Advanced Purification Technology for Cefodizime Sodium: Enhancing Quality and Commercial Viability
Advanced Purification Technology for Cefodizime Sodium: Enhancing Quality and Commercial Viability
The pharmaceutical landscape demands increasingly rigorous standards for antibiotic purity, particularly for third-generation cephalosporins like Cefodizime Sodium. Patent CN102010432B introduces a transformative approach to refining this critical compound, addressing long-standing challenges in impurity removal and product stability. This technology leverages a sophisticated combination of acid-base salification, activated carbon adsorption, and precise chromatographic separation to achieve purity levels exceeding 99.5%. For R&D directors and procurement specialists, understanding this methodology is essential for securing a reliable cefodizime sodium supplier capable of meeting global regulatory requirements. The structural integrity of the molecule, characterized by its unique thiazole rings and methoxyimino group, requires gentle yet effective processing to maintain immunoregulatory activity while eliminating toxic byproducts.
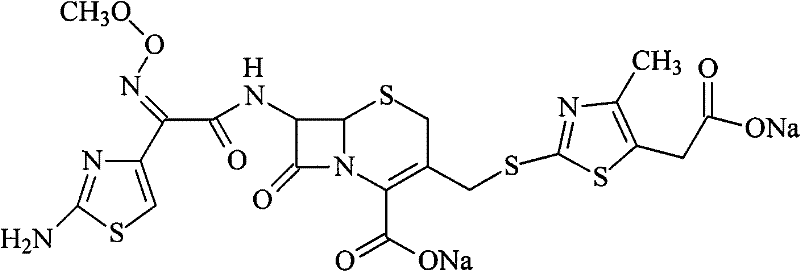
The innovation lies not just in the synthesis but in the downstream processing, which dictates the final quality of the API. By optimizing parameters such as pH control during precipitation and selecting specific stationary phases for chromatography, this method overcomes the limitations of earlier techniques that often resulted in dark-colored products or residual solvent issues. As we delve deeper into the technical specifics, it becomes clear that this process offers a robust pathway for cost reduction in antibiotic manufacturing by streamlining purification steps and enhancing yield consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Cefodizime Sodium has been plagued by inefficiencies that compromise both economic viability and product quality. Traditional routes often involve condensation reactions under alkaline conditions followed by salt formation using agents like sodium isooctanoate or sodium bicarbonate. However, these methods frequently yield products with undesirable dark colors, necessitating additional, costly decolorization steps that further reduce overall yield. Furthermore, prior art methods disclosed in various patents rely heavily on organic solvents such as methylene chloride or ethyl acetate, which pose significant environmental and safety hazards while complicating solvent recovery processes. The use of protection groups to avoid side reactions adds unnecessary complexity, increasing the number of operational steps and extending production cycles. Additionally, older purification techniques utilizing macroporous resins have been found to introduce secondary pollution due to resin fragmentation and the leaching of pore-creating agents, ultimately failing to meet the stringent purity specifications required for clinical safety.
The Novel Approach
In stark contrast, the novel method described in the patent data utilizes a streamlined three-step purification protocol that eliminates the need for complex protection-deprotection sequences. The process begins with a controlled acid-base reaction where crude Cefodizime Sodium is dissolved in water and acidified to a precise pH of 2-4, causing the selective precipitation of Cefodizime Acid. This step effectively separates the target molecule from highly soluble impurities. Following filtration, the acid is redissolved and treated with a optimized amount of activated carbon (0.2-0.8% g/ml) at moderate temperatures (40-50°C), which efficiently removes colored impurities without significant adsorption of the active pharmaceutical ingredient. The final and most critical stage involves chromatographic separation using silica gel or neutral alumina rather than fragile macroporous resins. This approach ensures a high-purity end product with minimal risk of contamination, representing a significant leap forward in process reliability and scalability for industrial applications.
Mechanistic Insights into Chromatographic Separation and Impurity Control
The core of this technological advancement lies in the meticulous selection of chromatographic conditions that maximize the separation factor between Cefodizime Sodium and its related substances. The mobile phase consists of a 3:1 volume ratio mixture of acetonitrile and an alkaline solution with a pH value strictly maintained between 9 and 11. This specific alkalinity is crucial because it ensures the ionization state of the carboxylic acid groups on the cephalosporin nucleus is optimal for interaction with the stationary phase, facilitating sharp peak resolution. The flow velocity is carefully controlled between 3.3 and 4.5 ml/min at a column temperature of 25-35°C to prevent thermal degradation of the sensitive beta-lactam ring. By collecting fractions where the drug content exceeds 90% and merging them, the process achieves a final purity greater than 99.5%, effectively removing maximum single impurities to below 1.0% and total impurities to below 1.5%, adhering to strict pharmacopoeia standards.
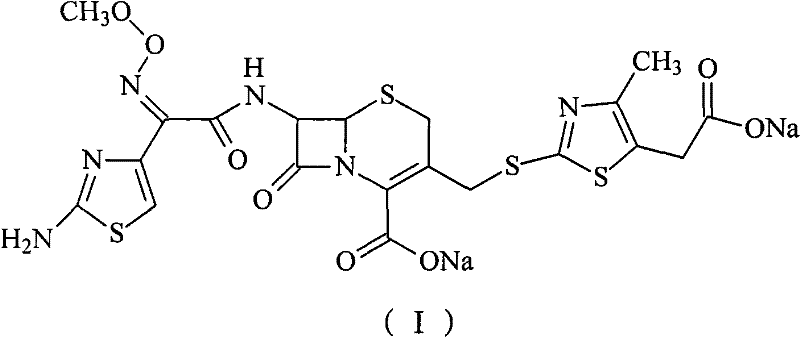
Furthermore, the choice of stationary phase plays a pivotal role in the success of this purification strategy. Unlike macroporous resins which can fragment and release synthetic residues, silica gel (particle diameter 45-250 μm) and neutral alumina (100-300 mesh) offer superior mechanical stability and consistent pore structures. These materials effectively adsorb pigment and composition impurities while allowing the target molecule to elute cleanly. The mechanism relies on the differential adsorption affinity where impurities are retained more strongly or eluted at different times compared to the main component. This selective retention is enhanced by the specific surface chemistry of the silica or alumina, which interacts favorably with the polar functional groups of the impurities. Consequently, this method not only improves the visual clarity and color of the solution but also ensures the chemical stability of the final product, reducing the formation of degradation products during storage.
