Advanced Synthesis of 1,3,4-Oxadiazole Derivatives for Commercial ATR Inhibitor Production
The pharmaceutical landscape for oncology treatments is continuously evolving, driven by the urgent need for more effective and safer therapeutic agents. Patent CN113354631B represents a significant technological breakthrough in the field of anticancer drug development, specifically disclosing a novel 1,3,4-oxadiazole derivative designed to function as a potent Ataxia Telangiectasia and Rad3-related (ATR) kinase inhibitor. This intellectual property outlines not only the chemical structure of the derivative but also details a sophisticated and environmentally benign preparation method that addresses long-standing challenges in heterocyclic synthesis. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this patent offers a compelling value proposition by introducing a synthetic route that avoids the pitfalls of traditional high-temperature and high-corrosion processes. The core innovation lies in the strategic application of dibromotriphenylphosphine as a dehydrating cyclization agent, which enables the formation of the critical oxadiazole ring under mild, room-temperature conditions. This shift away from aggressive reagents like phosphorus oxychloride or thionyl chloride marks a pivotal advancement in green chemistry within the fine chemical sector, promising enhanced safety profiles and simplified operational workflows for industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3,4-oxadiazole derivatives has been plagued by significant operational and safety hurdles that complicate both laboratory research and industrial production. The most prevalent traditional technique, known as the double hydrazide ring closure method, typically necessitates the use of aggressive dehydrating agents such as phosphorus oxychloride, phosphorus pentachloride, concentrated sulfuric acid, or polyphosphoric acid. These reagents are not only highly toxic and corrosive but also require elevated reflux temperatures to drive the cyclization reaction to completion, creating severe safety risks for personnel and demanding specialized, corrosion-resistant reactor infrastructure. Furthermore, the disposal of waste streams containing these hazardous chemicals poses a substantial environmental burden, increasing the overall cost of compliance and waste management for manufacturers. The harsh conditions often lead to the formation of complex impurity profiles, necessitating rigorous and costly purification steps to achieve the stringent purity specifications required for pharmaceutical applications. Consequently, the reliance on these conventional methods has often resulted in extended lead times for high-purity pharmaceutical intermediates and limited the scalability of potential drug candidates due to safety and engineering constraints.
The Novel Approach
In stark contrast to these archaic methodologies, the approach detailed in patent CN113354631B introduces a paradigm shift by utilizing dibromotriphenylphosphine as a mild yet highly effective dehydrating cyclization agent. This innovative reagent allows the critical ring-closing reaction to proceed efficiently at room temperature, thereby eliminating the need for energy-intensive heating and mitigating the risks associated with thermal runaway or pressure buildup. The mildness of the reaction conditions preserves the integrity of sensitive functional groups, such as the nitro and sulfonyl moieties present in the substrate, which are crucial for the biological activity of the final ATR inhibitor. By operating under ambient conditions, the process significantly reduces the corrosion load on manufacturing equipment, extending asset life and lowering maintenance costs. Moreover, the post-reaction workup is remarkably straightforward, involving simple filtration and washing steps that facilitate the isolation of the target intermediate with high efficiency. This streamlined process not only enhances the overall yield but also aligns with modern principles of sustainable manufacturing, making it an ideal candidate for cost reduction in API manufacturing where operational efficiency and environmental compliance are paramount.
