Advanced Manufacturing of Cefodizime Sodium: A Green Chemistry Approach for High-Purity API Intermediates
Advanced Manufacturing of Cefodizime Sodium: A Green Chemistry Approach for High-Purity API Intermediates
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical antibiotic intermediates. A pivotal advancement in this domain is detailed in patent CN102942575A, which outlines a novel preparation method for Cefodizime Sodium, a third-generation cephalosporin with dual antibiotic and immunoregulatory properties. This technology addresses significant bottlenecks in traditional manufacturing, specifically replacing hazardous boron trifluoride catalysts with a more manageable sulfuric acid system. By optimizing reaction conditions and solvent systems, this method achieves a total product yield exceeding 75% and an HPLC purity greater than 99.7%. For R&D directors and procurement specialists, understanding this shift is crucial for securing a reliable API intermediate supplier capable of delivering high-quality materials with reduced environmental impact.
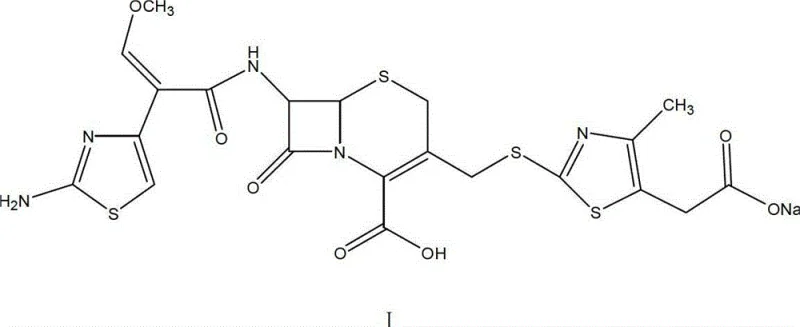
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefodizime Sodium has relied heavily on boron trifluoride gas or its complexes as acidic catalysts for the condensation of 7-amino-cephalosporanic acid (7-ACA) with side-chain precursors. While chemically effective, this approach presents severe logistical and safety challenges for large-scale operations. The use of boron trifluoride generates substantial noxious fumes, creating significant environmental pollution risks that require expensive scrubbing and containment infrastructure. Furthermore, the reliance on such specialized reagents drives up raw material costs and complicates supply chain continuity. Prior art methods often resulted in total yields hovering around 60%, necessitating extensive purification steps, such as macroporous resin chromatography, which further erodes profit margins and increases production lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally reengineers the synthetic pathway to eliminate these inefficiencies. By substituting boron trifluoride with concentrated sulfuric acid in an acetonitrile solvent system, the process drastically simplifies the operational workflow while mitigating safety hazards. This substitution not only removes the need for handling toxic gases but also leverages the cost-effectiveness of sulfuric acid. Crucially, the introduction of specific additives like sodium hydrosulfite (vat powder) counteracts the potential oxidative discoloration typically associated with sulfuric acid catalysis. The subsequent steps utilize dimethyl carbonate as a green solvent, which is easily recoverable, contrasting sharply with the difficult-to-recycle mixed solvents of previous methods. This holistic optimization results in a streamlined process that is highly favorable for the commercial scale-up of complex antibiotic intermediates.
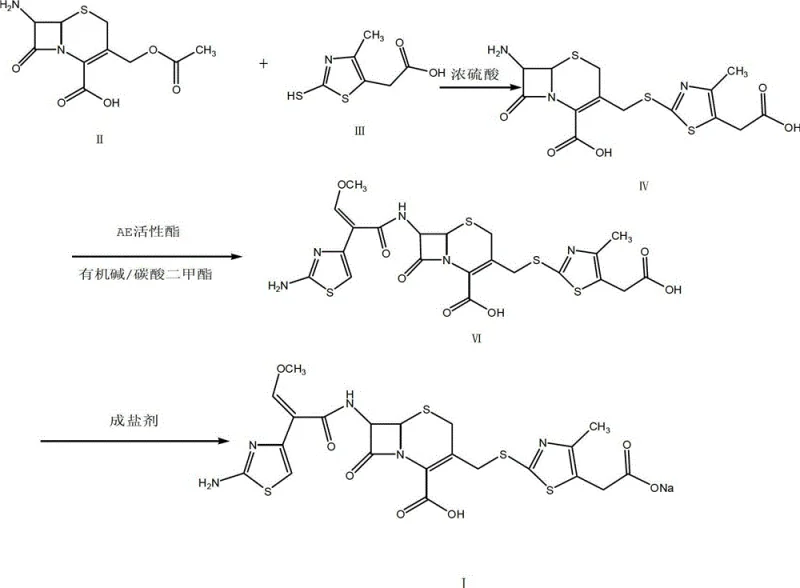
Mechanistic Insights into Sulfuric Acid-Catalyzed Condensation
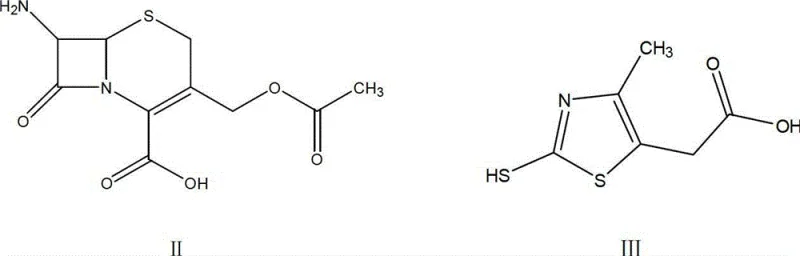
The core chemical innovation lies in the precise control of the condensation reaction between 7-ACA and 2-mercapto-4-methyl-5-thiazole acetic acid (MMTA). In traditional Lewis acid catalysis, boron trifluoride activates the carbonyl species, but its volatility makes stoichiometric control difficult. In this new protocol, concentrated sulfuric acid acts as a potent Brønsted acid catalyst within an acetonitrile medium. The reaction is conducted at a controlled temperature range of 28°C to 30°C, which is mild enough to preserve the integrity of the beta-lactam ring while providing sufficient energy for the nucleophilic attack. The mechanistic pathway involves the protonation of the leaving group or activation of the electrophilic center, facilitating the formation of the thioether linkage at the C-3 position of the cephem nucleus. This step is critical as it establishes the foundational scaffold for the subsequent acylation.