How to Synthesize Cefodizime Sodium Efficiently
Implementing this purification protocol requires precise adherence to the optimized parameters identified through extensive screening. The process transforms crude bulk material into a pharmaceutical-grade substance suitable for formulation into broad-spectrum antibacterial medicines. The following guide outlines the critical operational phases necessary to replicate the high yields and purity reported in the patent data. Operators must pay close attention to pH adjustments and temperature controls during the decolorization and chromatography stages to ensure reproducibility. Detailed standardized synthesis steps are provided below to facilitate technology transfer and process validation.
- Dissolve crude Cefodizime Sodium in water and adjust pH to 2-4 using acid to precipitate Cefodizime Acid.
- Dissolve the acid in solvent, add 0.2-0.8% activated carbon at 40-50°C, and filter to remove impurities.
- Concentrate filtrate and purify via chromatography using silica gel or neutral alumina with an acetonitrile-alkaline mobile phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel purification method translates into tangible strategic benefits that extend beyond mere technical specifications. By simplifying the production workflow and eliminating the need for expensive protection groups and excessive organic solvents, manufacturers can achieve substantial cost savings in raw material consumption and waste disposal. The robustness of the silica gel and alumina chromatography system reduces the risk of batch failures caused by resin degradation, thereby enhancing supply chain reliability and ensuring consistent delivery schedules. Moreover, the ability to produce high-purity material with reduced toxic side effects aligns perfectly with the growing regulatory emphasis on patient safety, minimizing the risk of costly recalls or compliance issues. This process is inherently scalable, allowing for seamless transition from pilot batches to large-scale commercial production without compromising quality.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection steps significantly reduces the number of unit operations, leading to lower labor and utility costs. By avoiding the use of large volumes of hazardous organic solvents like methylene chloride, the process minimizes solvent recovery expenses and environmental compliance costs associated with VOC emissions. The high yield achieved through optimized chromatographic fraction merging means less raw material is wasted, directly improving the cost-efficiency of the final API. Furthermore, the use of cost-effective stationary phases like silica gel instead of specialized macroporous resins lowers the consumable costs per batch.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the potential bottlenecks often associated with multi-step synthesis routes, ensuring a more predictable production timeline. The stability of the purification columns allows for longer campaign runs without frequent column packing or regeneration, which enhances equipment utilization rates. Sourcing of key reagents such as silica gel and neutral alumina is globally stable, reducing the risk of supply disruptions compared to specialized proprietary resins. This reliability enables suppliers to offer more consistent lead times, a critical factor for pharmaceutical companies managing tight inventory schedules.
- Scalability and Environmental Compliance: The method is designed with industrial scalability in mind, utilizing standard chromatographic equipment that can be easily scaled up from laboratory to plant scale. The reduction in organic solvent usage contributes to a greener manufacturing profile, aligning with modern sustainability goals and reducing the burden on wastewater treatment facilities. The absence of resin fragmentation issues ensures that the final product is free from particulate contamination, simplifying downstream filtration and drying processes. This environmental and operational efficiency makes the process highly attractive for long-term commercial partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and supply of high-purity Cefodizime Sodium. These answers are derived directly from the patented technology details and practical industry experience. Understanding these nuances helps stakeholders make informed decisions about sourcing and process integration. For further technical discussions, our team is available to provide detailed route feasibility assessments.
Q: Why is silica gel preferred over macroporous resin for Cefodizime Sodium purification?
A: Macroporous resins often suffer from fragmentation and pore-creating agent residues, leading to secondary pollution. Silica gel and neutral alumina provide superior rigidity and adsorption capacity for impurities without introducing contaminants, ensuring higher product purity.
Q: What is the critical pH range for the mobile phase in this purification process?
A: The mobile phase must be an alkaline solution with a pH value between 9 and 11, mixed with acetonitrile in a 3:1 volume ratio. This specific alkalinity is crucial for effective elution and separation of the target compound from related substances.
Q: How does this method improve upon traditional precipitation techniques?
A: Traditional methods using sodium isooctanoate often result in darker product colors and require careful handling. This novel method combines acid-base precipitation with chromatographic refinement, significantly improving clarity, color, and overall purity to meet stringent pharmacopoeia standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefodizime Sodium Supplier
The technological potential of this advanced purification route underscores the importance of partnering with a manufacturer who possesses deep process engineering expertise. NINGBO INNO PHARMCHEM stands as a premier CDMO partner, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Cefodizime Sodium meets the highest international standards. We understand the critical nature of antibiotic supply chains and are equipped to handle the complexities of cephalosporin manufacturing with precision and care.
We invite global pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to assist you in evaluating the feasibility of integrating this high-purity material into your supply chain. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our optimized processes can drive value for your organization. Together, we can ensure the continuous availability of safe and effective antibacterial therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
