Advanced Synthesis of Rivastigmine Intermediates via Reductive Amination for Commercial Scale-Up
Advanced Synthesis of Rivastigmine Intermediates via Reductive Amination for Commercial Scale-Up
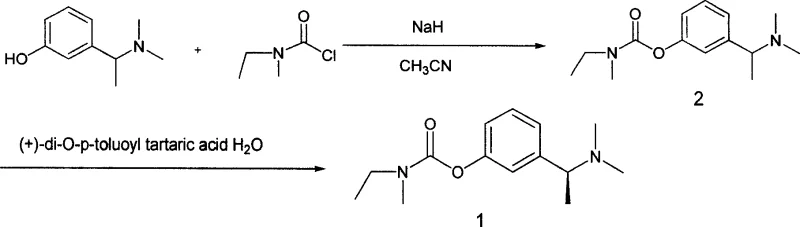
The pharmaceutical landscape for Alzheimer's disease treatments continues to evolve, driven by the need for more efficient and cost-effective manufacturing processes for key active ingredients. Patent CN101016257A introduces a significant technological breakthrough in the synthesis of Rivastigmine, a potent carbamate acetylcholinesterase inhibitor. This patent discloses a novel intermediate, N,N-ethyl-methyl-carbamate 3-(1-methylamino ethyl) phenyl ester, and a robust preparation method that fundamentally shifts the synthetic strategy from traditional phenol-based carbamoylation to a ketone-based reductive amination pathway. By prioritizing the construction of the chiral amine center prior to final methylation, this approach addresses critical bottlenecks in yield and enantiomeric purity that have long plagued conventional manufacturing lines. For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials at scale.
This innovative route leverages titanium isopropylate-catalyzed reductive amination followed by optical resolution, offering a distinct advantage in process control. The methodology ensures that the stereochemical integrity of the molecule is established early and maintained through subsequent transformations, thereby reducing the burden on downstream purification steps. As the demand for CNS therapeutics grows, the ability to produce complex chiral amines with high efficiency becomes a strategic asset. This report analyzes the technical merits of patent CN101016257A, highlighting its potential to drive cost reduction in API manufacturing while ensuring the stringent quality standards required for neurodegenerative disease medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Rivastigmine has relied on pathways that introduce the carbamate functionality at an early stage, often reacting a phenolic precursor with N-ethyl-N-methylcarbamoyl chloride. As illustrated in the prior art, this conventional approach typically generates a racemic mixture that requires extensive and repetitive recrystallization to achieve the necessary enantiomeric excess (ee value). The reliance on multiple crystallization steps not only drastically reduces the overall recovery rate but also introduces significant variability in product quality. Furthermore, handling reactive carbamoyl chlorides on a large scale presents safety challenges and requires rigorous moisture control, complicating the operational workflow. These inefficiencies result in higher production costs and longer lead times, creating vulnerabilities in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN101016257A reverses the synthetic logic by constructing the amine side chain via reductive amination of a ketone precursor, specifically N,N-ethyl-methyl-carbamate 3-acetyl phenyl ester. This strategy allows for the formation of the critical carbon-nitrogen bond under mild conditions using sodium borohydride and titanium isopropylate, avoiding the harsh reagents associated with traditional methods. The resulting racemic amine intermediate is then subjected to optical resolution using readily available chiral acids, such as di-O-p-toluoyl tartaric acid, to isolate the desired S-enantiomer with high fidelity. This decoupling of the carbamate formation from the chiral resolution step simplifies the purification profile and significantly enhances the total yield. By streamlining the process flow, manufacturers can achieve commercial scale-up of complex chiral amines with greater operational safety and economic efficiency.
Mechanistic Insights into Titanium-Catalyzed Reductive Amination
The core of this novel synthesis lies in the efficient conversion of the acetyl ketone group into a methylamino ethyl group through a titanium-mediated reductive amination mechanism. In this process, titanium isopropylate acts as a Lewis acid catalyst, facilitating the condensation of the ketone with methylamine to form an imine or iminium intermediate in situ. This activation lowers the energy barrier for the subsequent reduction by sodium borohydride, ensuring rapid and selective conversion to the secondary amine without affecting the sensitive carbamate moiety elsewhere in the molecule. The use of titanium isopropylate is particularly advantageous as it coordinates with the carbonyl oxygen, directing the hydride attack and minimizing side reactions such as over-reduction or ester hydrolysis. This precise control over chemoselectivity is crucial for maintaining the structural integrity of the intermediate and preventing the formation of difficult-to-remove impurities.