A major challenge in switching to sulfuric acid is the tendency for the reaction mixture to darken due to oxidation, which can compromise the aesthetic and chemical quality of the intermediate TACS. The patent elucidates a sophisticated impurity control mechanism involving the addition of sodium hydrosulfite (0.01 to 0.03 times the weight of 7-ACA). This reducing agent effectively scavenges dissolved oxygen and reactive oxidative species generated during the acid catalysis, thereby preserving the solution's transmittance. Following the reaction, the pH is carefully adjusted to 2.8–3.0 using an alkaline reagent like ammonia or triethylamine. This precise pH control ensures the precipitation of the desired TACS intermediate while keeping impurities in solution. The result is a crude intermediate with high purity (over 98%) and excellent flowability, setting the stage for the high-yield acylation step that follows.
How to Synthesize Cefodizime Sodium Efficiently
The synthesis of Cefodizime Sodium via this patented route involves a logical sequence of condensation, acylation, and salt formation, designed for maximum efficiency and minimal waste. The process begins with the generation of the TACS intermediate, followed by coupling with the AE active ester in a dimethyl carbonate solvent system, and concludes with a controlled salting-out procedure. Each step is optimized to minimize side reactions and maximize recovery. For process engineers and laboratory teams looking to implement this technology, understanding the specific parameters regarding temperature, pH, and additive timing is essential for replicating the reported success. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to facilitate immediate technology transfer.
- Condense 7-ACA with MMTA in acetonitrile using concentrated sulfuric acid, adding sodium hydrosulfite to prevent oxidation.
- React the resulting TACS intermediate with AE active ester in dimethyl carbonate solvent with an organic base.
- Perform salt formation with a sodium bicarbonate and sodium carbonate mixture in ethanol to obtain the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this sulfuric acid-based methodology represents a strategic opportunity to optimize cost structures and enhance supply reliability. The elimination of boron trifluoride removes a significant variable cost component and reduces the regulatory burden associated with hazardous material handling. Furthermore, the implementation of recoverable solvents like acetonitrile and dimethyl carbonate aligns with modern sustainability goals, potentially lowering waste disposal fees and improving the company's environmental compliance profile. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous boron trifluoride catalysts with commodity-grade sulfuric acid results in a direct and substantial reduction in raw material expenditures. Additionally, the simplified purification process, which avoids the need for costly macroporous resin columns, significantly lowers operational overheads. The high total yield of over 75% means that less starting material is required to produce the same amount of final API, effectively amplifying the return on investment for every kilogram of 7-ACA purchased. This economic efficiency is critical for maintaining competitive pricing in the generic antibiotic market.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents such as sulfuric acid and sodium hydrosulfite, the manufacturing process becomes less susceptible to supply disruptions often associated with specialized fluorine chemicals. The robustness of the reaction conditions, particularly the tolerance for slight variations in temperature and the use of stable solvents, ensures consistent batch-to-batch performance. This reliability allows for more accurate production planning and reduces the risk of stockouts, ensuring that downstream formulation partners receive their orders on time. The ability to source raw materials locally further shortens the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing solvents that are easy to recover and recycle through standard distillation techniques. The absence of noxious fumes simplifies the engineering controls required for reactor ventilation, making it easier to scale from pilot plants to multi-ton commercial production facilities. Moreover, the use of dimethyl carbonate, a recognized green solvent, minimizes the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the brand reputation of the manufacturer as a responsible producer of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Cefodizime Sodium. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. They are intended to clarify the operational benefits and quality assurances associated with this advanced manufacturing route. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: Why is sulfuric acid preferred over boron trifluoride in this synthesis?
A: Sulfuric acid eliminates the environmental hazards and fuming associated with boron trifluoride gas, significantly reducing raw material costs and improving operational safety while maintaining high yields.
Q: How does the process control product color and purity?
A: The addition of sodium hydrosulfite (vat powder) during the condensation step and sulfurous acid during the acylation step prevents oxidative discoloration, ensuring the final product achieves over 99.7% HPLC purity with excellent color properties.
Q: What are the solvent advantages in this new method?
A: The use of acetonitrile and dimethyl carbonate allows for easier solvent recovery and recycling compared to traditional mixed solvent systems, contributing to a more sustainable and cost-effective manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefodizime Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for life-saving antibiotics like Cefodizime Sodium. Our technical team has extensively analyzed this patented methodology and possesses the expertise to adapt and optimize it for large-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify every batch against the highest pharmacopoeial standards, guaranteeing that the impurity profiles and physical characteristics, such as color and flowability, match the superior benchmarks set by this innovative process.
We invite global pharmaceutical partners to collaborate with us to leverage these technological advancements for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates exactly how switching to this optimized route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable, high-quality supply of Cefodizime Sodium that drives your business forward.
