Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Introduction to Next-Generation Quinolinone Synthesis
The development of efficient synthetic routes for heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, particularly for privileged structures like quinolin-2(1H)-ones. These derivatives are ubiquitous in pharmaceutical landscapes, serving as critical cores for antibiotics, antiplatelet agents, and antitumor drugs, as evidenced by their presence in MAP Kinase inhibitors and HBV inhibitors. A groundbreaking methodology detailed in patent CN113045489B introduces a highly versatile palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm for accessing these valuable motifs. By leveraging benzisoxazole as a unique dual-purpose reagent acting as both a nitrogen donor and a formyl source, this innovation circumvents traditional limitations associated with toxic gas handling and complex multi-step sequences. This technical advancement not only streamlines the synthetic workflow but also enhances the safety profile of manufacturing processes, making it an ideal candidate for industrial adoption by forward-thinking pharmaceutical enterprises seeking reliable pharmaceutical intermediate suppliers.
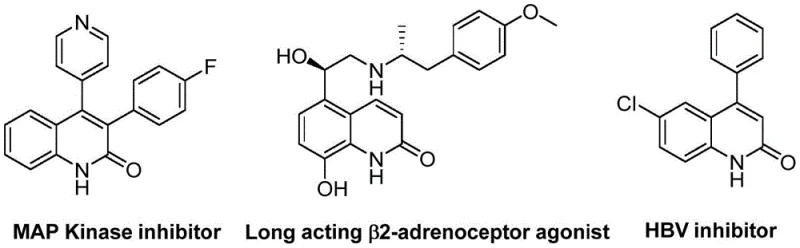
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolinone core has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, or Friedlander syntheses. While these methods are well-established, they often suffer from significant drawbacks when applied to complex molecule synthesis, including harsh reaction conditions, poor atom economy, and limited tolerance for sensitive functional groups. Furthermore, transition-metal catalyzed carbonylation approaches, although powerful, have traditionally required the use of high-pressure carbon monoxide gas, which poses severe safety hazards and necessitates specialized high-pressure reactor infrastructure that increases capital expenditure. The reliance on external CO sources also complicates the supply chain logistics and introduces variability in reaction kinetics due to gas-liquid mass transfer limitations. Consequently, there has been a persistent demand within the R&D community for a safer, more operationally simple alternative that maintains high efficiency without compromising on substrate scope or yield.
The Novel Approach
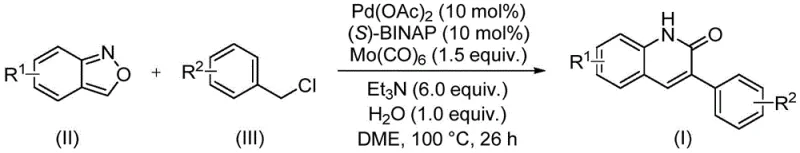
The methodology disclosed in the referenced patent offers a transformative solution by utilizing a solid-state carbonyl source strategy coupled with a clever ring-opening mechanism of benzisoxazoles. Instead of introducing exogenous CO gas, the process employs molybdenum hexacarbonyl [Mo(CO)6] in conjunction with benzisoxazole, which effectively delivers the necessary carbonyl and nitrogen atoms in situ under mild thermal conditions. This approach allows the reaction to proceed in standard sealed vessels at atmospheric pressure equivalents, drastically reducing the engineering controls required for scale-up. The use of readily available benzyl chlorides and substituted benzisoxazoles as starting materials further democratizes access to this chemistry, enabling the rapid generation of diverse libraries of 3-arylquinolin-2(1H)-one derivatives. This shift from gas-phase to solution-phase carbonylation represents a significant leap in process safety and operational simplicity, aligning perfectly with the goals of cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The catalytic cycle underpinning this transformation is a sophisticated interplay between palladium coordination chemistry and the unique reactivity of the benzisoxazole ring. The reaction initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species generated from Pd(OAc)2 and the chiral ligand (S)-BINAP. Subsequently, the benzisoxazole undergoes a ring-opening event, likely facilitated by the basic conditions provided by triethylamine and the presence of water, which releases the nitrogen nucleophile and potentially contributes to the carbonyl pool alongside the Mo(CO)6 decomposition. The resulting organopalladium intermediate then undergoes migratory insertion of the carbonyl species, followed by intramolecular cyclization to forge the characteristic lactam ring of the quinolinone. The inclusion of water as a stoichiometric additive is critical, presumably assisting in the hydrolysis steps required to liberate the final product and regenerate the catalyst. This intricate balance of reagents ensures high turnover numbers and minimizes the formation of side products such as homocoupled bibenzyls or unreacted starting materials.
