling, acidify the solution to pH 3-4 with dilute HCl to precipitate the product, then filter, wash with water, and vacuum dry to obtain the pure oxadiazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this green synthesis route translates directly into tangible operational efficiencies and risk mitigation strategies that protect the bottom line. The elimination of hazardous reagents like potassium cyanate and diketene removes the need for specialized storage facilities and expensive safety monitoring equipment, thereby lowering the overhead costs associated with regulatory compliance and insurance. Furthermore, the switch to water as a solvent dramatically reduces the cost of raw materials compared to high-grade organic solvents, while simultaneously eliminating the capital expenditure required for solvent recovery distillation columns. The simplified workup procedure, which relies on filtration rather than complex extraction or chromatography, significantly shortens the production cycle time, allowing facilities to increase throughput and respond more agilely to market demand fluctuations. These factors combined create a robust supply chain framework that is less susceptible to disruptions caused by raw material shortages or environmental regulatory changes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and dangerous reagents with commodity chemicals that are readily available in the global market. By removing the need for toxic cyanide handling, manufacturers save substantially on waste disposal fees and safety training programs, which are often hidden costs in traditional synthetic routes. The high atom economy of the one-pot reaction minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable saleable product. Additionally, the energy consumption is optimized since the reaction runs at the boiling point of water, avoiding the need for cryogenic cooling or high-pressure equipment that consumes excessive electricity. These cumulative savings allow for a more competitive pricing structure for the final oxadiazole derivatives, enhancing margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials such as chloroformates and simple inorganic bases, which are not subject to the same strict export controls or supply bottlenecks as specialized toxic reagents. The robustness of the aqueous reaction conditions means that the process is less sensitive to minor variations in temperature or moisture, reducing the rate of batch failures and ensuring consistent delivery schedules. Because the purification is achieved through crystallization and filtration, the process is inherently easier to scale from pilot plant to full commercial production without the linear increase in complexity seen in chromatographic separations. This reliability ensures that partners can maintain steady inventory levels and meet just-in-time delivery requirements without the fear of unexpected production stoppages due to safety incidents.
- Scalability and Environmental Compliance: From an environmental standpoint, this method aligns perfectly with modern green chemistry principles, generating minimal hazardous waste and utilizing a non-flammable solvent that simplifies fire safety management in large-scale plants. The aqueous waste stream is easier to treat in standard wastewater facilities compared to organic solvent mixtures, reducing the environmental compliance burden and associated treatment costs. The process is highly scalable, as demonstrated by the ability to run reflux reactions in large vessels without the heat transfer limitations often encountered in viscous organic media. This scalability ensures that the technology can support the growing global demand for oxadiazole-containing drugs, from early-stage clinical trials to multi-ton commercial manufacturing, without requiring significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The answers provided reflect the specific advantages of the aqueous system and the broad substrate tolerance observed during development.
Q: Why is this water-based synthesis method safer than traditional oxadiazole production?
A: Traditional methods often utilize potassium cyanate, which generates highly toxic cyanide gas upon contact with acid, or diketene, a dangerous lachrymator. This patented green method eliminates these hazardous reagents entirely, using stable chloroformates and water instead.
Q: What are the typical reaction conditions for this cyclization process?
A: The reaction typically proceeds under reflux conditions at 100°C in an aqueous medium. It requires an alkaline catalyst (organic or inorganic base) and completes within 1 to 5 hours, depending on the specific substrate substituents.
Q: How does this method impact downstream purification costs?
A: By using water as the primary solvent, the workup is significantly simplified. The product precipitates directly upon acidification, allowing for simple filtration and washing, which removes the need for energy-intensive solvent recovery or complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxadiazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthesis technology and have integrated similar advanced aqueous methodologies into our CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of oxadiazole derivative meets the highest international standards. Our commitment to green chemistry aligns with the global push for sustainable manufacturing, allowing us to deliver high-quality intermediates that support your regulatory filings and environmental goals.
We invite you to collaborate with us to leverage these cost-effective and safe synthetic routes for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall manufacturing costs.