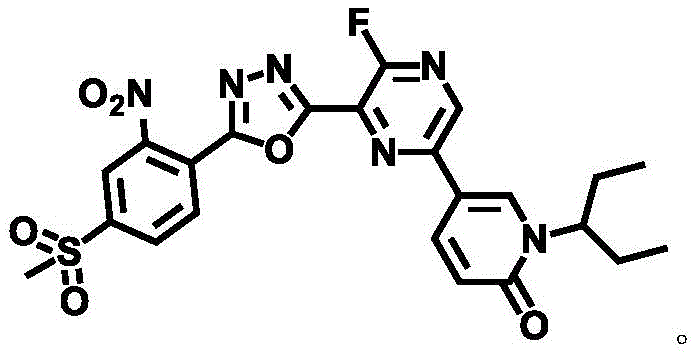
Mechanistic Insights into Dibromotriphenylphosphine-Mediated Cyclization
The mechanistic elegance of this synthesis lies in the activation of the carboxylic acid functionality by the phosphine reagent, which facilitates the nucleophilic attack by the hydrazide nitrogen without the need for extreme thermal energy. In the critical Step c of the synthesis, dibromotriphenylphosphine reacts with the carbohydrazide intermediate to form a reactive acyl phosphonium species, which is subsequently attacked by the adjacent nitrogen atom to close the oxadiazole ring. This pathway avoids the generation of hazardous gaseous byproducts often associated with thionyl chloride or oxalyl chloride methods, such as SO2 or CO, thereby improving the safety profile of the reaction vessel headspace. The presence of the base, N,N-diisopropylethylamine (DIEA), serves to neutralize the hydrogen bromide generated during the process, driving the equilibrium towards product formation and preventing acid-catalyzed degradation of the sensitive pyrazine core. This controlled environment ensures that the electron-withdrawing nitro group on the benzene ring remains intact, preserving the electronic properties necessary for the molecule to act as a potent ATR inhibitor. The specificity of this mechanism minimizes side reactions, resulting in a cleaner crude product that requires less intensive chromatographic purification, which is a significant advantage when scaling up complex pharmaceutical intermediates.
From an impurity control perspective, the room-temperature operation is instrumental in suppressing the formation of thermal degradation products that often contaminate batches produced via traditional reflux methods. The avoidance of strong mineral acids prevents the hydrolysis of the ester or amide linkages that might otherwise occur under harsh acidic conditions, ensuring a higher degree of chemical fidelity in the final structure. Additionally, the use of anhydrous acetonitrile as the solvent provides a polar aprotic environment that stabilizes the transition states involved in the cyclization while remaining easy to remove during the concentration phase. The subsequent washing steps with n-hexane and acetonitrile effectively remove triphenylphosphine oxide and other phosphorous-containing byproducts, which are common contaminants in phosphine-mediated reactions. This rigorous control over the impurity profile is essential for meeting the regulatory standards set by health authorities for clinical-grade materials, thereby reducing the risk of batch rejection and ensuring a consistent supply of high-purity 1,3,4-oxadiazole derivatives for downstream drug development.
How to Synthesize 1,3,4-Oxadiazole Derivative Efficiently
The synthesis of this advanced ATR inhibitor intermediate involves a convergent strategy that combines robust heterocyclic chemistry with precise functional group manipulation to ensure high fidelity and yield. The process begins with the diazotization and fluorination of a pyrazine precursor, followed by hydrazinolysis to generate the key carbohydrazide building block. The heart of the synthesis is the aforementioned room-temperature cyclization, which constructs the oxadiazole core, followed by parallel synthesis of the pyridone fragment via alkylation and borylation. These two fragments are finally united through a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction to deliver the target molecule. Each step has been optimized to balance reaction kinetics with product stability, ensuring that the sensitive nitro and sulfonyl groups are preserved throughout the sequence. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification protocols, are outlined in the guide below to assist process chemists in replicating this efficient route.
- Perform diazotization of methyl 3-amino-6-bromopyrazine-2-carboxylate followed by thermal decomposition to introduce the fluorine atom.
- Conduct hydrazinolysis with hydrazine hydrate to form the carbohydrazide intermediate.
- Execute the key cyclization using dibromotriphenylphosphine at room temperature to form the oxadiazole ring.
