Advanced Manufacturing of Cefodizime Sodium: Optimizing Yield and Purity for Global Supply Chains
Advanced Manufacturing of Cefodizime Sodium: Optimizing Yield and Purity for Global Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for third-generation cephalosporins, particularly for Cefodizime Sodium, a potent antimicrobial agent with unique immunomodulatory properties. The technical landscape for producing this complex molecule has been significantly advanced by the methodologies disclosed in patent CN101239985A, which outlines a superior preparation method addressing long-standing yield and purity challenges. This innovation focuses on a streamlined three-step sequence that begins with the condensation of 7-amino-cephalosporanic acid (7-ACA) and proceeds through a novel acylation and salt formation process. The core breakthrough lies in the strategic manipulation of reaction conditions to minimize side reactions and simplify downstream processing, thereby offering a compelling value proposition for large-scale manufacturing. By optimizing the catalytic environment and solvent systems, this approach ensures the production of high-quality active pharmaceutical ingredients that meet stringent global regulatory standards.
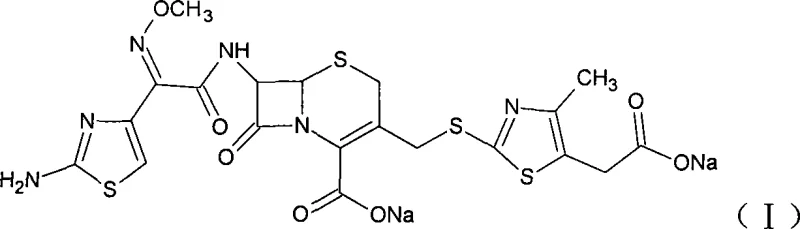
As illustrated in the structural diagram above, Cefodizime Sodium possesses a sophisticated molecular architecture featuring a beta-lactam fused to a dihydrothiazine ring, substituted with specific thiazole groups that confer its biological activity. The precise arrangement of the methoxyimino acetamido group at the C-7 position and the thiazolyl sulfur methyl group at the C-3 position is critical for its efficacy against a broad spectrum of bacteria. Achieving this specific configuration with high stereochemical fidelity is the primary objective of the synthetic route described in the patent data. The ability to construct this molecule efficiently without compromising the integrity of the sensitive beta-lactam ring represents a significant technical achievement, directly impacting the reliability of the supply chain for this essential medicine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefodizime Sodium has been plagued by operational complexities and quality inconsistencies inherent in earlier patented routes. For instance, methods disclosed in U.S. Pat 5126445 rely on triethylamine to dissolve Cefodizime acid, a process that frequently results in products with undesirable dark coloration, necessitating rigorous and costly decolorization treatments that inevitably reduce overall yield. Alternatively, routes utilizing sodium bicarbonate, as seen in U.S. Pat 4590267, suffer from prolonged dissolution times and the persistent presence of undissolved bicarbonate salts in the final product, which compromises pharmaceutical quality and stability. Furthermore, traditional synthetic pathways for the precursor Cefodizime acid often involve either the use of macroporous resins for purification, which adds significant operational cost and complexity, or high-temperature reaction conditions that promote degradation and lower yields. These legacy methods create bottlenecks in production capacity and inflate the cost of goods sold, making them less attractive for modern, cost-sensitive pharmaceutical manufacturing environments.
The Novel Approach
In stark contrast to these conventional limitations, the methodology presented in CN101239985A introduces a refined protocol that drastically simplifies the manufacturing workflow while enhancing product quality. The new approach utilizes a boron trifluoride catalyst system for the initial condensation step, which proceeds under mild conditions to generate the key intermediate with high purity, eliminating the need for complex resin-based purification. Subsequent acylation is performed in a carefully balanced mixed solvent system of halogenated alkanes and alcohols, which optimizes solubility and reaction kinetics without inducing thermal degradation. The most significant innovation, however, is the salt formation step, which employs a two-phase system containing sodium isooctanoate. This specific choice of salt-forming agent and solvent environment ensures complete dissolution of the acid precursor and facilitates the crystallization of the sodium salt in an off-white, high-purity form. This holistic optimization of reaction parameters translates directly into a more robust and economically viable production process.
Mechanistic Insights into BF3-Catalyzed Condensation and Salt Formation
The success of this synthetic route hinges on the precise mechanistic control exerted during the initial condensation of 7-ACA with the thiazole derivative. The use of boron trifluoride, either as a gas or a complex like boron trifluoride acetonitrile, acts as a potent Lewis acid catalyst that activates the carbonyl functionality of the reactants, facilitating nucleophilic attack under mild conditions. This catalytic activation allows the reaction to proceed efficiently at room temperature, thereby preserving the stereochemical integrity of the chiral centers within the cephalosporin nucleus. Following the reaction, the addition of water and careful pH adjustment to the range of 2.5 to 3.5 induces the precipitation of the intermediate as light yellow crystals. This pH-controlled crystallization is critical, as it selectively precipitates the desired product while leaving soluble impurities in the mother liquor, effectively serving as an in-process purification step that negates the need for chromatographic separation.
Furthermore, the mechanism of the final salt formation step is engineered to maximize recovery and purity through phase transfer principles. By suspending the Cefodizime acid in a two-phase system comprising an organic solvent like methylene dichloride and water, the process creates an interface where the salt-forming agent, sodium isooctanoate, can effectively interact with the acid. The organic salt-forming agent provides sodium ions in a manner that promotes the formation of the disodium salt without introducing inorganic contaminants that are difficult to remove. The subsequent addition of alcohol to the aqueous phase reduces the solubility of the product, driving crystallization. This controlled precipitation, combined with activated carbon decolorization, ensures that the final Cefodizime Sodium crystals are free from colored impurities and residual solvents, achieving purity levels exceeding 99.5% as demonstrated in the patent examples.
