Revolutionizing Aryl Thioamide Production: A Green Catalytic Route for Commercial Scale-Up
The landscape of organic synthesis is constantly evolving, driven by the urgent need for greener, safer, and more economically viable pathways to access critical pharmaceutical building blocks. A significant breakthrough in this domain is documented in patent CN112574081A, which discloses a novel, highly efficient method for preparing aryl thioamide compounds. This technology represents a paradigm shift from traditional, hazardous硫化 processes to a mild, catalytic one-pot reaction that utilizes cheap and readily available starting materials. By leveraging an alkali metal complex catalyst system, this invention enables the direct conversion of aryl methanols into valuable thioamides using elemental sulfur and formamide. For R&D directors and procurement strategists alike, this development offers a compelling solution to long-standing challenges in impurity control, operational safety, and cost efficiency, positioning it as a cornerstone technology for modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamide functionalities has been plagued by significant technical and safety hurdles that complicate both laboratory research and industrial scale-up. The first conventional route typically relies on the thionation of pre-formed amides using reagents such as Lawesson's reagent or phosphorus pentasulfide (P2S5). These reagents are notoriously unstable, expensive, and generate substantial amounts of phosphorus-containing waste, posing severe environmental disposal challenges and health risks to operators due to their toxicity and unpleasant odor. Furthermore, this approach suffers from poor atom economy and requires a multi-step sequence where the amide must be synthesized separately from carboxylic acids or acyl chlorides before thionation can occur. The second traditional method, the Willgerodt-Kindler reaction, while avoiding some phosphorus reagents, often demands extremely harsh reaction conditions, with temperatures soaring up to 320°C. Such extreme thermal requirements not only consume excessive energy but also limit substrate compatibility, often leading to decomposition of sensitive functional groups and difficult purification processes.
The Novel Approach
In stark contrast to these legacy methods, the process described in CN112574081A introduces a remarkably mild and streamlined three-component coupling strategy. This innovative route directly utilizes aryl methanols (benzyl alcohols), sublimed sulfur, and formamide in a single reaction vessel, eliminating the need for pre-synthesized amides or dangerous thionating agents. The reaction proceeds smoothly at temperatures ranging from room temperature to merely 60°C, drastically reducing energy consumption and thermal stress on the molecular framework. By employing formamide as both the solvent and the nitrogen source, the process simplifies the reaction matrix and enhances atom utilization. The use of an alkali metal complex formed from simple salts and ligands acts as a powerful yet inexpensive catalyst, facilitating the transformation with high efficiency. This approach not only mitigates the safety risks associated with volatile organic solvents and toxic reagents but also significantly shortens the synthetic timeline, offering a robust platform for the rapid generation of diverse thioamide libraries.
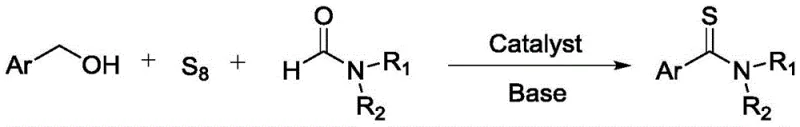
Mechanistic Insights into Alkali Metal Complex-Catalyzed Thioamidation
The core of this technological advancement lies in the sophisticated yet practical catalytic system involving an alkali metal salt paired with a specific ligand, such as cesium chloride with ethylenediaminetetraacetic acid (EDTA) or salicylaldehyde. Mechanistically, this catalyst system is believed to activate the elemental sulfur (S8), breaking its cyclic structure to generate reactive sulfur species capable of inserting into the carbon framework. Simultaneously, the base promoter facilitates the dehydrogenation or oxidation of the benzylic alcohol moiety, likely generating an aldehyde intermediate in situ which then undergoes condensation with the formamide and activated sulfur. The coordination environment provided by the ligand stabilizes the alkali metal cation, enhancing its Lewis acidity and ability to coordinate with the oxygen and sulfur atoms during the transition states. This precise tuning of the electronic environment allows the reaction to proceed under neutral to mildly basic conditions without the need for transition metals like palladium or copper, which are often costly and difficult to remove from final API intermediates to meet strict regulatory limits.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity aryl thioamides. Because the reaction avoids the use of phosphorus-based reagents, the final product is free from phosphine oxide byproducts that are notoriously difficult to separate and can interfere with downstream coupling reactions. The mild conditions also suppress side reactions such as over-oxidation to sulfones or polymerization of the aldehyde intermediates, which are common issues in high-temperature processes. The one-pot nature of the synthesis ensures that intermediate species are consumed immediately, minimizing the accumulation of unstable byproducts. Furthermore, the broad substrate tolerance demonstrated in the patent examples—ranging from electron-rich methoxy-substituted benzyl alcohols to electron-deficient nitro- and cyano-substituted analogs—suggests that the catalytic cycle is robust against various electronic effects. This reliability is crucial for maintaining consistent quality profiles across different batches, a key metric for supply chain stability in pharmaceutical manufacturing.
