Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks efficient and scalable routes to access privileged heterocyclic scaffolds that serve as the core structures for potent bioactive molecules. Among these, quinolin-2(1H)-one derivatives stand out due to their prevalence in natural products and their critical role in modern drug discovery programs targeting various therapeutic areas. As detailed in patent CN113045489B, a significant technological breakthrough has been achieved in the synthesis of 3-arylquinolin-2(1H)-one derivatives through a novel palladium-catalyzed aminocarbonylation strategy. This method addresses long-standing challenges in heterocycle synthesis by utilizing benzisoxazole as a versatile dual-purpose reagent, providing both the nitrogen atom and the formyl moiety required for ring construction. The implications of this development extend far beyond academic interest, offering a practical solution for the commercial scale-up of complex pharmaceutical intermediates with high efficiency and operational simplicity.
The biological significance of the quinolinone core cannot be overstated, as it forms the backbone of numerous clinical candidates and approved drugs ranging from antibiotics to antitumor agents. For instance, specific derivatives within this class have demonstrated potent activity as MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors, highlighting the urgent need for reliable supply chains capable of delivering these structures in high purity. By leveraging the innovative approach disclosed in the patent data, manufacturers can now access these valuable scaffolds through a streamlined process that avoids the harsh conditions and toxic reagents often associated with classical heterocyclic synthesis. This positions the technology as a cornerstone for cost reduction in API manufacturing, enabling faster progression from lead optimization to clinical supply.
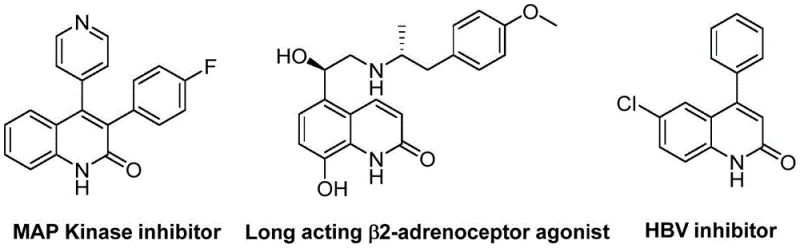
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)-one skeleton has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods are well-documented in organic chemistry textbooks, they frequently suffer from significant drawbacks when applied to the synthesis of complex, functionalized derivatives required by modern medicinal chemistry. Traditional approaches often necessitate the use of corrosive reagents, extreme temperatures, or highly reactive precursors that limit the scope of compatible functional groups. Furthermore, these legacy processes typically exhibit poor atom economy and generate substantial amounts of chemical waste, creating environmental burdens and increasing the cost of waste disposal for large-scale production facilities. The inability of these conventional methods to tolerate sensitive functionalities often forces chemists to employ lengthy protection-deprotection sequences, thereby reducing overall yield and extending production timelines.
The Novel Approach
In stark contrast to these outdated techniques, the palladium-catalyzed aminocarbonylation method described in patent CN113045489B represents a paradigm shift in heterocyclic synthesis. This innovative route utilizes readily available benzisoxazoles and benzyl chlorides as starting materials, reacting them under mild conditions in the presence of a palladium catalyst and molybdenum hexacarbonyl as a safe carbon monoxide surrogate. The reaction proceeds efficiently at 100°C in dimethoxyethane (DME) solvent, demonstrating remarkable compatibility with a wide array of substituents including halogens, alkoxy groups, and electron-withdrawing moieties like cyano and trifluoromethyl groups. This broad substrate scope eliminates the need for complex substrate modification prior to the cyclization step, significantly simplifying the synthetic pathway. By integrating the nitrogen and carbonyl sources into a single benzisoxazole molecule, the process reduces the number of reagents required and minimizes side reactions, resulting in a cleaner reaction profile and higher isolated yields.
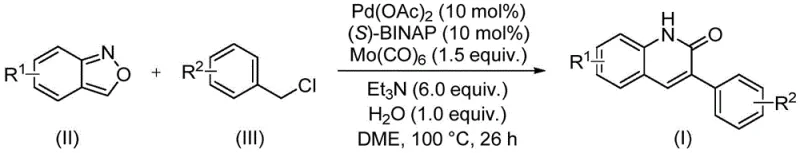
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The success of this transformation relies on a sophisticated catalytic cycle driven by the synergistic interaction between the palladium center and the specialized ligand system. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, which is generated in situ from palladium acetate and the chiral phosphine ligand (S)-BINAP. Simultaneously, molybdenum hexacarbonyl serves as a solid source of carbon monoxide, releasing CO gas slowly into the reaction medium to facilitate the carbonylation step without the safety hazards associated with handling high-pressure CO cylinders. The insertion of the carbonyl group into the palladium-carbon bond creates an acyl-palladium intermediate, which subsequently undergoes nucleophilic attack by the nitrogen species derived from the benzisoxazole ring opening. This intricate sequence of elementary steps ensures the precise formation of the quinolinone core while maintaining the integrity of sensitive functional groups present on the aromatic rings.