How to Synthesize Cefodizime Sodium Efficiently
The implementation of this advanced synthesis route requires strict adherence to the optimized reaction conditions and stoichiometric ratios defined in the patent to ensure reproducibility and high yield. The process is divided into three distinct operational stages: the catalytic condensation to form the C-3 side chain intermediate, the acylation at the C-7 position to build the full acid structure, and the final salt formation to generate the stable sodium salt. Each stage utilizes specific solvent systems and temperature controls designed to minimize side reactions such as beta-lactam ring opening or isomerization. Operators must pay particular attention to the pH adjustments and the ratios of organic solvents to water, as these parameters are the primary drivers of crystal quality and yield. The following guide outlines the standardized procedural framework derived from the patent data, providing a clear roadmap for technical teams aiming to adopt this superior manufacturing technology.
- Condense 7-ACA with MMTA using a boron trifluoride catalyst in an organic solvent to form the intermediate compound (IV).
- React compound (IV) with MAEM (V) in a dichloromethane and alcohol mixed solvent with an organic base to generate Cefodizime acid (VI).
- Dissolve Cefodizime acid (VI) in a two-phase system and add sodium isooctanoate to effect salt formation and crystallization of the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic advantages by addressing key pain points associated with traditional cephalosporin manufacturing. The elimination of expensive and operationally intensive purification steps, such as macroporous resin chromatography, directly translates to a reduction in raw material consumption and waste generation. Furthermore, the use of readily available and cost-effective reagents like boron trifluoride complexes and sodium isooctanoate stabilizes the input cost structure, shielding the supply chain from volatility associated with specialized catalysts. The robustness of the crystallization steps ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This reliability is crucial for maintaining continuous supply lines to downstream formulation partners and meeting the demanding delivery schedules of global pharmaceutical markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the downstream processing workflow. By avoiding the use of triethylamine, which causes discoloration requiring expensive remediation, and eliminating the need for high-temperature reactions that degrade yield, the overall cost of production is significantly lowered. The high yield reported in the patent data indicates a more efficient conversion of starting materials into the final product, maximizing the utility of every kilogram of 7-ACA processed. Additionally, the avoidance of complex resin purification columns reduces capital expenditure on equipment and lowers the ongoing costs associated with resin replacement and regeneration. These cumulative efficiencies result in a leaner manufacturing model that enhances profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on common industrial solvents such as methylene dichloride, ethanol, and ethyl acetate ensures that raw material sourcing is secure and resilient. Unlike processes that depend on niche or proprietary reagents, this method utilizes chemicals that are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The operational simplicity of the method, characterized by straightforward filtration and crystallization steps, also reduces the dependency on highly specialized labor, allowing for more flexible production scheduling. This flexibility enables manufacturers to respond more agilely to fluctuations in market demand, ensuring that critical antibiotic supplies remain uninterrupted even during periods of high global consumption.
- Scalability and Environmental Compliance: The environmental profile of this synthesis route is markedly improved compared to legacy methods, aligning with increasingly stringent global regulations on pharmaceutical waste. The reduction in solvent usage and the elimination of solid waste streams associated with resin purification contribute to a lower environmental footprint. The process is inherently scalable, as the liquid-phase reactions and crystallization steps can be easily transferred from pilot plants to large-scale commercial reactors without significant re-engineering. The high purity of the final product also minimizes the need for re-processing or recycling off-spec batches, further reducing the environmental burden. This combination of scalability and compliance makes the technology an ideal candidate for long-term commercial investment and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefodizime Sodium synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the source patent, offering clarity on process capabilities and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term viability of the supply source. The responses highlight the specific advantages of the boron trifluoride catalytic system and the two-phase salt formation technique, demonstrating how they resolve historical manufacturing challenges.
Q: How does the new method improve upon traditional Cefodizime Sodium synthesis routes?
A: The novel method overcomes the limitations of prior art, such as the dark coloration associated with triethylamine use and the incomplete dissolution issues with sodium bicarbonate. By utilizing a boron trifluoride catalyst and a specific two-phase salt formation system, the process achieves higher yields and superior product clarity without complex purification steps like macroporous resin chromatography.
Q: What are the critical parameters for controlling impurities during the salt formation step?
A: Critical parameters include maintaining the pH between 2.5 and 3.5 during crystallization to prevent hydrolysis of the beta-lactam ring. Additionally, the volume ratio of the organic solvent to water in the two-phase system (optimized at 3:1 to 1:1) and the specific ratio of water to alcohol during the final crystallization are essential for ensuring high purity and removing residual organic bases effectively.
Q: Is this synthesis route scalable for industrial commercial production?
A: Yes, the route is designed for scalability. It replaces expensive and operationally complex steps, such as high-temperature reactions that cause discoloration or resin-based purifications, with robust liquid-phase reactions. The use of common solvents like methylene dichloride and ethanol, along with standard crystallization techniques, facilitates easy scale-up from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefodizime Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative methods described in patent CN101239985A can be seamlessly integrated into our manufacturing operations. We are committed to delivering Cefodizime Sodium that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this process, guaranteeing a consistent supply of high-quality intermediates and APIs for our partners.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this superior manufacturing method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to enhance the efficiency and reliability of your Cefodizime Sodium supply, ensuring that your patients receive the highest quality medication without interruption.
