Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
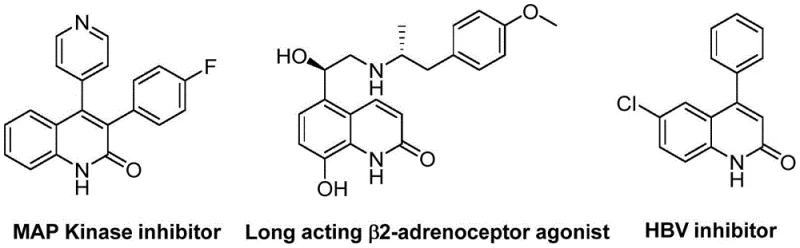
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical backbones for bioactive molecules. Patent CN113045489B discloses a groundbreaking preparation method for 3-arylquinoline-2(1H) ketone derivatives, addressing long-standing challenges in heterocycle synthesis. This innovation leverages a palladium-catalyzed aminocarbonylation strategy that transforms simple benzisoxazoles and benzyl chlorides into valuable quinolinone structures with exceptional efficiency. The significance of this technology lies in its ability to utilize benzisoxazole simultaneously as a nitrogen source and a formyl source, thereby streamlining the synthetic pathway and reducing the reliance on hazardous gaseous carbon monoxide. As illustrated in the structural diversity of bioactive targets below, these derivatives are pivotal in developing antibiotics, antitumor agents, and receptor antagonists.
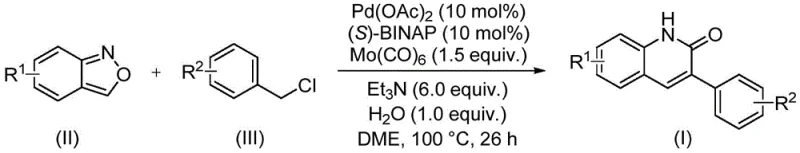
For R&D directors and process chemists, the versatility of this method offers a compelling alternative to traditional cyclization protocols. The reaction operates under relatively mild thermal conditions at 100°C for 26 hours, utilizing a catalytic system composed of palladium acetate, (S)-BINAP, and molybdenum hexacarbonyl. This specific combination ensures high functional group tolerance, allowing for the incorporation of diverse substituents such as halogens, alkoxy groups, and cyano groups without compromising yield. By establishing a reliable pharmaceutical intermediate supplier capability based on this patent, manufacturers can secure a steady supply of high-purity building blocks essential for next-generation drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While effective in specific contexts, these traditional routes often suffer from significant drawbacks that hinder their applicability in modern, green chemistry-focused manufacturing environments. Conventional methods frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, many established protocols necessitate the use of toxic gaseous carbon monoxide for carbonylation steps, posing severe safety risks and requiring specialized high-pressure equipment that increases capital expenditure. The multi-step nature of some older syntheses also results in lower overall atom economy and increased waste generation, creating substantial burdens for environmental compliance and cost reduction in API manufacturing.
The Novel Approach
The novel approach detailed in the patent data represents a paradigm shift by employing a transition metal-catalyzed cascade reaction that constructs the quinolinone core in a single operational step. By utilizing benzisoxazole as a dual-purpose reagent, this method elegantly bypasses the need for external formylating agents or pre-functionalized aniline derivatives. The use of molybdenum hexacarbonyl as a solid CO surrogate allows the reaction to proceed safely in a sealed tube at atmospheric pressure equivalents, drastically simplifying the engineering requirements for scale-up. This strategy not only enhances the safety profile of the operation but also broadens the substrate scope, enabling the synthesis of derivatives with electron-withdrawing and electron-donating groups alike. The result is a streamlined process that delivers high-purity OLED material precursors and pharmaceutical intermediates with minimal downstream purification effort.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this transformative synthesis lies in the intricate catalytic cycle driven by the palladium complex. The reaction initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from palladium acetate and the chiral phosphine ligand (S)-BINAP. This step forms an organopalladium intermediate that is primed for subsequent insertion reactions. Crucially, the molybdenum hexacarbonyl acts as a controlled release source of carbon monoxide, which inserts into the palladium-carbon bond to generate an acyl-palladium species. This mechanistic pathway avoids the direct handling of toxic CO gas while maintaining the necessary concentration of carbonyl units for the reaction to proceed efficiently. The presence of water and triethylamine facilitates the activation of the benzisoxazole ring, promoting its opening to release the nitrogen nucleophile required for the final cyclization step.
