Advanced Synthesis of Cytosine Nucleoside: A Cost-Effective Route for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical nucleoside intermediates, and the technology disclosed in patent CN112480197B represents a significant leap forward in the manufacturing of cytosine nucleoside. This innovative method shifts the paradigm from relying on expensive natural nucleosides or complex protected bases to utilizing cyanoacetaldehyde urea acetal as a foundational building block. By employing a strategic sequence of silicon etherification followed by Lewis acid-catalyzed condensation and a final one-pot cyclization-deprotection step, this process addresses long-standing inefficiencies in nucleoside chemistry. For R&D directors and procurement specialists, this patent offers a compelling alternative that promises not only high chemical fidelity but also substantial economic advantages through simplified operations and reduced raw material costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cytosine nucleoside has been plagued by significant technical and economic hurdles that hinder efficient commercial production. Traditional routes often rely on starting materials such as uridine, which is a natural nucleoside with limited availability and high market price, creating a bottleneck for large-scale supply chains. Alternative methods involving N4-acetyl cytosine require complex protection and deprotection strategies that frequently result in low overall yields and difficult post-reaction processing. Furthermore, older methodologies, such as those proposed by Nishimura et al., often generate mixtures of alpha and beta anomers, necessitating tedious and costly chromatographic separations to isolate the biologically active beta-configuration. These conventional processes typically involve harsh reaction conditions, including high-pressure ammonolysis at elevated temperatures, which pose safety risks and increase energy consumption, thereby inflating the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes cyanoacetaldehyde urea acetal, a readily available and cost-effective precursor that bypasses the need for expensive natural nucleosides. This method streamlines the synthesis into a continuous three-step sequence where the intermediate does not require isolation until the final stage, significantly reducing solvent usage and processing time. The core innovation lies in the use of Lewis acid catalysis to facilitate the glycosidic bond formation between the silylated urea acetal and tetraacetyl ribose, ensuring high stereoselectivity and yield. By integrating the cyclization and deprotection steps into a single pot using sodium alkoxide, the process eliminates multiple workup stages, enhancing operational simplicity and stability. This streamlined workflow is particularly advantageous for a reliable pharmaceutical intermediate supplier aiming to optimize production throughput while maintaining rigorous quality standards.
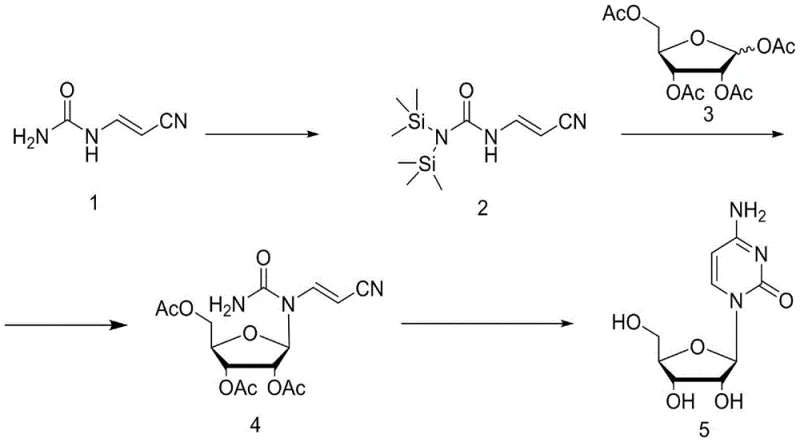
Mechanistic Insights into Lewis Acid-Catalyzed Glycosylation
The chemical elegance of this synthesis lies in its precise control over reactivity and stereochemistry through Lewis acid catalysis. The process initiates with the silylation of cyanoacetaldehyde urea acetal using reagents like hexamethyldisilazane (HMDS) or trimethylchlorosilane (TMCS), which activates the nitrogen nucleophile for subsequent attack. In the critical condensation step, a Lewis acid such as stannic chloride (SnCl4) or titanium tetrachloride (TiCl4) coordinates with the anomeric acetate of tetraacetyl ribose, generating a highly reactive oxocarbenium ion intermediate. This electrophilic species is then attacked by the silylated urea derivative, forming the N-glycosidic bond with high regioselectivity. The choice of Lewis acid and the maintenance of low temperatures during this addition are paramount to suppressing side reactions and ensuring the formation of the desired beta-anomer, which is crucial for the biological activity of the final nucleoside product.
