Revolutionizing 1,3,4-Oxadiazole Production: A High-Yield 4-Step Process for Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient pathways to synthesize complex heterocyclic scaffolds, particularly those serving as critical intermediates for protease inhibitors. Patent CN1350525A, filed in May 2002, introduces a groundbreaking methodology for the preparation of 1,3,4-oxadiazole derivatives, specifically targeting the synthesis of compounds useful as serine protease inhibitors, such as elastase inhibitors. This patent addresses a significant bottleneck in the existing art by proposing a novel synthetic route that drastically reduces the number of operational steps while simultaneously boosting overall yield. For R&D directors and process chemists, the shift from a cumbersome multi-step sequence to a streamlined four-step protocol represents a substantial leap in process efficiency. The core innovation lies in the utilization of a new intermediate, represented by general formula (I), which serves as a versatile pivot point for constructing the final target molecule, general formula (II). By leveraging this intermediate, the inventors have successfully circumvented the low-yielding oxidation and protection/deprotection cycles that plagued previous methodologies, thereby establishing a more robust and economically viable manufacturing process for these high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 1,3,4-oxadiazole derivatives, particularly those intended for use as elastase inhibitors, relied heavily on convoluted synthetic pathways that were neither time-efficient nor cost-effective. As detailed in the background art referencing WO9824806, conventional methods often necessitated up to ten distinct reaction steps to reach the desired structural complexity. For instance, Reaction Scheme 1 in the prior art illustrates a pathway involving multiple protection and deprotection sequences, alongside challenging oxidation steps that frequently resulted in material loss. Even the slightly optimized Reaction Scheme 2 and 3 still required six steps to achieve the target structure, yet the cumulative yield remained dismally low at approximately 18% for specific methyl-substituted variants. These inefficiencies stem from the reliance on harsh oxidation conditions, such as Swern oxidation, and the need for rigorous purification after every minor transformation. For a procurement manager, these factors translate directly into inflated raw material costs, extended lead times, and a larger environmental footprint due to excessive solvent and reagent consumption. The inability to secure high-purity intermediates consistently through such long sequences also poses a significant risk to supply chain reliability, as each additional step introduces a potential point of failure or impurity generation that could compromise the final API quality.
The Novel Approach
In stark contrast to the laborious conventional routes, the methodology disclosed in CN1350525A offers a remarkably concise four-step synthesis that achieves a total yield of 65%, representing a three-to-four-fold improvement over the prior art. This novel approach, illustrated in Reaction Scheme 4, centers on the strategic coupling of a Weinreb amide derivative (Formula V) with a lithiated 1,3,4-oxadiazole species (Formula VI). This key carbon-carbon bond-forming reaction allows for the direct assembly of the core ketone functionality without the need for intermediate oxidation steps that typically degrade yield. Furthermore, the process employs mild deprotection conditions, utilizing acidic hydrolysis or hydrogenolysis, which are far more compatible with sensitive functional groups than the aggressive reagents used in older protocols. 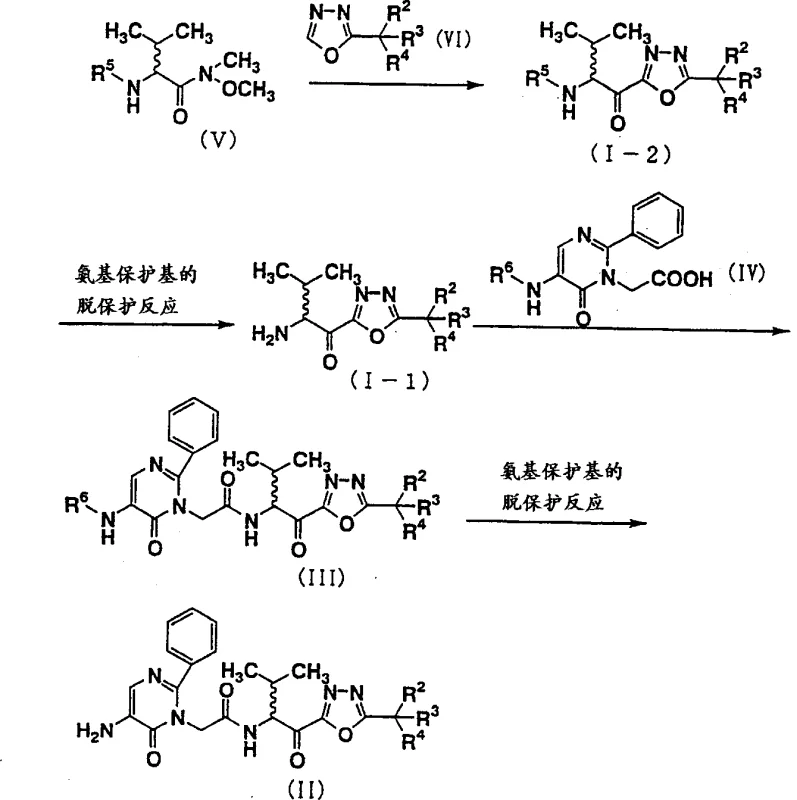 The streamlined nature of this route not only accelerates the timeline from raw materials to finished intermediate but also significantly simplifies the purification workflow. By reducing the step count from six or ten down to four, the process inherently minimizes the accumulation of impurities and reduces the volume of waste generated per kilogram of product. For supply chain heads, this translates to a more predictable and resilient production schedule, capable of meeting the demanding timelines of downstream drug development projects. The ability to produce high-purity 1,3,4-oxadiazole derivatives with such efficiency positions this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
