Advanced Manufacturing of Rivastigmine Intermediates via Optimized Leuckart Reaction Pathways
The global demand for effective treatments for neurodegenerative disorders, particularly Alzheimer's disease, has placed immense pressure on the pharmaceutical supply chain to deliver high-quality active pharmaceutical ingredients (APIs) with consistent reliability. Rivastigmine, a potent carbamate acetylcholinesterase inhibitor, stands as a cornerstone therapy in this sector, necessitating robust and efficient manufacturing processes to meet market needs. Patent CN103664702A introduces a transformative production process that fundamentally restructures the synthetic pathway for Rivastigmine, moving away from cumbersome multi-step reductions toward a streamlined Leuckart reaction methodology. This technical breakthrough addresses critical bottlenecks in traditional manufacturing, specifically targeting yield enhancement and cost efficiency without compromising the stringent purity standards required for central nervous system medications. By leveraging a direct amination strategy, this innovation offers a compelling value proposition for stakeholders seeking to optimize their supply chains for complex neurological therapeutics.
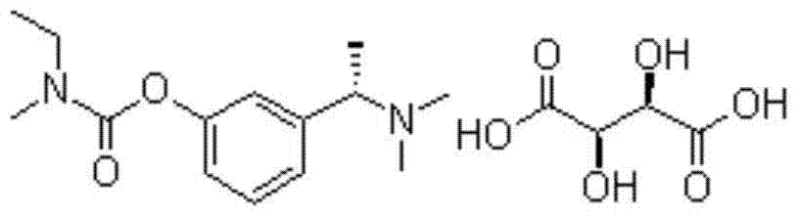
For R&D directors and process chemists, understanding the structural integrity and synthetic accessibility of the target molecule is paramount. The chemical architecture of Rivastigmine Tartrate, as depicted in the structural analysis, relies heavily on the precise stereochemistry of the ethyl-methyl-amino ethyl side chain attached to the phenolic core. Any deviation in the synthetic route can lead to racemization or the formation of structurally related impurities that are difficult to remove. The patented process acknowledges these challenges by implementing a controlled environment for the formation of the chiral center, ensuring that the downstream resolution steps are not overwhelmed by excessive diastereomeric impurities. This focus on molecular precision from the earliest synthetic stages underscores the sophistication of the proposed methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Rivastigmine intermediates has relied heavily on the formation of oximes followed by catalytic or chemical reduction to generate the necessary amine functionality. This traditional pathway is fraught with inefficiencies, primarily stemming from the requirement for multiple discrete reaction vessels and isolation procedures. The formation of the oxime intermediate typically involves the use of hydroxylamine hydrochloride, a reagent that poses significant safety hazards and generates substantial aqueous waste streams requiring specialized treatment. Furthermore, the subsequent reduction step often suffers from incomplete conversion or over-reduction, leading to a complex crude reaction mixture that demands extensive purification efforts. These cumulative inefficiencies result in lower overall yields, often exacerbated by mechanical losses during the filtration and drying of unstable intermediates, ultimately driving up the cost of goods sold for the final API.
The Novel Approach
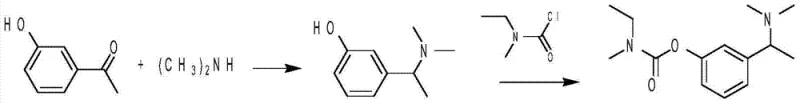
In stark contrast, the novel approach detailed in the patent utilizes a direct Leuckart reaction to construct the critical carbon-nitrogen bond in a single operational step. By reacting m-hydroxyacetophenone directly with dimethylamine in the presence of formic acid, the process achieves both imine formation and reduction simultaneously, effectively bypassing the need for isolated oxime intermediates. This telescoping of reactions not only reduces the total number of processing steps but also significantly minimizes the exposure of reactive intermediates to potential degradation pathways. The subsequent carbamate formation is executed under mild conditions using ethylmethyl-carbamic chloride, ensuring high conversion rates with minimal side reactions. This streamlined workflow represents a paradigm shift in process chemistry, prioritizing atom economy and operational simplicity to deliver a more sustainable manufacturing route.
Mechanistic Insights into Leuckart-Catalyzed Amination and Carbamate Coupling
The core of this technological advancement lies in the mechanistic elegance of the modified Leuckart reaction employed in the first step. Unlike standard reductive amination which might utilize expensive metal catalysts or high-pressure hydrogenation, this process leverages formic acid as both the solvent and the hydride source. The reaction proceeds through the initial formation of an iminium ion intermediate, which is subsequently reduced by the formate anion via a concerted hydride transfer mechanism. The inclusion of magnesium chloride acts as a crucial Lewis acid catalyst, coordinating with the carbonyl oxygen of the acetophenone to enhance its electrophilicity, thereby accelerating the rate of imine formation. This catalytic effect allows the reaction to proceed efficiently at elevated temperatures between 160°C and 180°C, ensuring complete conversion of the starting ketone while maintaining the integrity of the phenolic hydroxyl group.