Following the carbonyl insertion, the intramolecular nucleophilic attack by the nitrogen atom derived from the benzisoxazole ring closure leads to the formation of the quinolinone skeleton. The chiral ligand environment provided by (S)-BINAP may also impart stereochemical control in specific asymmetric variants, although the primary focus here is on the efficient construction of the heterocyclic core. The final reductive elimination releases the 3-arylquinolin-2(1H)one product and regenerates the palladium catalyst, completing the cycle. This mechanism explains the high functional group tolerance observed, as the mild conditions prevent side reactions such as homocoupling of the benzyl chloride or decomposition of the sensitive benzisoxazole moiety. Understanding these mechanistic details is vital for optimizing reaction parameters and troubleshooting potential scale-up issues in a commercial setting.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction conditions to maximize yield and purity. The protocol dictates a specific molar ratio of reagents, typically employing a slight excess of the benzyl chloride coupling partner relative to the benzisoxazole to drive the equilibrium forward. The choice of solvent, specifically ethylene glycol dimethyl ether (DME), is critical for solubilizing both the organic substrates and the inorganic base, ensuring a homogeneous reaction mixture. Operators must adhere strictly to the temperature profile of 100°C and the reaction time of 26 hours to ensure complete consumption of starting materials, as indicated by the extensive optimization data in the patent. Detailed standardized synthesis steps see the guide below.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 26 hours to ensure complete conversion of starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers distinct strategic advantages that directly impact the bottom line and operational resilience. The shift towards this catalytic process eliminates the dependency on volatile and hazardous gaseous reagents, thereby reducing the regulatory burden and insurance costs associated with handling toxic substances. Moreover, the use of commercially available and inexpensive starting materials like benzisoxazole and substituted benzyl chlorides ensures a stable and cost-effective supply chain, mitigating the risk of raw material shortages that often plague specialty chemical manufacturing. The high yields reported across a wide range of substrates translate directly into improved material throughput and reduced waste disposal costs, aligning perfectly with sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the elimination of expensive protecting group strategies often required in traditional methods. By using benzisoxazole as a dual nitrogen and carbon source, the number of synthetic steps is reduced, which inherently lowers labor costs, solvent consumption, and energy usage per kilogram of product. Additionally, the replacement of gaseous CO with solid Mo(CO)6 removes the need for specialized high-pressure reactors, allowing production to occur in standard glass-lined or stainless steel vessels, representing a significant capital expenditure saving. These factors combine to deliver substantial cost savings without compromising the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the widespread availability of the key reagents, benzisoxazole and benzyl chlorides, which are commodity chemicals produced by numerous global suppliers. This diversification of the supply base reduces the risk of single-source dependency and price volatility. Furthermore, the robustness of the reaction conditions, which tolerate a wide array of functional groups, means that the process is less susceptible to batch failures caused by minor variations in raw material quality. This reliability ensures consistent delivery schedules for downstream customers, fostering stronger long-term partnerships and reducing the need for safety stock inventory.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the reaction and the straightforward workup procedure involving filtration and column chromatography. The absence of hazardous gas evolution simplifies the engineering controls required for venting and scrubbing, making it easier to obtain environmental permits for new production lines. The high atom economy of the transformation minimizes the generation of chemical waste, supporting corporate sustainability initiatives and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles positions the technology favorably for future regulatory landscapes focused on emissions reduction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions about adopting this route for their specific product portfolios.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate formylation reagents and simplifying the synthetic route significantly.
Q: What is the typical yield range for this palladium-catalyzed process?
A: The patented method demonstrates high reaction efficiency, with isolated yields ranging from 68% to 97% across various substrates, ensuring robust production capabilities.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials and standard purification techniques like column chromatography, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the innovative palladium-catalyzed aminocarbonylation technology described in CN113045489B, we can offer our partners a competitive edge through superior quality and consistent supply.
We invite you to collaborate with us to explore how this cutting-edge synthesis can optimize your specific project requirements. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this route can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary targets. Let us be your partner in turning complex chemical challenges into commercial successes.