The streamlined nature of this route not only accelerates the timeline from raw materials to finished intermediate but also significantly simplifies the purification workflow. By reducing the step count from six or ten down to four, the process inherently minimizes the accumulation of impurities and reduces the volume of waste generated per kilogram of product. For supply chain heads, this translates to a more predictable and resilient production schedule, capable of meeting the demanding timelines of downstream drug development projects. The ability to produce high-purity 1,3,4-oxadiazole derivatives with such efficiency positions this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lithiated Oxadiazole Coupling
The heart of this innovative synthesis lies in the nucleophilic addition of a lithiated 1,3,4-oxadiazole to a Weinreb amide, a transformation that demands precise control over reaction parameters to ensure high fidelity and yield. The mechanism initiates with the generation of a highly reactive nucleophile by treating the 1,3,4-oxadiazole derivative (Formula VI) with a strong base, specifically lithium diisopropylamide (LDA), in the presence of a chelating ligand like tetramethylethylenediamine (TMEDA). This step must be conducted at cryogenic temperatures, typically ranging from -78°C to -65°C, to prevent side reactions such as self-condensation or decomposition of the sensitive heterocyclic ring. The subsequent addition of the Weinreb amide (Formula V) allows for the formation of a stable tetrahedral intermediate, which upon aqueous workup collapses to release the desired ketone product (Formula I-2). The use of the Weinreb amide is critical here, as it prevents over-addition of the nucleophile, a common pitfall when reacting organolithiums with standard esters or acid chlorides. This mechanistic precision ensures that the carbonyl group is installed cleanly, setting the stage for the subsequent amidation without the need for further oxidation.
Following the coupling, the process relies on selective deprotection strategies to unveil the reactive amine necessary for the final assembly. The patent specifies that acidic conditions, such as treatment with hydrogen chloride in ethyl acetate or dioxane, are highly effective for removing tert-butoxycarbonyl (Boc) or benzyloxycarbonyl (Cbz) protecting groups. Alternatively, catalytic hydrogenolysis using palladium on carbon can be employed for Cbz removal, offering a orthogonal deprotection pathway that is gentle on the oxadiazole ring. This flexibility in deprotection chemistry is vital for impurity control, as it allows process chemists to tailor the conditions to the specific substituents present on the molecule, thereby minimizing the formation of degradation byproducts. The final amidation step couples the liberated amine with a pyrimidine carboxylic acid derivative (Formula IV), typically using mixed acid anhydrides or carbodiimide coupling agents like EDC. This final linkage forms the peptide-mimetic backbone essential for the biological activity of the resulting elastase inhibitor, completing the synthesis with high atom economy and structural integrity.
How to Synthesize 1,3,4-Oxadiazole Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical progression designed to maximize yield and minimize operational complexity. The process begins with the preparation of the key ketone intermediate via the lithiation-coupling sequence described above, followed by a straightforward deprotection to generate the free amine. This amine is then coupled with the pyrimidine fragment, and a final global deprotection yields the target compound. The detailed standardized synthetic steps, including specific reagent quantities, temperature profiles, and workup procedures derived from the patent examples, are outlined below to guide process implementation.
