Scalable Synthesis of Aryl Thioamides via Alkali Metal Complex Catalysis for Global Pharma Supply Chains
Introduction to Advanced Thioamide Manufacturing Technology
The pharmaceutical and agrochemical industries continuously demand efficient, sustainable routes for constructing sulfur-containing heterocycles and bioactive motifs. Patent CN112574081B introduces a groundbreaking methodology for the preparation of aryl thioamide compounds, addressing critical bottlenecks in traditional synthetic pathways. This technology leverages a unique alkali metal complex catalytic system to transform readily available aryl methanols into high-value thioamides using sublimed sulfur and formamide. For R&D directors and procurement strategists, this represents a significant shift towards greener chemistry, offering a robust alternative to hazardous reagents while maintaining high substrate universality across diverse electronic environments.
The significance of this invention extends beyond mere academic interest; it provides a tangible solution for the reliable supply of complex pharmaceutical intermediates. By utilizing a three-component one-pot reaction, the process minimizes waste generation and simplifies downstream processing. The ability to operate at mild temperatures ranging from room temperature to 60°C drastically reduces energy consumption compared to classical methods requiring extreme heat. This technical advancement positions manufacturers to meet stringent environmental regulations while securing a cost-effective supply chain for essential drug building blocks used in DNA gyrase inhibitors and other therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamide compounds has relied heavily on two primary strategies, both of which present substantial drawbacks for modern industrial application. The first method involves the sulfurization of carbonamides using Lawson's reagent or phosphorus pentasulfide. These reagents are notoriously unstable, expensive, and generate significant amounts of toxic phosphorus-containing waste, posing severe environmental and safety challenges. Furthermore, the prerequisite synthesis of the amide precursor adds an extra step, reducing overall step economy and increasing production costs. The second conventional approach, the Willgestrom-Kindler reaction, utilizes elemental sulfur but often requires harsh reaction conditions, with temperatures soaring up to 320°C. Such extreme thermal requirements limit substrate compatibility, degrade sensitive functional groups, and necessitate specialized, high-cost reactor equipment capable of withstanding such stress.
The Novel Approach
In stark contrast, the methodology disclosed in CN112574081B offers a streamlined, mild, and highly efficient pathway. By employing aryl methanol as the starting substrate, the process bypasses the need for pre-formed amides or carboxylic acids. The reaction utilizes sublimed sulfur as a clean sulfur source and formamide, which serves a dual role as both the solvent and the amine source. This dual functionality eliminates the need for additional organic solvents, thereby simplifying the workup procedure and reducing solvent waste. The reaction proceeds smoothly under inert gas protection at temperatures between room temperature and 60°C, ensuring the integrity of sensitive functional groups on the aromatic ring.
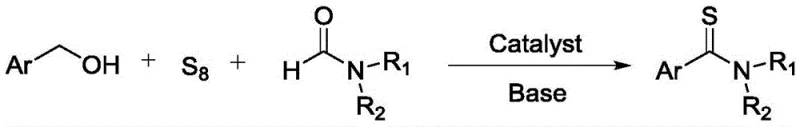
The visual representation of this transformation highlights the elegance of the one-pot design. As shown in the reaction scheme, the direct coupling of the alcohol, sulfur, and formamide leads to the target thioamide with high atom economy. This approach not only mitigates the safety risks associated with handling volatile or toxic reagents but also significantly lowers the barrier to entry for manufacturing these valuable intermediates. The broad scope of compatible substrates, including various substituted benzyl alcohols and heteroaromatic methanols, underscores the versatility of this novel catalytic system for diverse chemical libraries.
Mechanistic Insights into Alkali Metal Complex Catalyzed Thioamidation
The core of this technological breakthrough lies in the sophisticated catalytic system comprising an alkali metal salt and a specific ligand. The patent details the formation of an active alkali metal complex, such as those derived from cesium chloride paired with ethylenediaminetetraacetic acid (EDTA) or salicylaldehyde. This complex acts as a powerful promoter for the activation of elemental sulfur, facilitating its insertion into the carbon-nitrogen bond formation process. The presence of a base, such as sodium hydroxide or potassium carbonate, further accelerates the reaction by deprotonating intermediates and driving the equilibrium towards product formation. This synergistic interaction between the metal complex and the base allows the reaction to proceed efficiently at low temperatures, avoiding the thermal degradation often seen in uncatalyzed sulfurizations.
Understanding the substrate scope is crucial for R&D teams evaluating this technology for specific API intermediates. The aryl methanol component, represented generally as Ar-CH2-OH, tolerates a wide array of substituents. Electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, trifluoromethyl, and nitro, are all compatible with the reaction conditions.
Similarly, the amine source, derived from formamide derivatives, offers flexibility in the final product structure. The general structure H-C(=O)-N(R1)(R2) indicates that primary, secondary, and cyclic amines can be incorporated into the thioamide motif.
