Advanced Manufacturing Strategy for High-Purity Dexamethasone and Derivatives via Optimized Steroidal Intermediates
Advanced Manufacturing Strategy for High-Purity Dexamethasone and Derivatives via Optimized Steroidal Intermediates
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of critical corticosteroids like dexamethasone. Patent CN101397320A introduces a transformative methodology that fundamentally restructures the synthesis of dexamethasone and its series products, including dexamethasone acetate and dexamethasone sodium phosphate. This innovation pivots away from convoluted historical routes, instead leveraging 1,4,9,16-tetraene-pregna-3,20-diketone as a strategic starting material. By focusing modifications on the 9, 11, 16, 17, and 21 positions through a streamlined sequence of reactions, this approach offers a compelling solution for reliable pharmaceutical intermediate supplier networks aiming to optimize their API manufacturing portfolios. The technical breakthrough lies not just in the chemical transformations themselves, but in the strategic selection of precursors that align with existing industrial capabilities, thereby facilitating a seamless transition from laboratory scale to commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for dexamethasone have been plagued by excessive complexity and economic inefficiency, creating significant bottlenecks for cost reduction in API manufacturing. For instance, the route described in document GB869511A relies on 4,16-pregnadiene-3,11,20-triketone as a starting material and necessitates a cumbersome 18-step transformation sequence to reach the final product. As illustrated in the legacy pathway below, this method requires extensive protection and de-protection of ketone groups at multiple stages, which not only lowers overall yield but also generates substantial chemical waste.
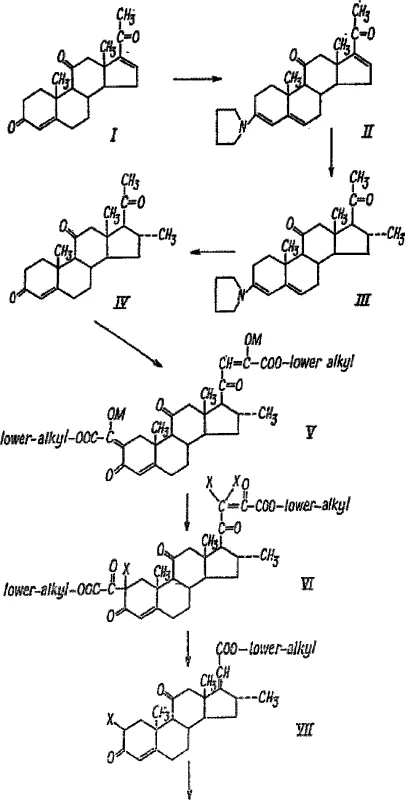
Furthermore, alternative routes such as those found in document ES8800260A utilize expensive starting materials like 9-alpha-fluoro-11-beta,21-dihydroxy-pregna-1,4,16-triene-3,20-dione-11-beta,21-diethyl ester. These legacy methods often involve difficult purification steps, such as column chromatography after Grignard reactions, which are notoriously difficult to scale industrially. The combination of low yields, high material costs, and operational complexity in these conventional methods renders them suboptimal for modern, high-volume commercial scale-up of complex steroidal intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in CN101397320A capitalizes on the availability of 1,4,9,16-tetraene-pregna-3,20-diketone, an intermediate that is already produced in large quantities by many manufacturers for other steroid lines. This strategic choice eliminates the need for expensive, specialized precursors and allows for the implementation of a much shorter, more direct synthetic sequence. The new route achieves the necessary structural modifications through a logical progression of Grignard addition, epoxidation, fluorination, iodination, and substitution. This simplicity translates directly into operational efficiency, as fewer unit operations mean reduced processing time and lower energy consumption.
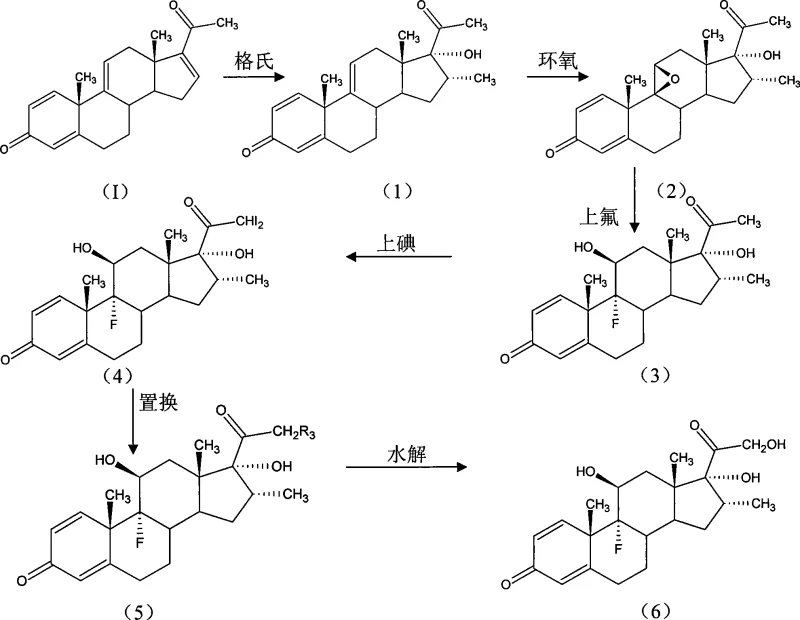
Moreover, this methodology enables a unique "doubling production" capability, where the same production lines used for betamethasone series products can be adapted for dexamethasone with minimal retooling. This flexibility is a game-changer for supply chain heads, as it maximizes asset utilization and provides a buffer against market fluctuations. By avoiding the use of costly auxiliary materials and simplifying the purification of intermediates through standard chemical methods, this novel approach ensures that high-purity dexamethasone can be produced with dramatically better economics than prior art methods.
