Advanced Synthesis of Cefodizime Sodium: Technical Breakthroughs and Commercial Scalability
Advanced Synthesis of Cefodizime Sodium: Technical Breakthroughs and Commercial Scalability
The pharmaceutical landscape for third-generation cephalosporins continues to evolve, driven by the relentless demand for higher purity antibiotics and more efficient manufacturing processes. Patent CN101239985B introduces a significant methodological advancement in the preparation of Cefodizime Sodium, a potent antimicrobial agent known for its dual antibiotic and immunoregulatory properties. This technical insight report dissects the proprietary synthesis route disclosed in the patent, highlighting how it overcomes the historical bottlenecks of yield loss and complex purification associated with legacy methods. By shifting from cumbersome resin-based purification to a streamlined acid-catalyzed condensation and optimized salt formation strategy, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chains. The following analysis provides a deep dive into the chemical mechanisms, operational parameters, and commercial implications of this novel pathway.
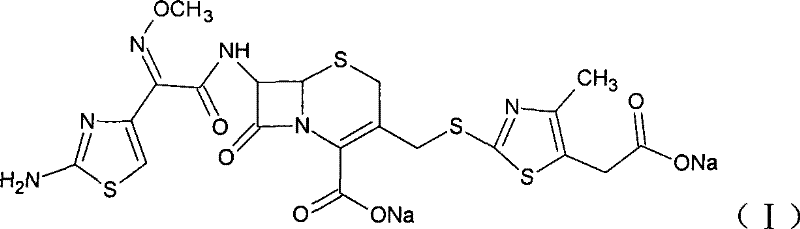
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Cefodizime Sodium has been plagued by inefficiencies inherent in traditional synthetic routes, particularly regarding the conversion of Cefodizime acid to its sodium salt and the synthesis of the acid precursor itself. Prior art, such as the methods disclosed in U.S. Pat 5126445 and U.S. Pat 4590267, often relies on dissolving the acid in water with triethylamine or sodium bicarbonate. These approaches frequently result in products with darker coloration that require meticulous and costly care during processing, or they suffer from prolonged dissolution times and the risk of undissolved inorganic bases contaminating the final product. Furthermore, the synthesis of the Cefodizime acid precursor typically involves either complex purification steps using macroporous resins to isolate intermediates or high-temperature reactions that degrade product quality and lower overall yields. These legacy constraints not only inflate the cost of goods sold but also introduce variability that complicates quality control in a regulated pharmaceutical environment.
The Novel Approach
The methodology outlined in patent CN101239985B presents a robust alternative that fundamentally restructures the synthesis workflow to enhance efficiency and product integrity. Instead of relying on aqueous dissolution with inorganic bases, the new process employs a sophisticated two-phase system for salt formation, utilizing organic salt-forming agents like sodium isooctanoate to ensure complete dissolution and superior crystal quality. In the upstream synthesis of the acid intermediate, the invention replaces the need for macroporous resin purification with a direct acid-catalyzed condensation using boron trifluoride complexes. This shift allows the intermediate to be isolated simply by pH adjustment and filtration, yielding a light yellow crystal of high purity that can be directly fed into the subsequent acylation step without further purification. This integrated approach drastically reduces the number of unit operations, minimizes solvent consumption, and elevates the overall yield from the historical average of approximately 50% to over 60%, representing a substantial leap in process economy.
Mechanistic Insights into BF3-Catalyzed Condensation and Acylation
The core of this technological breakthrough lies in the precise manipulation of reaction conditions during the formation of the critical 7-amino-3-substituted cephalosporin intermediate. The process initiates with the condensation of 7-amino-cephalosporanic acid (7-ACA) and 2-sulfhydryl-4-methyl-5-thiazole acetate under the influence of a Lewis acid catalyst, specifically boron trifluoride gas or its acetonitrile complex. The boron trifluoride acts as a potent activator, facilitating the nucleophilic attack of the thiol group on the cephalosporin nucleus while maintaining the stereochemical integrity of the beta-lactam ring. Unlike alkaline conditions which can lead to ring opening or degradation, this acidic environment ensures a clean reaction profile. Following the reaction, the addition of water and careful pH adjustment to the range of 2.5 to 3.5 precipitates the intermediate as a high-purity solid. This mechanistic control is pivotal, as it eliminates the formation of polymeric byproducts and ensures that the resulting intermediate possesses the necessary reactivity for the subsequent acylation step without requiring chromatographic purification.
Following the formation of the intermediate, the process proceeds to the acylation stage where the amino group at the 7-position is coupled with the methoxyimino side chain. This step is conducted in a mixed solvent system comprising a halogenated alkane, such as methylene dichloride, and a lower alcohol like methanol or ethanol. The presence of an organic base, such as triethylamine or N-methylmorpholine, serves to scavenge the acid byproduct generated during the amide bond formation, driving the equilibrium towards the desired Cefodizime acid. The choice of a mixed solvent system is critical here; it balances the solubility of the polar reactants with the precipitation characteristics of the product. Upon completion, the product is isolated through extraction and acidification, yielding Cefodizime acid with purity levels exceeding 98%. This high level of purity entering the final salt formation step is what enables the downstream process to achieve such exceptional final product quality, minimizing the burden on final crystallization and drying stages.