- Couple a Weinreb amide derivative (Formula V) with a lithiated 1,3,4-oxadiazole (Formula VI) using LDA and TMEDA at low temperatures (-78°C to 0°C) to form the protected intermediate (Formula I-2).
- Perform deprotection of the amino protecting group on Formula I-2 under acidic conditions (e.g., HCl/EtOAc) to obtain the free amine intermediate (Formula I-1).
- Conduct an amidation reaction between the free amine (Formula I-1) and a pyrimidine carboxylic acid derivative (Formula IV) using mixed acid anhydrides or condensing agents to form Formula III.
- Execute a final deprotection step, preferably via hydrogenolysis, to remove remaining protecting groups and yield the target 1,3,4-oxadiazole derivative (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN1350525A offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The reduction in synthetic steps from a double-digit count to merely four fundamentally alters the cost structure of manufacturing these intermediates. Fewer steps mean significantly lower consumption of solvents, reagents, and energy, which directly correlates to a substantial reduction in the cost of goods sold (COGS). Moreover, the elimination of hazardous oxidation steps like Swern oxidation reduces the need for specialized waste treatment and safety infrastructure, further driving down operational expenditures. This efficiency allows for a more competitive pricing strategy, enabling suppliers to offer high-purity pharmaceutical intermediates at a price point that supports the economic viability of the final drug product. The robustness of the route also implies a lower risk of batch failure, ensuring a more consistent supply of critical materials for downstream formulation.
- Cost Reduction in Manufacturing: The primary driver for cost reduction is the dramatic shortening of the synthetic route. By bypassing multiple protection and oxidation cycles, the process eliminates the associated material losses and labor costs inherent in long sequences. The high yield of 65% compared to the historical 18% means that significantly less starting material is required to produce the same amount of final product, effectively lowering the raw material intensity per unit. Additionally, the use of common industrial reagents like LDA and standard coupling agents avoids the need for exotic or prohibitively expensive catalysts, keeping input costs stable and predictable.
- Enhanced Supply Chain Reliability: A shorter, more robust synthesis translates directly into improved supply chain reliability. With fewer unit operations, the lead time for production batches is significantly compressed, allowing manufacturers to respond more agilely to fluctuations in demand. The simplified purification requirements reduce the bottleneck often caused by chromatography or complex crystallization steps in longer routes. This agility ensures that pharmaceutical partners can maintain continuous production schedules without the fear of raw material shortages caused by protracted synthesis times. Furthermore, the scalability of the reaction conditions, which utilize standard low-temperature reactors and filtration equipment, ensures that supply can be ramped up from pilot scale to commercial tonnage without requiring bespoke engineering solutions.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with modern green chemistry principles. The reduction in step count inherently reduces the E-factor (mass of waste per mass of product), minimizing the environmental burden of the manufacturing process. The avoidance of toxic oxidants and the use of recyclable solvents where possible contribute to a cleaner production profile, facilitating easier regulatory compliance and waste disposal. The process is designed to be scalable, with examples demonstrating successful execution on multi-kilogram scales, proving its readiness for commercial adoption. This scalability ensures that as the demand for elastase inhibitors grows, the supply of the intermediate can expand seamlessly to meet market needs without compromising on quality or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these 1,3,4-oxadiazole derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals seeking to implement this technology.
Q: What is the primary advantage of the process described in CN1350525A over prior art?
A: The primary advantage is a drastic reduction in synthetic steps from 6-10 steps in conventional methods to just 4 steps, resulting in a significantly higher total yield (65% vs 18%) and reduced production costs.
Q: What specific reaction conditions are critical for the key coupling step?
A: The coupling of the Weinreb amide with the oxadiazole requires strict temperature control between -78°C and 0°C, utilizing strong bases like LDA and ligands like TMEDA in inert solvents such as THF to ensure high selectivity.
Q: Is this process suitable for large-scale manufacturing of elastase inhibitors?
A: Yes, the process utilizes standard industrial reagents and avoids excessively hazardous conditions where possible, making it highly scalable for the commercial production of pharmaceutical intermediates like elastase inhibitors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4-Oxadiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the technology disclosed in CN1350525A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this high-yield process to life. We are committed to delivering high-purity 1,3,4-oxadiazole derivatives that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest industry standards. Our state-of-the-art facilities are equipped to handle the low-temperature reactions and sensitive intermediates involved in this synthesis, guaranteeing a reliable supply of material for your drug development programs.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this streamlined process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your path to market with superior pharmaceutical intermediates.
