Advanced Rh(III)-Catalyzed Synthesis of Quinoline Amides for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to construct complex amide scaffolds, which serve as critical backbones in countless bioactive molecules. Patent CN107721920B, published in July 2020, introduces a groundbreaking methodology for the synthesis of quinoline amide compounds that addresses long-standing challenges in C-H bond functionalization. This technology leverages a transition metal catalyst, specifically a Rh(III) complex, to directly catalyze the insertion of isocyanates into inert C(sp3)-H bonds. Unlike traditional methods that require harsh pre-functionalization steps, this approach operates under mild conditions with exceptional atom economy, ensuring that no atoms are wasted during the transformation. The ability to synthesize these valuable intermediates in a single reactor without intermediate separation steps represents a significant leap forward in green chemistry and process intensification.
For R&D directors evaluating new synthetic routes, the mechanistic elegance of this Rh(III)-catalyzed system offers a compelling alternative to legacy chemistries. Conventional methods for preparing aromatic amides typically rely on the coupling of carboxylic acids or their activated derivatives with amines, processes that often necessitate the use of stoichiometric coupling reagents, strong oxidants, or protection-deprotection sequences. These traditional pathways are not only costly due to the price of activating agents but also generate substantial amounts of chemical waste, complicating downstream purification and environmental compliance. Furthermore, the requirement for pre-functionalized starting materials limits the structural diversity that can be accessed rapidly during lead optimization phases, creating a bottleneck in the drug discovery pipeline.
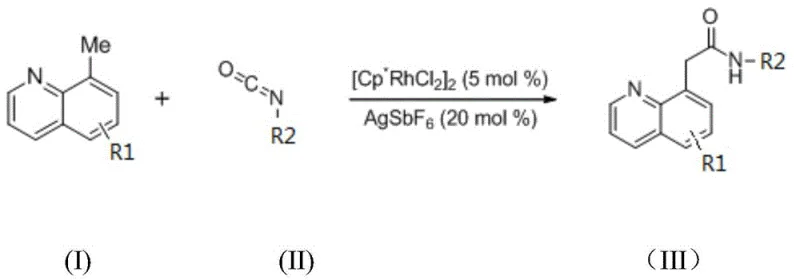
In stark contrast, the novel approach detailed in the patent utilizes a direct C-H activation strategy that bypasses the need for pre-installed leaving groups. By employing a cationic Rh(III) species generated in situ from [Cp*RhCl2]2 and AgSbF6, the system activates the methyl C-H bond at the 8-position of the quinoline ring with high regioselectivity. This catalytic cycle allows for the direct insertion of the isocyanate moiety, forming the new carbon-nitrogen bond in a single operational step. The reaction demonstrates broad substrate scope, tolerating various electronic and steric environments on both the quinoline and isocyanate components, which is crucial for generating diverse libraries of analogs. This shift from step-intensive classical synthesis to direct catalytic functionalization fundamentally alters the cost and time structure of producing these key intermediates.
Mechanistic Insights into Rh(III)-Catalyzed C(sp3)-H Amidation
The success of this transformation hinges on the unique ability of the Rh(III) catalyst to overcome the kinetic barrier associated with breaking strong aliphatic C-H bonds. The catalytic cycle initiates with the coordination of the quinoline nitrogen to the rhodium center, directing the metal to the adjacent methyl group. Through a concerted metalation-deprotonation or sigma-bond metathesis pathway, the C(sp3)-H bond is cleaved to form a stable five-membered rhodacycle intermediate. This step is facilitated by the specific electronic properties of the pentamethylcyclopentadienyl ligand, which stabilizes the high-valent metal center while allowing sufficient lability for substrate binding. The subsequent coordination and insertion of the isocyanate into the Rh-C bond is the turnover-limiting step, where the electrophilic carbon of the isocyanate attacks the nucleophilic rhodium-carbon bond.
Following the insertion event, protonolysis of the resulting Rh-N species releases the final quinoline amide product and regenerates the active Rh(III) catalyst, closing the catalytic loop. A critical component of this mechanism is the role of the silver salt additive, AgSbF6, which acts as a halide scavenger to generate the coordinatively unsaturated cationic rhodium species essential for high reactivity. Without this additive, the neutral rhodium precursor exhibits significantly lower activity, as evidenced by comparative experiments in the patent data. The choice of solvent, specifically 1,2-dichloroethane (DCE), further optimizes the reaction environment by stabilizing the charged intermediates without interfering with the catalytic cycle, unlike coordinating solvents like THF or DMF which can poison the catalyst. This deep understanding of the mechanistic nuances allows for precise tuning of reaction parameters to maximize yield and purity.
