Advanced Copper-Catalyzed C-H Sulfonylation for High-Purity Aryl Sulfone Manufacturing
Advanced Copper-Catalyzed C-H Sulfonylation for High-Purity Aryl Sulfone Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex molecular architectures, particularly those containing sulfone motifs which are ubiquitous in bioactive molecules. Patent CN111393364B, published in July 2021, introduces a groundbreaking synthetic methodology for constructing C(sp2)-S bonds through a direct sulfonylation strategy. This technology represents a significant leap forward in organic synthesis, specifically targeting the preparation of aryl sulfone compounds using 5-chloro-8-aminoquinoline aromatic amides as key substrates. Unlike conventional approaches that often rely on harsh conditions or multi-step sequences, this invention leverages a transition metal-catalyzed C-H activation protocol. By utilizing the 5-chloro-8-aminoquinoline moiety as an effective directing group, the process achieves high regioselectivity and functional group tolerance. For R&D directors and process chemists, this patent offers a compelling route to access high-purity aryl sulfone intermediates with reduced environmental impact, addressing the growing demand for greener and more cost-effective manufacturing processes in the production of advanced pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfone compounds has been fraught with significant chemical and operational challenges that hinder efficient large-scale production. Traditional methods typically involve the oxidation of sulfides or sulfoxides, which necessitates the preparation of sulfur-containing precursors through separate, often hazardous, synthetic steps. Alternatively, classical approaches such as the Ramberg-Backlund reaction or Julia olefination, while useful for specific transformations, often suffer from poor regioselectivity and limited functional group compatibility. Furthermore, many existing protocols require the use of strong acidic conditions for the sulfonylation of aromatic hydrocarbons or the addition of sulfonyl radical precursors to alkenes and alkynes. These conditions can lead to the degradation of sensitive functional groups, resulting in complex impurity profiles that are difficult and costly to remove during downstream purification. The reliance on stoichiometric oxidants or pre-functionalized starting materials not only increases the raw material costs but also generates substantial chemical waste, posing severe environmental compliance issues for modern manufacturing facilities striving for sustainability.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111393364B offers a streamlined, direct C-H bond functionalization strategy that fundamentally simplifies the synthetic workflow. The core innovation lies in the use of a copper-catalyzed system that activates inert C(sp2)-H bonds directly, bypassing the need for pre-halogenation or other activating groups on the aromatic ring. As illustrated in the general reaction scheme below, the process couples 5-chloro-8-aminoquinoline aromatic amides with readily available aryl sulfinates under mild aerobic conditions.

This novel approach utilizes air as the terminal oxidant, effectively replacing expensive and dangerous chemical oxidants like peroxides or hypervalent iodine reagents. The 5-chloro-8-aminoquinoline group acts as a powerful bidentate directing group, coordinating with the copper catalyst to ensure precise ortho-selectivity during the C-H activation step. This level of control minimizes the formation of regioisomers, thereby drastically simplifying the purification process and improving overall yield. For procurement managers, this translates to a supply chain less dependent on exotic reagents and more focused on commodity chemicals, significantly enhancing supply security and cost predictability.
Mechanistic Insights into Copper-Catalyzed C-H Sulfonylation
The mechanistic elegance of this transformation relies on a synergistic interplay between the copper catalyst, the base, and the coordinating ligands to achieve efficient C-S bond formation. The reaction initiates with the coordination of the copper(II) species, typically copper(II) acetate, to the nitrogen atoms of the 5-chloro-8-aminoquinoline directing group. This coordination brings the metal center into close proximity with the target ortho-C-H bond, facilitating a concerted metalation-deprotonation (CMD) process assisted by the pivalate base. The resulting organocopper intermediate is then intercepted by the aryl sulfinate species, leading to the insertion of the sulfonyl group. Crucially, the regeneration of the active copper(II) catalyst is achieved through oxidation by molecular oxygen from the air, closing the catalytic cycle without generating stoichiometric metal waste. This mechanism not only ensures high turnover numbers but also maintains a clean reaction profile, as the only byproduct of the oxidation step is water.
From an impurity control perspective, the high selectivity imparted by the quinoline directing group is paramount. In traditional electrophilic aromatic substitution, mixtures of ortho, meta, and para isomers are common, requiring energy-intensive separation techniques like preparative HPLC or repeated recrystallizations. In this copper-catalyzed system, the rigid coordination geometry effectively blocks alternative reaction sites, ensuring that the sulfonyl group is installed exclusively at the desired position. This inherent selectivity reduces the burden on quality control laboratories and ensures that the final active pharmaceutical ingredient (API) meets stringent purity specifications with minimal effort. Furthermore, the compatibility of the reaction with various functional groups—such as halogens, alkyl chains, and ethers—allows for late-stage functionalization, enabling medicinal chemists to rapidly generate diverse analog libraries for structure-activity relationship (SAR) studies without protecting group manipulations.
How to Synthesize Aryl Sulfone Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction parameters to maximize efficiency. The patent provides detailed experimental procedures that serve as a robust foundation for process optimization. The standard protocol involves dissolving the 5-chloro-8-aminoquinoline aromatic amide substrate and the aryl sulfinate salt in a polar aprotic solvent such as DMF or DMA. To this mixture, the copper catalyst, benzoic acid additive, and potassium pivalate base are added in specific molar ratios optimized for high conversion. The reaction is then heated to a moderate temperature, typically around 80°C, and stirred under an open atmosphere to allow for continuous oxygen exchange. Below is a representative example of the reaction conditions and outcome, demonstrating the practicality of the method.