Furthermore, the role of water and triethylamine in the reaction mixture is critical for driving the equilibrium towards the desired product and neutralizing the hydrochloric acid byproduct generated during the dehydrohalogenation of the benzyl chloride. The presence of water facilitates the hydrolysis of the intermediate species, effectively unmasking the formyl group from the benzisoxazole precursor and enabling the final cyclization to occur. This mechanistic understanding allows for precise control over impurity profiles, as the mild basic conditions prevent the degradation of acid-sensitive groups that might otherwise decompose under the harsh acidic conditions of traditional Friedlander syntheses. Consequently, the resulting crude product contains fewer byproducts, simplifying the downstream purification process and ensuring that the final high-purity pharmaceutical intermediates meet stringent quality specifications required for regulatory submission.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the catalytic components and the reaction parameters to maximize efficiency. The standard protocol involves charging a sealed vessel with the palladium catalyst, ligand, carbonyl source, base, and substrates in the appropriate solvent, followed by heating to the optimized temperature for a defined period. The robustness of the method allows for flexibility in scaling, as the reaction kinetics remain consistent across different batch sizes provided that mixing and heat transfer are adequately managed. Operators should note that the use of sealed tubes or autoclaves is necessary to retain the volatile solvent and carbon monoxide generated in situ, ensuring that the reaction proceeds to completion without loss of pressure. For detailed operational parameters and specific workup procedures tailored to your facility's capabilities, please refer to the standardized guide below.
- Charge a sealed reactor with palladium acetate, (S)-BINAP ligand, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole substrate, and benzyl chloride derivative in dimethoxyethane (DME) solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and perform column chromatography purification to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic methodology offers compelling advantages that directly address the pain points of sourcing complex heterocyclic building blocks. The reliance on commercially available and inexpensive starting materials, such as substituted benzisoxazoles and benzyl chlorides, mitigates the risk of supply chain disruptions often caused by bespoke or hard-to-source reagents. Because the raw materials are commodity chemicals produced by multiple vendors globally, procurement managers can leverage competitive bidding to secure favorable pricing and ensure continuity of supply. Additionally, the simplified reaction workflow reduces the demand for specialized equipment or hazardous gas handling infrastructure, lowering the barrier to entry for contract manufacturing organizations (CMOs) and enabling a broader base of qualified suppliers to participate in the production network.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal scavengers and the reduction in purification steps significantly lower the overall cost of goods sold (COGS). By avoiding the use of gaseous carbon monoxide cylinders, the process removes the need for specialized high-pressure reactors and the associated safety compliance costs, leading to substantial capital expenditure savings. Furthermore, the high atom economy of the reaction means that less raw material is wasted, translating directly into improved material utilization rates and reduced expenditure on starting materials per kilogram of final product.
- Enhanced Supply Chain Reliability: The broad functional group tolerance of this method ensures that supply chains remain resilient even when specific substituted analogs are required for structure-activity relationship (SAR) studies. Since the reaction conditions are mild and do not degrade sensitive moieties, manufacturers can produce a diverse library of derivatives from a common set of intermediates without needing to qualify entirely new synthetic routes for each variant. This flexibility allows supply chain heads to consolidate inventory and reduce lead times for high-purity intermediates, ensuring that R&D pipelines are not delayed by material shortages.
- Scalability and Environmental Compliance: The use of dimethoxyethane (DME) as a solvent and the absence of highly toxic reagents simplify the waste management process, facilitating compliance with increasingly stringent environmental regulations. The reaction generates minimal hazardous waste compared to traditional methods, reducing the costs associated with waste treatment and disposal. Moreover, the straightforward workup procedure involving filtration and chromatography is easily adaptable to large-scale industrial processes, ensuring that the technology can be seamlessly transferred from gram-scale laboratory optimization to multi-kilogram commercial production without loss of efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational boundaries and capabilities of the technology. Understanding these details is crucial for project managers evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: What is the unique role of benzisoxazole in this synthetic route?
A: In this novel methodology, benzisoxazole serves a dual function as both the nitrogen source and the formyl group donor, which significantly simplifies the reagent requirements compared to traditional multi-component couplings.
Q: What catalytic system is employed for this transformation?
A: The reaction utilizes a robust palladium catalytic system comprising palladium acetate and the chiral ligand (S)-BINAP, with molybdenum hexacarbonyl acting as the solid carbon monoxide source.
Q: Does this method tolerate diverse functional groups on the substrates?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating electron-withdrawing groups like cyano and trifluoromethyl, as well as halogens and alkoxy substituents on both aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in accelerating drug development timelines. Our team of expert chemists has extensively evaluated the palladium-catalyzed aminocarbonylation route and possesses the technical expertise to implement this technology effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from preclinical research to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-arylquinolin-2(1H)-one derivative delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced synthetic platform for your next generation of therapeutic candidates. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage you to reach out today to request specific COA data for our existing inventory or to discuss route feasibility assessments for novel derivatives, ensuring that your supply chain is optimized for speed, cost, and reliability.