- Complete the synthesis via alkylation, borylation, and final Suzuki coupling to attach the pyridone moiety.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic advantages that extend beyond mere chemical curiosity. The elimination of highly corrosive and toxic reagents fundamentally alters the cost structure of production by removing the need for specialized Hastelloy reactors or glass-lined vessels that are prone to rapid degradation. This shift allows for the utilization of more standard stainless steel equipment, significantly lowering capital expenditure requirements for new production lines and reducing the frequency of maintenance shutdowns. Furthermore, the simplified workup procedure, which relies on filtration and washing rather than complex aqueous extractions or distillations, reduces the consumption of solvents and utilities, leading to substantial cost savings in operational expenditures. The ability to run the critical cyclization step at room temperature also enhances energy efficiency, contributing to a lower carbon footprint and aligning with corporate sustainability goals that are increasingly important in global supply chains. These factors collectively enhance supply chain reliability by minimizing the risk of production delays caused by equipment failure or safety incidents, ensuring a steady flow of materials for clinical and commercial needs.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous dehydrating agents with dibromotriphenylphosphine eliminates the downstream costs associated with neutralizing and disposing of large volumes of acidic waste. By avoiding the use of reagents like phosphorus oxychloride, manufacturers save significantly on waste treatment fees and reduce the environmental compliance burden. The higher selectivity of the room-temperature reaction minimizes the formation of difficult-to-remove impurities, which reduces the loss of material during purification and improves the overall mass balance of the process. This efficiency gain directly impacts the cost of goods sold, making the final API more competitive in the marketplace while maintaining healthy margins for the supplier.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as dibromotriphenylphosphine ensures that the supply chain is not vulnerable to the volatility often seen with specialized or highly regulated hazardous chemicals. The mild reaction conditions reduce the likelihood of batch failures due to thermal excursions or pressure issues, leading to more predictable production schedules and shorter lead times for customers. Additionally, the robustness of the synthesis allows for greater flexibility in sourcing raw materials, as the process is less sensitive to minor variations in reagent quality compared to high-temperature reflux methods. This resilience is critical for maintaining continuity of supply in the face of global logistical challenges, ensuring that pharmaceutical partners can rely on consistent delivery of key intermediates.
- Scalability and Environmental Compliance: The inherent safety of the room-temperature cyclization step makes this process exceptionally well-suited for scale-up from kilogram to multi-ton production scales without the need for complex engineering controls. The absence of exothermic spikes or gas evolution simplifies the design of large-scale reactors, reducing the time and cost required for process validation and technology transfer. From an environmental perspective, the reduction in hazardous waste generation and energy consumption supports compliance with increasingly stringent global environmental regulations, such as REACH and TSCA. This proactive approach to green chemistry not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible and sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel 1,3,4-oxadiazole derivative. These insights are derived directly from the technical specifications and experimental data provided in patent CN113354631B, offering clarity on the process capabilities and biological potential of the compound. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their drug development pipelines or supply networks.
Q: How does this synthesis method improve upon traditional 1,3,4-oxadiazole preparation?
A: Traditional methods often require harsh dehydrating agents like phosphorus oxychloride or concentrated sulfuric acid at high reflux temperatures, leading to corrosion and safety hazards. This patented method utilizes dibromotriphenylphosphine at room temperature, significantly reducing equipment corrosion risks and simplifying post-reaction processing.
Q: What is the biological significance of the nitro group in this derivative?
A: The electron-withdrawing nitro group on the side chain benzene ring is structurally critical as it enhances the antitumor activity of the derivative. This specific substitution pattern contributes to the compound's efficacy as an ATR inhibitor, particularly in synergistic applications with chemotherapeutic agents like doxorubicin.
Q: Can this intermediate be scaled for commercial API production?
A: Yes, the elimination of high-temperature reflux conditions and highly corrosive reagents makes this route highly amenable to scale-up. The use of milder conditions and standard workup procedures like filtration and silica gel purification supports robust commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4-Oxadiazole Derivative Supplier
As the demand for targeted oncology therapies continues to surge, the ability to source high-quality ATR inhibitor intermediates with consistent purity and reliable availability has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring complex molecules like this 1,3,4-oxadiazole derivative to market. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of phosphine reagents and the execution of sensitive palladium-catalyzed couplings under strict inert atmospheres. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for clinical trials and eventual commercial launch, providing our partners with the confidence they need to advance their pipelines.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this innovative synthetic route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall manufacturing costs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will validate the suitability of this intermediate for your next-generation anticancer drug projects. Let us be your trusted partner in transforming cutting-edge patent technology into life-saving medicines.