Following the successful construction of the amine scaffold, the second stage involves the nucleophilic attack of the phenolic oxygen on the carbonyl carbon of ethylmethyl-carbamic chloride. This step is facilitated by the use of sodium hydride in tetrahydrofuran (THF), which serves to deprotonate the phenol, generating a highly reactive phenoxide anion in situ. The choice of THF as a solvent is strategic, providing excellent solubility for both the organic substrate and the inorganic base, thus ensuring homogeneous reaction conditions that promote rapid kinetics. The resulting carbamate linkage is thermodynamically stable, yet the reaction conditions are mild enough to prevent elimination side reactions or racemization of the adjacent chiral center. This precise control over reaction parameters is essential for maintaining the high optical purity required for the final pharmaceutical product.
How to Synthesize Rivastigmine Efficiently
Implementing this synthesis route requires careful attention to reaction stoichiometry and thermal management to maximize the benefits of the Leuckart transformation. The process begins with the preparation of the formic acid-dimethylamine complex, which must be handled with care due to the exothermic nature of salt formation. Subsequent addition of the ketone substrate and the magnesium chloride catalyst initiates the amination sequence, where temperature control is critical to drive the equilibrium towards the desired amine product. Following the workup and isolation of the intermediate phenol, the carbamate coupling step demands anhydrous conditions to prevent hydrolysis of the acid chloride reagent. The detailed standardized synthesis steps for executing this protocol are outlined in the guide below.
- Perform a one-step Leuckart reaction between m-hydroxyacetophenone and dimethylamine in the presence of formic acid and magnesium chloride at elevated temperatures to synthesize 3-(1-(dimethylamino) ethyl) phenol.
- Conduct a condensation reaction between the resulting phenol intermediate and ethylmethyl-carbamic chloride in THF using sodium hydride as a base to form the rivastigmine free base.
- Purify the racemic mixture through resolution using D-(+)-di-p-toluoyl tartaric acid (DTTA) to obtain the optically active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The reduction in process steps directly correlates to a decrease in capital expenditure on equipment utilization time and labor costs, as fewer batch cycles are required to produce the same quantity of finished goods. Moreover, the elimination of hazardous reagents like hydroxylamine simplifies regulatory compliance and reduces the overhead associated with waste disposal and environmental safety monitoring. These operational efficiencies translate into a more resilient supply chain capable of responding rapidly to fluctuations in market demand for Alzheimer's therapeutics.
- Cost Reduction in Manufacturing: The streamlined nature of the Leuckart reaction pathway inherently lowers the cost of production by minimizing raw material consumption and energy usage. By avoiding the multi-step oxime reduction sequence, manufacturers eliminate the costs associated with purchasing additional reagents and solvents required for intermediate isolations. The high recovery rate of unreacted starting materials, as demonstrated in the patent examples, further contributes to cost optimization by allowing for the recycling of valuable precursors back into the process. This circular approach to material usage significantly enhances the overall economic viability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as m-hydroxyacetophenone and dimethylamine ensures a stable and secure supply of raw materials, mitigating the risk of shortages that often plague specialized reagent markets. The robustness of the reaction conditions, which do not require exotic catalysts or extreme pressures, means that production can be easily distributed across multiple manufacturing sites without significant technology transfer barriers. This geographic flexibility strengthens the supply chain against regional disruptions and ensures continuous availability of the critical API intermediate for downstream formulation.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates significantly less waste compared to traditional methods, aligning with modern green chemistry principles and corporate sustainability goals. The simplified workup procedures reduce the volume of aqueous and organic waste streams, lowering the burden on effluent treatment plants. Furthermore, the scalability of the high-temperature Leuckart reaction is well-established in the fine chemical industry, allowing for seamless transition from pilot plant trials to full-scale commercial production ranging from hundreds of kilograms to multi-ton annual capacities without loss of efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Rivastigmine synthesis process. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential manufacturing partners. Understanding these nuances is crucial for making informed decisions about technology adoption and supply chain integration.
Q: What are the primary advantages of the Leuckart reaction route over traditional oxime reduction for Rivastigmine?
A: The Leuckart reaction route significantly simplifies the synthetic pathway by combining amination and reduction into a single operational step. Unlike the traditional method which requires separate oxime formation and subsequent reduction stages, this novel approach eliminates intermediate isolation steps, thereby reducing mechanical losses and improving overall process yield. Furthermore, it avoids the use of hazardous hydroxylamine salts often required in oxime synthesis, enhancing operational safety.
Q: How does the new process impact the purity profile of the final API intermediate?
A: By minimizing the number of unit operations and utilizing a highly selective condensation step for the carbamate formation, the process inherently reduces the generation of complex side-product impurities. The implementation of specific crystallization protocols using chiral resolving agents like DTTA ensures that the enantiomeric excess is maintained at stringent levels required for CNS active pharmaceutical ingredients, resulting in a cleaner impurity profile compared to multi-step reduction routes.
Q: Is this synthesis method scalable for commercial production volumes?
A: Yes, the process is specifically designed with industrial scalability in mind. The reaction conditions, such as the use of common solvents like THF and ether, along with robust temperature parameters that do not require cryogenic cooling, make it highly adaptable for large-scale reactor systems. The high recovery rate of unreacted starting materials further supports economic viability at the metric ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivastigmine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex neurological drugs depends on a partnership grounded in technical excellence and supply chain reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to market-ready reality. We are committed to delivering high-purity Rivastigmine intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch conforms to the highest international standards, providing you with the confidence needed to navigate the competitive pharmaceutical landscape.
We invite you to collaborate with us to leverage this innovative synthesis technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