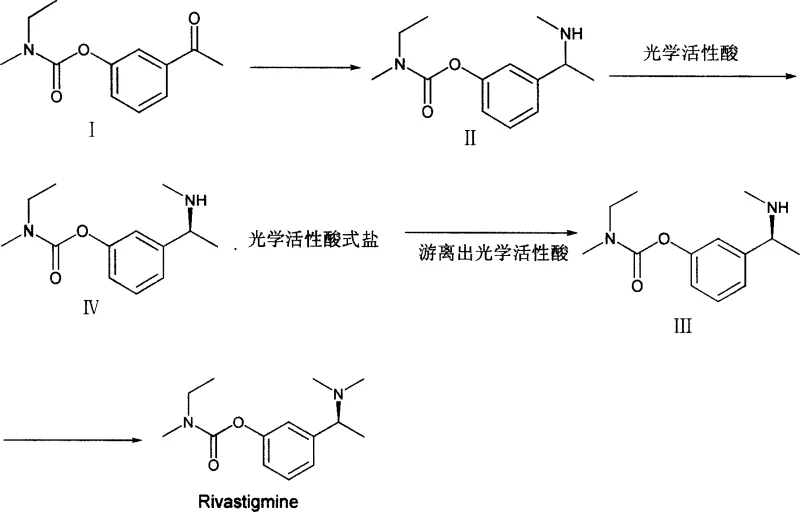
Following the reductive amination, the process employs a classical resolution strategy to secure the required stereochemistry. The racemic amine is reacted with an optically active acid to form a diastereomeric salt, which exhibits different solubility properties allowing for the selective crystallization of the desired S-enantiomer salt. The patent highlights the versatility of this step, noting that various chiral acids including tartaric acid derivatives and camphorsulfonic acid can be effectively utilized. Once the pure salt is isolated, it is treated with a base to liberate the free amine, yielding the optically pure intermediate ready for final methylation. This robust resolution protocol ensures that the final API meets the rigorous enantiomeric purity specifications demanded by regulatory bodies, thereby reducing lead time for high-purity pharmaceutical intermediates by minimizing the need for corrective processing.
How to Synthesize Rivastigmine Intermediate Efficiently
The synthesis of the key Rivastigmine intermediate described in this patent offers a practical and scalable pathway for industrial production. The process begins with the reductive amination of the ketone precursor, followed by chiral resolution and final methylation. Each step is optimized for high yield and ease of operation, utilizing common laboratory reagents that are readily available in bulk quantities. The detailed procedure involves dissolving the starting ketone in a methylamine solution, adding titanium isopropylate, and subsequently reducing with sodium borohydride under controlled temperatures. For the complete standardized operating procedures, safety data, and specific stoichiometric ratios required for GMP manufacturing, please refer to the technical guide below.
- Perform reductive amination on N,N-ethyl-methyl-carbamate 3-acetyl phenyl ester using titanium isopropylate and sodium borohydride.
- Resolve the racemic amine intermediate using optically active acids such as di-O-p-toluoyl tartaric acid to isolate the S-enantiomer.
- Execute final methylation via Eschweiler-Clarke reaction or reductive methylation to obtain Rivastigmine.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthetic route outlined in CN101016257A presents substantial strategic benefits for procurement managers and supply chain leaders focused on optimizing the cost structure of Alzheimer's drug production. By shifting to a reductive amination pathway, manufacturers can eliminate the need for expensive and hazardous carbamoyl chloride reagents in the early stages, replacing them with more stable and cost-effective ketone precursors. This change not only lowers raw material costs but also simplifies the handling requirements, reducing the need for specialized containment equipment and lowering operational expenditures. Furthermore, the improved yield profile of this method means that less starting material is required to produce the same amount of final API, directly contributing to significant cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The elimination of multiple recrystallization steps required in conventional methods drastically reduces solvent consumption and processing time. By achieving high crude yields in the reductive amination step, the process minimizes material loss, leading to a more economical production cycle. Additionally, the use of common reducing agents like sodium borohydride instead of precious metal catalysts ensures that the process remains affordable and accessible for large-scale operations without compromising on efficiency.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as titanium isopropylate and methylamine ensures a stable supply of raw materials, mitigating the risk of shortages that can disrupt production schedules. The robustness of the resolution step, which tolerates a variety of chiral acids, provides flexibility in sourcing resolving agents, further strengthening supply chain resilience. This adaptability allows manufacturers to maintain continuous production even when specific reagent markets experience volatility, ensuring consistent delivery of critical intermediates to downstream API producers.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup procedures associated with this method facilitate easier scale-up from pilot plant to commercial tonnage. The reduction in hazardous waste generation, particularly from avoiding excessive halogenated solvents and reactive chlorides, aligns with modern environmental regulations and sustainability goals. This greener profile not only reduces waste disposal costs but also enhances the corporate social responsibility standing of the manufacturing facility, making it a preferred partner for environmentally conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Rivastigmine intermediate synthesis. These insights are derived directly from the experimental data and claims presented in patent CN101016257A, providing a factual basis for evaluating the technology's viability. Understanding these details is essential for technical teams assessing the feasibility of integrating this route into existing production facilities.
Q: What is the primary advantage of the reductive amination route over conventional carbamoylation?
A: The reductive amination route avoids the harsh conditions and low yields associated with early-stage carbamoylation, allowing for better impurity control and higher overall throughput.
Q: Which resolving agents are effective for isolating the S-enantiomer in this process?
A: The patent specifies several effective resolving agents including (+)-di-O-p-toluoyl tartaric acid, D-tartaric acid, and D-amydalic acid, offering flexibility in sourcing.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes common reagents like sodium borohydride and titanium isopropylate under mild conditions, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivastigmine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of CNS drug manufacturing. Our team of expert chemists has extensively evaluated the reductive amination strategy described in CN101016257A and possesses the technical capability to implement this process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Rivastigmine intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate the delivery of life-saving medications to patients worldwide.