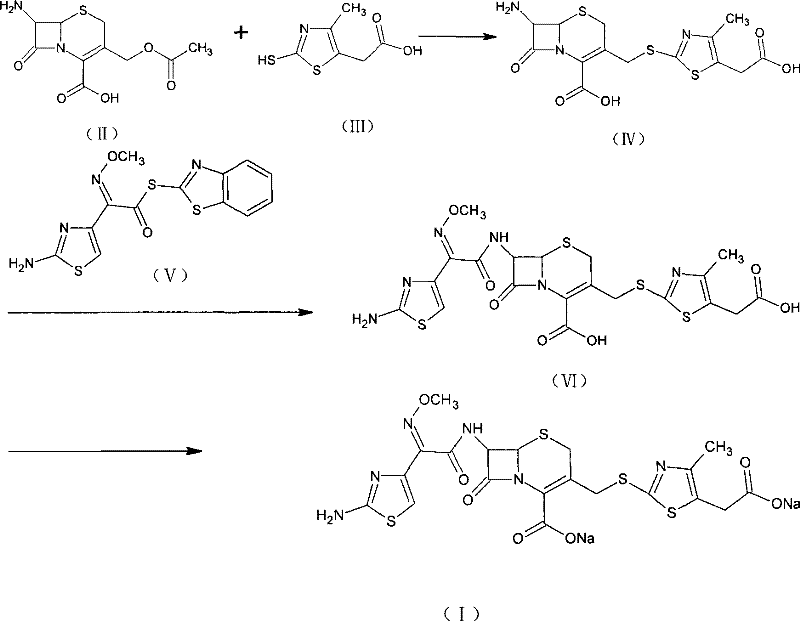
How to Synthesize Cefodizime Sodium Efficiently
The implementation of this patented synthesis route requires strict adherence to the specified stoichiometric ratios and solvent systems to replicate the high yields reported in the experimental data. The process is divided into three distinct stages: the initial condensation to form the thioether intermediate, the acylation to install the aminothiazole side chain, and the final salt formation. Each stage has been optimized to minimize waste and maximize throughput, making it an ideal candidate for transfer to commercial scale manufacturing facilities. Operators must pay particular attention to the temperature controls during the boron trifluoride addition and the pH adjustments during isolation, as these are the critical control points that determine the success of the synthesis. For a detailed breakdown of the specific reagent quantities, reaction times, and workup procedures, please refer to the standardized synthesis guide provided below.
- React 7-amino-cephalosporanic acid (II) with 2-sulfhydryl-4-methyl-5-thiazole acetate (III) using a boron trifluoride catalyst to form intermediate (IV).
- Acylation of intermediate (IV) with active ester (V) in a mixed solvent system containing an organic base to yield Cefodizime acid (VI).
- Convert Cefodizime acid (VI) to the final sodium salt (I) using an organic salt-forming agent in a two-phase solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates directly into tangible operational improvements and risk mitigation. The elimination of macroporous resin purification, which is a capital-intensive and time-consuming step in conventional routes, significantly streamlines the production timeline. This reduction in processing complexity means that manufacturing cycles can be shortened, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations. Furthermore, the use of common, commercially available solvents like acetonitrile and methylene dichloride ensures that raw material sourcing remains stable and cost-effective, avoiding reliance on specialized or exotic reagents that might introduce supply chain vulnerabilities. The robustness of the process also implies a lower rate of batch failures, which is a critical factor in maintaining consistent inventory levels for high-demand antibiotic intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the macroporous resin purification step and the simplification of the salt formation procedure. By avoiding the need for expensive resin columns and the associated solvent regeneration costs, the variable cost per kilogram of the final API is substantially decreased. Additionally, the higher overall yield means that less starting material is required to produce the same amount of finished product, effectively lowering the raw material cost basis. The ability to use the intermediate directly in the next step without additional purification further reduces labor and utility costs associated with extra isolation and drying phases, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on standard industrial solvents and reagents such as boron trifluoride complexes and sodium isooctanoate ensures a resilient supply chain that is less susceptible to disruptions. Unlike processes that depend on niche catalysts or difficult-to-source chiral auxiliaries, the inputs for this synthesis are widely available from multiple global suppliers. This diversity in sourcing options provides procurement teams with greater negotiating power and security against regional shortages. Moreover, the simplified workflow reduces the dependency on specialized equipment, meaning that production can be easily scaled or shifted between different manufacturing sites without significant retooling investments, thereby enhancing overall supply continuity.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers distinct advantages by reducing the volume of waste generated per unit of product. The avoidance of resin regeneration wastes and the minimization of solvent usage through efficient mixed-solvent systems contribute to a smaller environmental footprint. The process generates fewer hazardous byproducts compared to high-temperature alternatives, simplifying waste treatment and disposal compliance. As production scales from pilot batches to multi-ton commercial runs, the exothermic nature of the reactions can be managed effectively with standard cooling systems, ensuring that safety and environmental standards are maintained even at high volumes, facilitating smoother regulatory approvals for commercial expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefodizime Sodium synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology. Understanding these nuances is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios and for anticipating potential challenges during technology transfer.
Q: What are the key advantages of the BF3-catalyzed route for Cefodizime Sodium?
A: The patented method eliminates the need for complex macroporous resin purification required in conventional routes, significantly simplifying the workflow and reducing production costs while maintaining high purity levels above 99%.
Q: How does the new salt formation process improve product quality?
A: By utilizing a two-phase system with organic salt-forming agents like sodium isooctanoate, the process avoids the issues of undissolved inorganic bases and dark coloration often seen in traditional triethylamine or bicarbonate methods.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents such as acetonitrile and methylene dichloride and operates under mild conditions, making it highly scalable for commercial production of antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefodizime Sodium Supplier
The technical advancements detailed in patent CN101239985B underscore the potential for significant efficiency gains in the production of this critical antibiotic. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to life. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic conditions described, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we guarantee that our Cefodizime Sodium supplies are consistent, high-quality, and fully compliant with international pharmaceutical standards, providing a secure foundation for your drug development and manufacturing needs.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the global antibiotic market.
