Next-Generation Electron Transport Materials for High-Efficiency Red OLED Manufacturing
Introduction to Advanced Electroluminescent Material Technology
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that offer not only high quantum efficiency but also exceptional thermal and morphological stability. Patent CN1546477A introduces a groundbreaking class of electron transport and hole blocking materials based on non-branched conjugated oligomers containing oxadiazole groups. This technology addresses the critical bottleneck of charge imbalance in red electroluminescent devices by providing a robust alternative to traditional small molecules. By leveraging organometallic catalyzed reactions, specifically palladium-mediated cross-coupling, this innovation enables the synthesis of derivatives with significantly enhanced electron transport capabilities. For R&D directors and procurement specialists in the display sector, understanding the structural nuances and synthetic feasibility of these materials is paramount for securing a competitive edge in next-generation display manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on materials like 2-(4-biphenylyl)-5-(4-tert-butylphenyl)-1,3,4-oxadiazole (PBD) and 2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline (BCP) for electron transport and hole blocking functions. However, PBD suffers from a critically low melting point of approximately 138°C, which leads to easy recrystallization during the vacuum evaporation process, thereby destroying film homogeneity and causing rapid device degradation. While embedding PBD in an inert polymer matrix can suppress recrystallization to some extent, doping techniques often fail to guarantee long-term morphological stability. Similarly, while BCP is widely used, its stability within the device architecture remains a subject of concern for long-lifetime applications. These inherent physical limitations restrict the operational lifespan and efficiency of high-brightness red OLED panels, creating a pressing need for materials with higher glass transition temperatures and superior thermal resilience.
The Novel Approach
The patented approach circumvents these thermal instability issues by extending the conjugation length of the oxadiazole core through the attachment of bulky and rigid aryl groups. Instead of simple small molecules, the invention focuses on synthesizing unbranched conjugated oligomers that possess dramatically higher melting points, with specific derivatives recorded at 258.2°C, 259.3°C, and even reaching 376.3°C for fluorene-substituted variants. This substantial increase in thermal stability prevents the recrystallization phenomena observed in PBD, ensuring a uniform amorphous film state during device operation. Furthermore, the electron-deficient nature of the 1,3,4-oxadiazole ring is preserved and enhanced, maintaining low hole affinity which is essential for effective hole blocking. This structural modification results in a material that not only withstands the thermal stress of device fabrication but also actively improves charge carrier confinement within the luminescent layer.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core synthetic strategy employed in this patent utilizes the Suzuki-Miyaura cross-coupling reaction, a powerful tool for constructing carbon-carbon bonds between aryl halides and aryl boronic acids. The reaction mechanism involves a catalytic cycle mediated by tetrakis(triphenylphosphine)palladium(0), Pd(PPh3)4. Initially, the oxidative addition of the palladium catalyst to the carbon-bromine bond of the 2,5-bis(4-bromophenyl)-1,3,4-oxadiazole core generates an aryl-palladium(II) intermediate. This is followed by transmetallation with the aryl boronic acid species, which is activated by the base (typically potassium carbonate or sodium carbonate) present in the reaction mixture. Finally, reductive elimination releases the desired conjugated product and regenerates the active Pd(0) catalyst. This mechanism is highly tolerant of various functional groups, allowing for the introduction of diverse substituents such as trifluoromethyl, fluoro, and spiro-fluorene groups without compromising the integrity of the oxadiazole ring.
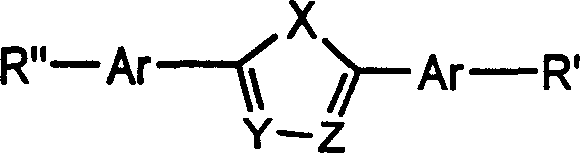
Structural analysis of the resulting compounds reveals that the choice of the aryl boronic acid directly influences the electronic and thermal properties of the final material. For instance, the incorporation of electron-withdrawing groups like trifluoromethyl or pentafluorophenyl can further lower the LUMO energy level, potentially enhancing electron injection efficiency. Conversely, bulky groups like spiro-fluorene introduce steric hindrance that inhibits close packing of molecules, thereby suppressing crystallization and promoting the formation of stable amorphous films. The patent details specific molar ratios, recommending a boronic acid to bromo-derivative ratio of 2.0 to 3.0:1 to drive the reaction to completion and minimize the presence of mono-substituted impurities. This precise control over stoichiometry and catalytic conditions is crucial for achieving the high purity levels required for electronic grade materials, where trace metal residues or structural defects can act as quenching sites for excitons.