How to Synthesize Aryl Thioamide Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometric ratios and the order of addition to maximize yield and safety. The general protocol involves dissolving the aryl methanol substrate in formamide, followed by the sequential addition of sublimed sulfur and a base such as sodium hydroxide or potassium carbonate. The catalyst system, comprising the alkali metal salt and ligand, is then introduced to initiate the transformation. The reaction mixture is stirred under an inert atmosphere, typically nitrogen, to prevent unwanted oxidation of the sulfur or alcohol components. Detailed standard operating procedures regarding specific molar equivalents, stirring speeds, and workup protocols are essential for reproducibility.
- Under inert gas protection, combine aryl methanol, sublimed sulfur, and a base (e.g., NaOH) in formamide solvent.
- Add the alkali metal salt catalyst and ligand (e.g., CsCl and EDTA), then stir at room temperature to 60°C for 6-12 hours.
- Quench with water, extract with ethyl acetate, wash with brine, dry, and purify via column chromatography to isolate the target thioamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic thioamidation process translates into tangible strategic benefits that extend far beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing expensive, specialty reagents like Lawesson's reagent with commodity chemicals such as benzyl alcohol, elemental sulfur, and formamide, manufacturers can significantly reduce raw material costs and mitigate the risk of supply disruptions. These starting materials are produced on a massive global scale, ensuring consistent availability and price stability, which is critical for long-term project planning. Moreover, the elimination of hazardous phosphorus reagents removes the need for specialized waste treatment facilities and expensive disposal contracts, further driving down the total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several cost-saving factors. Firstly, the catalyst system utilizes earth-abundant alkali metals and simple organic ligands, avoiding the volatility and high expense associated with precious transition metal catalysts. Secondly, the reaction operates at near-ambient temperatures, leading to substantial savings in energy consumption compared to processes requiring reflux or high-pressure autoclaves. Thirdly, the one-pot design eliminates the isolation and purification of intermediate amides, reducing solvent usage, labor hours, and equipment occupancy time. These cumulative efficiencies result in a leaner manufacturing process that delivers high-purity products at a fraction of the cost of traditional methods, enhancing the overall margin profile for the final API.
- Enhanced Supply Chain Reliability: In the context of global pharmaceutical supply chains, reliability is paramount. This synthesis route enhances reliability by decoupling production from the supply of niche, hazardous reagents that often face regulatory scrutiny or logistical bottlenecks. The use of stable, non-hazardous solids like sublimed sulfur and common liquid solvents simplifies storage and transportation requirements, reducing the risk of delays due to hazardous material shipping restrictions. Additionally, the robustness of the reaction across a wide range of substrates means that a single manufacturing line can be flexibly adapted to produce various thioamide derivatives without extensive retooling or requalification. This flexibility allows suppliers to respond rapidly to changing market demands and ensures continuity of supply even when specific precursor availability fluctuates.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but this method is inherently designed for scalability. The absence of exothermic spikes and the use of low-viscosity formamide solvent facilitate efficient heat transfer and mixing in large reactors. From an environmental compliance standpoint, the process aligns perfectly with green chemistry principles by maximizing atom economy and minimizing waste generation. The lack of toxic phosphorus byproducts simplifies effluent treatment, making it easier for manufacturing sites to meet stringent environmental regulations. This 'green' credential is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies who are committed to sustainable sourcing, thereby opening up new business opportunities for suppliers who adopt this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aryl thioamide synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the safety advantages of this thioamide synthesis compared to traditional methods?
A: Unlike traditional methods using toxic Lawesson's reagent or phosphorus pentasulfide which generate hazardous waste, this process utilizes stable elemental sulfur and formamide, significantly reducing operator risk and environmental impact.
Q: Can this process accommodate diverse substrate structures for drug discovery?
A: Yes, the method demonstrates strong substrate universality, successfully converting various substituted benzyl alcohols (including electron-withdrawing and electron-donating groups) and heteroaromatic alcohols into corresponding thioamides with good yields.
Q: Is this catalytic system suitable for large-scale industrial production?
A: The process operates at mild temperatures (Rt-60°C) and atmospheric pressure using inexpensive, commercially available catalysts, making it highly scalable and cost-effective for industrial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Thioamide Supplier
The transition from academic innovation to industrial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering comprehensive CDMO services tailored to the commercialization of advanced synthetic routes like the alkali metal-catalyzed thioamidation process. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the laboratory are faithfully reproduced on a manufacturing scale. We understand that high-purity aryl thioamides are critical intermediates for a wide array of bioactive molecules, and our rigorous QC labs are equipped to handle stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite R&D leaders and procurement professionals to engage with us for a Customized Cost-Saving Analysis specific to your target molecules. By leveraging this novel catalytic technology, we can help you optimize your supply chain, reduce manufacturing costs, and accelerate your time-to-market. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and detailed project timelines. Let us collaborate to transform your chemical challenges into commercial successes through innovation and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