This structural diversity is vital for medicinal chemists seeking to optimize the pharmacokinetic properties of lead compounds. The mechanism ensures that impurities arising from over-oxidation or side reactions are minimized, resulting in a cleaner crude product profile. The use of formamide as a solvent also helps in solubilizing the inorganic sulfur and the polar intermediates, creating a homogeneous reaction environment that enhances mass transfer and reaction kinetics. This level of control over the reaction pathway is what enables the high yields reported in the patent examples, often exceeding 70-80% even on a laboratory scale.
How to Synthesize Aryl Thioamide Compounds Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and order of addition to maximize the efficiency of the alkali metal catalyst. The standard protocol involves charging the reactor with the aryl methanol substrate, followed by the addition of sublimed sulfur and the base. The formamide solvent is then introduced, serving as the reaction medium. Finally, the catalytic system, consisting of the alkali metal salt and ligand, is added to initiate the transformation. The detailed standardized synthesis steps below outline the precise operational parameters required to achieve optimal results.
- Under inert gas protection, mix aryl methanol, sublimed sulfur, alkali, alkali metal salt catalyst, and ligand in formamide.
- Stir the reaction mixture at temperatures between room temperature and 60°C for 6 to 12 hours to ensure complete conversion.
- Quench with water, extract with ethyl acetate, wash with brine, dry, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic thioamidation technology offers profound strategic advantages beyond simple yield improvements. The shift away from expensive and hazardous reagents like Lawson's reagent directly translates to substantial cost savings in raw material procurement. By utilizing commodity chemicals such as benzyl alcohol and sublimed sulfur, manufacturers can decouple their production costs from the volatility of specialty reagent markets. Furthermore, the elimination of toxic phosphorus byproducts simplifies waste treatment protocols, reducing the environmental compliance burden and associated disposal fees. This green chemistry approach aligns perfectly with the sustainability goals of major multinational pharmaceutical corporations, enhancing the marketability of the supplied intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the synthetic route. By combining the sulfur source and amine source in a one-pot reaction, the need for intermediate isolation and purification steps is removed. This reduction in unit operations lowers labor costs, decreases solvent consumption, and minimizes equipment occupancy time. Additionally, the use of inexpensive alkali metal salts as catalysts, rather than precious transition metals, ensures that the catalyst cost remains negligible. The mild reaction conditions (Rt-60°C) also result in significantly lower energy expenditures for heating and cooling compared to traditional high-temperature sulfurization methods, contributing to a leaner overall manufacturing cost structure.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-restricted starting materials. Benzyl alcohols and formamides are produced on a massive global scale, ensuring a stable supply even during market fluctuations. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, reducing the risk of batch failures. Moreover, the simplified post-treatment procedure, involving standard aqueous workup and extraction, allows for faster turnaround times from reaction completion to finished goods. This agility enables suppliers to respond more quickly to urgent customer demands, reducing lead times for high-purity pharmaceutical intermediates and strengthening the reliability of the supply partnership.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is straightforward due to the absence of exothermic hazards or high-pressure requirements. The reaction can be safely conducted in standard glass-lined or stainless steel reactors without the need for specialized high-pressure autoclaves. From an environmental perspective, the process generates minimal hazardous waste, primarily consisting of aqueous saline solutions that are easier to treat than phosphorus-containing sludge. The high atom economy of using elemental sulfur ensures that nearly all sulfur atoms end up in the desired product, minimizing waste generation. This compliance with green chemistry principles facilitates smoother regulatory approvals and supports the long-term sustainability of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aryl thioamide synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production workflows.
Q: Why is this method superior to using Lawson's Reagent for thioamide synthesis?
A: Traditional methods using Lawson's Reagent suffer from poor atom economy, high cost, instability, and significant odor issues. This novel catalytic approach utilizes cheap sublimed sulfur and benign formamide, operating under mild conditions (Rt-60°C) without the safety hazards associated with phosphorus pentasulfide derivatives.
Q: What represents the key innovation in the catalytic system described in CN112574081B?
A: The core innovation lies in the use of an alkali metal complex formed by combining an alkali metal salt (e.g., CsCl) with a ligand (e.g., EDTA or salicylaldehyde). This complex effectively activates the sulfur source and facilitates the oxidative thioamidation of benzyl alcohols at significantly lower temperatures than classical Willgestrom-Kindler reactions.
Q: Is this process suitable for large-scale industrial production of drug intermediates?
A: Yes, the process is highly scalable due to its one-pot nature, which eliminates intermediate isolation steps. The use of inexpensive, commercially available starting materials like benzyl alcohol and the absence of harsh reaction conditions (max 60°C) make it economically viable and safer for commercial scale-up compared to high-temperature alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Thioamide Supplier
The technological potential of the alkali metal complex catalyzed thioamidation route represents a significant opportunity for optimizing the production of sulfur-containing drug intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch. We understand that consistency is key in the pharmaceutical supply chain, and our robust process controls guarantee that our aryl thioamide products meet the highest international standards.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. We encourage you to contact our technical procurement team to obtain specific COA data for our catalog compounds and to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and innovation in the global supply of fine chemical intermediates.