Mechanistic Insights into the Optimized Steroidal Transformation
The core of this technological advancement lies in the precise control of stereochemistry and functional group interconversion, particularly at the C9, C11, and C21 positions. The process initiates with a Grignard reaction where the starting tetraene-diketone is treated with a methyl magnesium halide in a polar organic solvent like tetrahydrofuran (THF), followed by oxidation to install the 17-alpha-hydroxyl group. This is followed by a critical epoxidation step involving a halogenating agent and acid catalyst to form the 9-beta,11-beta-epoxide, which sets the stage for the subsequent introduction of the fluorine atom. The regioselectivity and stereoselectivity of these steps are paramount, as they determine the biological activity of the final corticosteroid.
Following epoxidation, the ring is opened via fluorination using hydrogen fluoride to yield the 9-alpha-fluoro-11-beta-hydroxy configuration, a hallmark of potent glucocorticoids. The synthesis then proceeds to the C21 position, where iodination introduces a leaving group that facilitates nucleophilic substitution. By reacting the iodide intermediate with alkyl carboxylates (such as potassium acetate), the process efficiently installs the ester functionality required for derivatives like dexamethasone acetate. Finally, a controlled hydrolysis step removes the ester group to yield the free alcohol of dexamethasone. Each step is optimized for mild conditions and high conversion, minimizing the formation of impurities and ensuring a clean impurity profile suitable for pharmaceutical applications.
How to Synthesize Dexamethasone Efficiently
The synthesis of dexamethasone via this patented route involves a sequence of six distinct chemical transformations that convert the readily available tetraene-diketone precursor into the final active pharmaceutical ingredient. The process is designed to be operationally simple, utilizing common solvents and reagents that are easily sourced in a GMP environment. Detailed below is the strategic overview of the workflow, which balances reaction efficiency with ease of purification to ensure consistent quality.
- Perform a Grignard reaction on the starting material using methyl magnesium bromide and an oxidant to form the 17-alpha-hydroxy intermediate.
- Execute an epoxidation reaction followed by a fluorination step to introduce the critical 9-alpha-fluoro and 11-beta-hydroxy groups.
- Complete the synthesis via iodination at the 21-position, substitution with an alkyl carboxylate, and final hydrolysis to yield dexamethasone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain; by utilizing 1,4,9,16-tetraene-pregna-3,20-diketone, manufacturers can leverage existing inventory and supplier relationships, thereby reducing lead time for high-purity corticosteroids. The elimination of complex protection-deprotection sequences means fewer raw materials need to be sourced, qualified, and managed, which streamlines the entire procurement lifecycle and reduces administrative overhead.
- Cost Reduction in Manufacturing: The economic impact of this route is profound due to the removal of expensive auxiliary materials and the reduction in total processing steps. Unlike legacy methods that require costly protecting groups and specialized reagents, this process relies on commodity chemicals and straightforward reaction conditions. The avoidance of difficult purification techniques like column chromatography in favor of crystallization and extraction further drives down operational expenditures, resulting in substantial cost savings that can be passed down the value chain.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of starting materials that are common intermediates in the steroid industry. This reduces the risk of supply disruption associated with niche, single-source raw materials. Furthermore, the compatibility of this route with existing betamethasone production lines allows for flexible capacity allocation. If demand for dexamethasone spikes, manufacturers can quickly ramp up production without the need for significant capital investment in new infrastructure, ensuring continuity of supply for critical medications.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the shorter synthetic route inherently generates less waste and consumes less energy per kilogram of product. The simplified workup procedures, which avoid heavy metal catalysts and toxic solvents where possible, align with modern green chemistry principles. This makes the process easier to scale from pilot batches to multi-ton commercial production while maintaining compliance with increasingly stringent environmental regulations regarding waste disposal and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dexamethasone synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical scope of the method for potential partners and licensees.
Q: What are the primary advantages of this new dexamethasone synthesis route compared to traditional methods?
A: The new route utilizes readily available existing intermediates (1,4,9,16-tetraene-pregna-3,20-diketone), significantly shortening the synthetic sequence compared to the 18-step traditional routes. It eliminates the need for expensive protecting groups and auxiliary materials, thereby drastically reducing manufacturing costs and simplifying industrial purification processes.
Q: Can this process be adapted for producing dexamethasone derivatives like acetate or phosphate?
A: Yes, the process is highly versatile. By modifying the substitution step with specific alkyl carboxylates (such as acetate) or performing a subsequent phosphorylation reaction on the final dexamethasone product, manufacturers can efficiently produce dexamethasone acetate and dexamethasone sodium phosphate without altering the core upstream synthetic pathway.
Q: How does this method impact supply chain reliability for corticosteroid APIs?
A: This method enhances supply chain reliability by enabling 'doubling production' on existing betamethasone production lines. Since it uses common intermediates already stocked by many manufacturers, it reduces dependency on scarce, specialized starting materials and minimizes the risk of supply disruptions associated with complex, multi-step legacy syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexamethasone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthesis routes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex steroidal intermediates like dexamethasone are manufactured with the highest standards of quality and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the exacting requirements of global regulatory bodies, providing our partners with absolute confidence in our supply.
We invite you to collaborate with us to leverage this advanced technology for your API needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience and profitability.