How to Synthesize Quinoline Amide Efficiently
Implementing this synthesis route requires strict adherence to the optimized conditions identified during the patent's development to ensure reproducible high yields. The process is designed as a one-pot reaction, meaning all reagents including the catalyst, additive, substrates, and solvent are combined initially, eliminating the need for handling sensitive intermediates. The reaction is typically conducted under an inert nitrogen atmosphere to prevent oxidation of the catalyst or substrates, maintaining the integrity of the active species throughout the 24-hour heating period at 60°C. Following the reaction completion, standard workup procedures involving column chromatography are employed to isolate the pure product, leveraging the high conversion rates to minimize purification losses. For detailed operational parameters and safety considerations, please refer to the standardized protocol below.
- Charge a reaction vessel with 5 mol% [Cp*RhCl2]2 catalyst, 20 mol% AgSbF6 additive, 8-methylquinoline substrate, and isocyanate coupling partner in DCE solvent.
- Maintain the reaction mixture under nitrogen atmosphere at 60°C for 24 hours to ensure complete conversion via C-H activation.
- Upon completion, isolate the target quinoline amide product through standard column chromatography purification techniques.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this Rh(III)-catalyzed methodology offers distinct advantages that translate directly into operational efficiency and cost competitiveness. The elimination of pre-activation steps means that manufacturers can source simpler, more commoditized starting materials rather than expensive, specialized building blocks. This simplification of the bill of materials reduces the complexity of the supply chain, mitigating risks associated with the availability of niche reagents. Furthermore, the high atom economy inherent in direct C-H functionalization ensures that a larger proportion of the input mass ends up in the final product, drastically reducing the volume of waste streams that require treatment and disposal. This aligns perfectly with modern sustainability goals and regulatory pressures facing the fine chemical industry.
- Cost Reduction in Manufacturing: The removal of stoichiometric coupling reagents and the reduction in synthetic steps lead to a substantial decrease in raw material costs. By avoiding the purchase of expensive activating agents and reducing solvent consumption through a one-pot design, the overall cost of goods sold is significantly optimized. Additionally, the high yields reported, often exceeding 90 percent under optimal conditions, mean that less starting material is required to produce a fixed amount of product, further enhancing economic efficiency without compromising quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available 8-methylquinoline derivatives and commercial isocyanates ensures a robust and resilient supply chain. These starting materials are produced at scale by multiple global suppliers, reducing the risk of single-source bottlenecks. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, removing the need for specialized high-pressure or cryogenic equipment that could limit production capacity or extend lead times for campaign scheduling.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and uses common chlorinated solvents that are well-understood in industrial settings, facilitating straightforward scale-up from laboratory to commercial tonnage. The absence of heavy metal waste, aside from the trace rhodium catalyst which can be recovered, simplifies effluent treatment protocols. This environmental profile supports faster regulatory approvals and reduces the long-term liability associated with hazardous waste management, making it an attractive option for sustainable manufacturing partnerships.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational requirements. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is critical for successful technology transfer and risk mitigation during the scale-up phase.
Q: What is the primary advantage of this Rh(III) catalytic method?
A: This method enables direct C(sp3)-H bond functionalization without pre-activation, significantly improving atom economy and reducing waste compared to traditional amidation routes.
Q: What catalyst system is required for this transformation?
A: The process utilizes a pentamethylcyclopentadienyl rhodium(III) chloride dimer [Cp*RhCl2]2 combined with silver hexafluoroantimonate (AgSbF6) as a critical additive.
Q: Is this method scalable for industrial production?
A: Yes, the reaction operates under mild temperatures (60°C) in common solvents like DCE, avoiding hazardous reagents and facilitating easier scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating the delivery of high-value intermediates to the market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this Rh(III)-catalyzed process can be successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global pharmaceutical and agrochemical clients, providing you with a secure and compliant supply source.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can optimize your supply chain and reduce your overall manufacturing costs while ensuring consistent quality and delivery performance.