How to Synthesize High-Purity Oxadiazole Derivatives Efficiently
The synthesis of these advanced electron transport materials requires a meticulous approach to reaction conditions and purification protocols to ensure suitability for vacuum thermal evaporation. The process begins with the preparation of the key intermediate, 2,5-bis(4-bromophenyl)-1,3,4-oxadiazole, which serves as the modular building block for all subsequent derivatives. Following the coupling reaction, the crude products typically undergo rigorous purification, either through silica gel column chromatography or recrystallization from appropriate solvents, to remove palladium residues and unreacted starting materials. The detailed standardized synthesis steps for producing these high-performance OLED intermediates are outlined below.
- Preparation of the 2,5-bis(4-bromophenyl)-1,3,4-oxadiazole core module through cyclodehydration of diacylhydrazines using phosphorus oxychloride.
- Execution of the Suzuki-Miyaura cross-coupling reaction between the brominated oxadiazole core and various aryl boronic acids using Pd(PPh3)4 catalyst.
- Purification of the final conjugated oligomers via column chromatography or recrystallization to achieve electronic-grade purity suitable for vacuum evaporation.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and cost perspective, the adoption of this synthetic route offers significant strategic advantages over the procurement of legacy materials like BCP or unstable PBD derivatives. The reliance on the Suzuki coupling reaction utilizes widely available and commercially scalable reagents, such as aryl boronic acids and palladium catalysts, which are staples in the fine chemical industry. This ensures a robust and continuous supply chain, reducing the risk of raw material shortages that often plague specialty chemical markets. Furthermore, the high yields reported in the patent examples, such as 98% for the core intermediate and up to 85% for specific derivatives, indicate a highly efficient process that minimizes waste and maximizes output per batch. This efficiency translates directly into better cost structures for large-scale manufacturing.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for complex multi-step sequences often required to build the oxadiazole ring from scratch for every derivative, as the core module is pre-synthesized and reused. By utilizing a convergent synthesis strategy where diverse functionalities are introduced in a single coupling step, the overall process time and labor costs are drastically simplified. Additionally, the ability to purify products via recrystallization rather than exclusively relying on expensive chromatography for all batches suggests a viable path towards ton-scale production with reduced solvent consumption and waste disposal costs. The removal of transition metal catalysts, while standard, is streamlined by the robust nature of the reaction, allowing for efficient scavenging methods that meet stringent electronic material specifications without excessive processing.
- Enhanced Supply Chain Reliability: The starting materials, including 4-bromobenzoic acid derivatives and various substituted phenylboronic acids, are commodity chemicals produced by multiple global suppliers, ensuring that the supply chain is not dependent on a single source. This diversification of raw material sources mitigates the risk of supply disruptions and price volatility. Moreover, the reaction conditions described, such as refluxing in toluene or THF, are standard industrial processes that do not require exotic high-pressure or cryogenic equipment, making it easier for contract manufacturing organizations (CMOs) to adopt the technology quickly. This ease of technology transfer ensures that production capacity can be scaled up rapidly to meet surging demand from the display industry.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, as evidenced by the successful synthesis of various derivatives with consistent quality. The use of palladium catalysts, while requiring recovery to meet cost and environmental goals, is a well-established practice in the pharmaceutical and fine chemical sectors, with mature technologies available for metal scavenging and recycling. The high thermal stability of the final products also implies a longer operational life for the end-user devices, which indirectly contributes to sustainability by reducing electronic waste. Furthermore, the avoidance of unstable materials that degrade quickly reduces the failure rate in downstream device manufacturing, optimizing the overall yield of the OLED panel production line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these oxadiazole-based materials in industrial settings. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and synthesis feasibility. Understanding these details is essential for technical teams evaluating the integration of new materials into existing device architectures.
Q: What are the thermal stability advantages of these new oxadiazole derivatives compared to PBD?
A: Unlike traditional PBD which has a low melting point of 138°C and suffers from recrystallization, these new conjugated oligomers exhibit significantly elevated melting points, ranging from 215°C to over 376°C, ensuring superior film homogeneity and device longevity.
Q: Can these materials replace BCP in red electroluminescent devices?
A: Yes, experimental data indicates that devices utilizing these materials demonstrate satisfactory results in brightness and current density, with comprehensive performance superior to the widely used BCP (2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline) electron transport/hole blocking layers.
Q: What is the primary synthetic methodology used for these intermediates?
A: The core synthesis relies on organometallic catalyzed reactions, specifically the Suzuki coupling between 2,5-bis(4-bromophenyl)-1,3,4-oxadiazole and substituted aryl boronic acids under alkaline conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxadiazole Derivatives Supplier
As the demand for high-efficiency red OLED materials continues to grow, partnering with an experienced chemical manufacturer is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of electron transport material meets the exacting standards required for display manufacturing. We understand the nuances of organometallic chemistry and are fully capable of optimizing the Suzuki coupling conditions to maximize yield and minimize metal residuals.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant operational efficiencies. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for these high-performance oxadiazole derivatives. Let us help you secure a stable supply of next-generation OLED materials that drive superior device performance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