- Charge a reaction vessel with DMF solvent, 5-chloro-8-aminoquinoline aromatic amide substrate, aryl sulfinate salt, copper(II) acetate catalyst, benzoic acid additive, and potassium pivalate base.
- Heat the reaction mixture to 80°C in an oil bath and stir for approximately 8 hours under air atmosphere to facilitate oxidative sulfonylation.
- Upon completion, cool the mixture, dilute with ethyl acetate, wash with water and brine, dry over anhydrous sodium sulfate, and purify via silica gel chromatography.
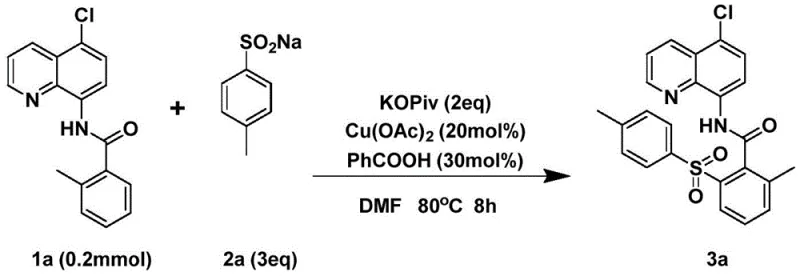
As shown in the specific example above, the reaction proceeds smoothly to afford the desired aryl sulfone product in high yield (80%) and excellent purity (>95%) after a simple workup involving aqueous extraction and silica gel chromatography. This operational simplicity is a key driver for adoption, as it reduces the technical barrier for implementation across different manufacturing scales.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic viability and continuity of chemical supply chains, the adoption of this copper-catalyzed sulfonylation technology offers transformative benefits. The shift from multi-step, pre-functionalized routes to a direct C-H activation strategy fundamentally alters the cost structure of aryl sulfone production. By eliminating the need for separate halogenation or lithiation steps, the overall number of unit operations is significantly reduced. This consolidation of steps leads to a drastic reduction in solvent consumption, energy usage, and labor hours, all of which are primary drivers of manufacturing costs. Furthermore, the use of air as the oxidant removes the procurement risk and safety hazards associated with storing and handling large quantities of hazardous chemical oxidants, thereby lowering insurance and compliance costs.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the simplification of the synthetic route. Traditional methods often require expensive palladium catalysts or stoichiometric amounts of silver salts, whereas this method utilizes abundant and low-cost copper salts. Additionally, the high atom economy of the direct coupling reaction means that a larger proportion of the raw material mass ends up in the final product, minimizing waste disposal fees. The elimination of pre-functionalization steps also reduces the cumulative yield loss typically seen in linear synthetic sequences, effectively increasing the overall throughput of the manufacturing line without requiring additional capital investment in new equipment.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available commodity chemicals. Sodium aryl sulfinates and 5-chloro-8-aminoquinoline derivatives are commercially accessible from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of substituents and moisture to some extent, ensures consistent batch-to-batch quality even when raw material specifications vary slightly. This reliability is critical for maintaining uninterrupted production schedules for downstream API manufacturing, preventing costly delays and stockouts that can disrupt the entire pharmaceutical value chain.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the mild reaction conditions and the absence of exothermic hazards associated with strong oxidants. The use of DMF or DMA as solvents allows for high substrate concentrations, improving reactor productivity. From an environmental standpoint, the replacement of toxic oxidants with air aligns perfectly with green chemistry principles, significantly reducing the E-factor (mass of waste per mass of product). This environmental advantage simplifies the permitting process for new manufacturing lines and helps companies meet increasingly stringent corporate sustainability goals and regulatory emissions standards.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational parameters. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what can be expected during process development. Understanding these nuances is essential for accurately projecting timelines and resource requirements for potential scale-up campaigns.
Q: What are the primary advantages of this C-H sulfonylation method over traditional sulfide oxidation?
A: This method eliminates the need for pre-functionalization steps required in traditional sulfide oxidation or sulfonyl chloride additions. By utilizing direct C-H bond activation guided by a quinoline moiety, it significantly reduces synthetic steps, improves atom economy, and avoids the use of harsh strong acids or unstable radical precursors often associated with older methodologies.
Q: What specific catalyst system enables this transformation?
A: The reaction utilizes a robust copper catalytic system comprising copper(II) acetate as the metal source, potassium pivalate (KOPiv) as the base, and benzoic acid as a coordinating ligand. This specific combination facilitates the critical C-H activation and subsequent sulfonylation under mild aerobic conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly amenable to scale-up. It employs inexpensive and commercially available raw materials, operates at moderate temperatures (80°C), and uses air as the terminal oxidant, which removes the safety hazards and costs associated with stoichiometric chemical oxidants. The simple workup procedure further supports industrial viability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the copper-catalyzed C-H sulfonylation described in CN111393364B. As a leading CDMO partner, we possess the technical expertise to translate such innovative academic and patent literature into robust, commercial-grade manufacturing processes. Our team of process chemists is adept at optimizing reaction conditions, screening alternative solvents for greener profiles, and developing scalable crystallization processes to ensure the highest quality standards. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive impurity profiling and stringent purity specifications analysis to guarantee that every batch meets your exact requirements.
We invite you to leverage our capabilities to accelerate your drug development timeline. Whether you require custom synthesis of complex aryl sulfone intermediates or full-scale commercial manufacturing, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule. Contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how we can collaborate to bring your next-generation therapeutics to market faster and more efficiently.