From an impurity control perspective, the robustness of this catalytic system is remarkable, as it tolerates a wide array of electronic environments on both aromatic rings. Whether the substrates bear electron-withdrawing groups like cyano or trifluoromethyl moieties, or electron-donating groups like methoxy and tert-butyl, the reaction maintains high fidelity. The use of (S)-BINAP, while primarily known for asymmetric catalysis, here likely provides a steric environment that favors the desired reductive elimination pathway over competing beta-hydride elimination processes. This selectivity is crucial for maintaining high purity profiles, as it reduces the burden on downstream purification steps. For process chemists, understanding that the reaction tolerates halogens (Cl, F) without undergoing dehalogenation is particularly valuable, as it preserves handles for further functionalization in late-stage drug discovery campaigns, thereby enhancing the overall utility of the synthetic route.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and mixing efficiency to ensure consistent results. The protocol is designed to be user-friendly, utilizing common organic solvents like ethylene glycol dimethyl ether (DME) and standard heating equipment. The key to success lies in the precise stoichiometric balance of the catalyst system, specifically the 1:1 ratio of Pd(OAc)2 to (S)-BINAP, and the sufficient excess of the base and carbonyl source to drive the equilibrium towards product formation. Detailed standardized operating procedures regarding the addition order of reagents and the specific workup filtration steps are essential for reproducibility. For a comprehensive guide on executing this reaction with optimal parameters, please refer to the step-by-step instructions provided below.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture to ensure homogeneous suspension of all catalytic components and reagents.
- Heat the reaction mixture to 100°C for 26 hours, then filter, adsorb onto silica gel, and purify via column chromatography to isolate the target quinolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substitution of hazardous gaseous carbon monoxide with solid, bench-stable reagents like Mo(CO)6 and benzisoxazole. This change eliminates the need for expensive high-pressure autoclaves and specialized gas handling infrastructure, leading to substantial cost savings in facility setup and maintenance. Furthermore, the starting materials—benzyl chlorides and benzisoxazoles—are commodity chemicals available from multiple global vendors, ensuring a resilient supply chain that is not dependent on single-source specialty reagents. This diversification of the supply base mitigates the risk of production delays caused by raw material shortages, a critical factor for maintaining continuous manufacturing operations in the competitive pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the reaction setup and the elimination of high-pressure safety protocols. By avoiding the use of pressurized CO gas, facilities can operate with standard glass-lined or stainless steel reactors rated for lower pressures, significantly lowering capital investment barriers. Additionally, the high yields reported (often exceeding 90% for unsubstituted or simply substituted substrates) mean less raw material waste and reduced solvent consumption per kilogram of product. The simplified workup, involving basic filtration and silica treatment, reduces the labor hours and consumables associated with complex extraction or crystallization processes, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The reliance on widely available building blocks ensures that production schedules remain robust against market fluctuations. Benzyl chlorides and substituted benzisoxazoles are produced at scale for various industries, meaning lead times are generally short and inventory levels are stable. This reliability allows for better forecasting and just-in-time manufacturing strategies, reducing the need for large safety stocks of finished goods. Moreover, the tolerance for diverse functional groups means that a single platform technology can be used to produce a wide family of intermediates, allowing manufacturers to respond quickly to changing customer demands without retooling or developing entirely new processes for each new derivative.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is straightforward due to the homogeneous nature of the reaction mixture and the absence of gas-liquid mass transfer limitations. The use of DME as a solvent, while requiring appropriate recovery systems, is well-understood in industrial settings. From an environmental standpoint, the atom economy is improved by incorporating the nitrogen and carbon atoms directly from the heterocyclic starting material, reducing the generation of stoichiometric salt waste compared to traditional condensation methods. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the process attractive for environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is vital for R&D teams evaluating this route for their specific pipeline candidates, ensuring that potential challenges are anticipated and mitigated early in the development cycle.
Q: What is the primary advantage of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for hazardous gaseous carbon monoxide and simplifying the reagent profile significantly.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds at 100°C in DME solvent for approximately 26 hours, utilizing a Pd(OAc)2/(S)-BINAP catalyst system with Mo(CO)6 and triethylamine.
Q: Does this method tolerate diverse functional groups on the substrates?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating substituents such as halogens, alkoxy groups, cyano, and trifluoromethyl groups on both the benzisoxazole and benzyl chloride rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 3-arylquinolin-2(1H)-one derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement advanced palladium-catalyzed transformations allows us to offer superior quality intermediates that accelerate your drug development timelines while maintaining cost efficiency.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain needs and drive your innovation forward.