Following the condensation, the reaction mixture undergoes a transformative cyclization and deprotection phase mediated by sodium alkoxide in an alcoholic solvent. The alkoxide base serves a dual function: it catalyzes the intramolecular cyclization of the cyanoacetaldehyde moiety to form the pyrimidine ring of the cytosine base, and simultaneously hydrolyzes the acetyl protecting groups on the ribose sugar. This tandem reaction mechanism is highly efficient, converting the linear precursor directly into the target cytosine nucleoside without the need for intermediate isolation. The resulting crude product is of sufficient quality that high-purity pharmaceutical intermediates can be obtained simply through recrystallization from ethanol, demonstrating the robustness of the impurity control mechanism inherent in this synthetic design.
How to Synthesize Cytosine Nucleoside Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of reagent stoichiometry and temperature control. The process begins with the reflux of cyanoacetaldehyde urea acetal with HMDS in dichloroethane, followed by the direct addition of tetraacetyl ribose and the Lewis acid catalyst at sub-zero temperatures to manage exothermicity. After the condensation is complete, the reaction mixture is quenched and processed to yield the key intermediate, which is then subjected to reflux with sodium methoxide in methanol to effect cyclization. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios required for GMP-compliant manufacturing, please refer to the standardized synthesis steps provided below.
- Perform silylation of cyanoacetaldehyde urea acetal using hexamethyldisilazane (HMDS) or trimethylchlorosilane (TMCS) in a suitable solvent like dichloroethane.
- Conduct condensation of the silylated intermediate with tetraacetyl ribose under Lewis acid catalysis (e.g., SnCl4) at controlled low temperatures.
- Execute one-pot cyclization and deprotection by heating the condensation product with sodium alkoxide (e.g., sodium methoxide) in alcohol solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound benefits that directly address the pain points of procurement managers and supply chain heads in the fine chemical sector. The substitution of expensive starting materials like uridine or protected cytosine with cyanoacetaldehyde urea acetal results in a drastic reduction in raw material expenditure, which is a primary driver of overall manufacturing costs. Furthermore, the elimination of chromatographic purification steps in favor of simple recrystallization significantly lowers the consumption of silica gel and organic solvents, reducing both waste disposal costs and environmental impact. This efficiency translates into a more competitive pricing structure for the final API intermediate, allowing downstream pharmaceutical manufacturers to optimize their own cost structures without compromising on quality or supply reliability.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals and the minimization of unit operations. By avoiding the need for high-pressure reactors and expensive biochemical reagents, the capital expenditure required for setting up production lines is substantially lowered. The ability to perform the cyclization and deprotection in a single pot reduces labor hours and energy consumption associated with heating and cooling cycles. Additionally, the high yield and purity achieved reduce the loss of valuable material during purification, ensuring that the maximum amount of input mass is converted into saleable product, thereby maximizing return on investment for chemical manufacturing projects.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available industrial solvents such as dichloroethane, methanol, and ethanol, which are less susceptible to market volatility compared to specialized biochemical reagents. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, ensuring consistent batch-to-batch performance. This stability is critical for maintaining continuous production schedules and meeting the stringent delivery timelines required by global pharmaceutical clients. The simplified workflow also reduces the risk of production bottlenecks, facilitating a smoother flow of materials from synthesis to packaging and distribution.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, featuring straightforward workup procedures that are easily adaptable to large reactor volumes. The reduction in solvent usage and the avoidance of heavy metal catalysts in the final steps align with increasingly strict environmental regulations regarding waste discharge and residual metals in drug substances. The ability to recover and recycle solvents like dichloroethane further enhances the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only mitigates regulatory risks but also appeals to environmentally conscious stakeholders and partners in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs and quality requirements.
Q: What are the primary advantages of this synthesis method over traditional uridine-based routes?
A: This method utilizes cyanoacetaldehyde urea acetal, a significantly cheaper and more accessible raw material compared to uridine or protected cytosine derivatives. It avoids harsh high-pressure ammonolysis conditions and complex chromatographic separations, relying instead on simple recrystallization for purification.
Q: What purity levels can be achieved with this process?
A: The patent data indicates that the final product can achieve an HPLC purity of greater than 99.5% after a single recrystallization step from ethanol, meeting stringent pharmaceutical intermediate specifications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It features stable yields around 80%, uses common industrial solvents like dichloroethane and methanol, and eliminates the need for column chromatography, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cytosine Nucleoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our partners receive a consistent and high-quality supply of essential intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of cytosine nucleoside meets the highest international standards. We are committed to leveraging advanced technologies like the one described in CN112480197B to drive innovation and efficiency in our manufacturing processes.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this cost-effective synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while optimizing your supply chain economics.
