Advanced Synthesis of Pyrido[2,3-d]pyrimidine Intermediates for Commercial API Production
The global demand for next-generation kinase inhibitors continues to drive innovation in the synthesis of critical pharmaceutical intermediates. Among these, the pyrido[2,3-d]pyrimidine scaffold serves as the structural core for Palbociclib, a potent CDK4/6 inhibitor widely utilized in the treatment of HR-positive, HER2-negative advanced breast cancer. The efficient manufacturing of this core structure, specifically referred to as Compound 1 in recent technical literature, remains a pivotal challenge for process chemists aiming to balance yield, purity, and cost. A significant technological breakthrough in this domain is detailed in Chinese patent CN108117550B, which discloses a robust preparation method that addresses the longstanding inefficiencies of traditional catalytic systems. This patent introduces a streamlined approach utilizing simple palladium chloride (PdCl2) as the primary catalyst, offering a compelling alternative to complex ligand-based systems. By optimizing reaction parameters such as solvent choice and temperature, this method achieves superior yields and purity profiles, positioning it as a preferred route for industrial scale-up.
![Chemical structure of Compound 1, a key pyrido[2,3-d]pyrimidine intermediate for Palbociclib](/insights/img/pyrido-pyrimidine-synthesis-pdcl2-pharma-supplier-20260309075033-01.png)
For R&D directors and process development teams, understanding the mechanistic advantages of this new route is essential for evaluating its integration into existing manufacturing pipelines. The transition from legacy methods to this novel PdCl2-catalyzed protocol represents more than just a reagent swap; it signifies a fundamental improvement in reaction kinetics and thermodynamic control. The ability to operate effectively at moderate temperatures while maintaining high conversion rates suggests a lower activation energy barrier for the cyclization step, which directly correlates to reduced energy consumption and improved safety profiles in a plant setting. Furthermore, the simplified catalyst system reduces the complexity of the downstream purification process, as there are fewer phosphine oxide byproducts to remove, thereby enhancing the overall mass balance of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements described in the subject patent, the synthesis of Compound 1 was predominantly achieved through methods that suffered from significant operational and economic drawbacks. Traditional protocols often relied on palladium acetate (Pd(OAc)2) in conjunction with expensive and sensitive phosphine ligands, such as triphenylphosphine or specialized bulky phosphines. These legacy routes typically required elevated reaction temperatures, often exceeding 90°C, to drive the cyclization to completion. Such harsh thermal conditions not only increased energy costs but also promoted the formation of thermal degradation byproducts and difficult-to-remove impurities, complicating the purification workflow. Additionally, the reliance on air-sensitive ligands necessitated stringent inert atmosphere controls throughout the entire process, increasing the operational burden and the risk of batch failure due to catalyst deactivation. Yields in these conventional methods were frequently capped around 80%, with reaction times extending beyond 6 hours, creating a bottleneck for high-volume production schedules.
The Novel Approach
In stark contrast, the methodology disclosed in CN108117550B leverages a simplified catalytic system centered on PdCl2, which demonstrates remarkable efficiency without the need for auxiliary phosphine ligands. This novel approach operates effectively within a milder temperature window of 60°C to 85°C, with optimal results observed between 65°C and 75°C. The elimination of complex ligands not only reduces the raw material cost but also simplifies the reaction setup, making it more robust against minor variations in atmospheric conditions. The reaction utilizes readily available water-soluble organic solvents like N-methyl-2-pyrrolidone (NMP) or tetrahydrofuran (THF), which facilitate excellent solubility of the reactants and ensure homogeneous reaction conditions. As illustrated in the reaction scheme below, the direct coupling of the halogenated precursor (Compound 2) with the acrylate derivative (Compound 3) proceeds with high atom economy.
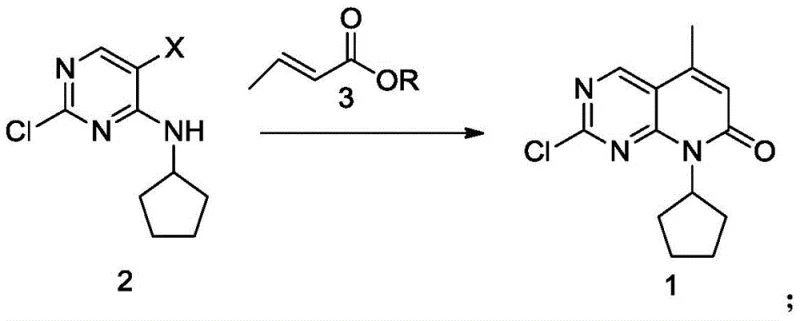
The implementation of this novel route allows for reaction times that are competitive with or superior to prior art, often reaching completion within 6 hours while achieving yields that can exceed 94% under optimized conditions. The use of triethylamine or DIPEA as the base further enhances the practicality of the process, as these reagents are inexpensive and easy to handle on a large scale. By addressing the key pain points of temperature sensitivity and catalyst complexity, this method provides a clear pathway for cost reduction in API manufacturing, enabling producers to meet the rigorous quality standards required for oncology therapeutics while maintaining a competitive cost structure.
Mechanistic Insights into PdCl2-Catalyzed Cyclization
The success of this synthetic route lies in the unique reactivity of the PdCl2 catalyst in facilitating the oxidative addition and subsequent migratory insertion steps required for the formation of the pyrido[2,3-d]pyrimidine ring system. Mechanistically, the Pd(II) species is likely reduced in situ to the active Pd(0) species by the amine base or the substrate itself, initiating the catalytic cycle. The oxidative addition of the palladium center into the carbon-halogen bond (where X is Br or I) of Compound 2 is the rate-determining step in many cross-coupling reactions. In this specific system, the electronic environment of the pyrimidine ring appears to be sufficiently activated to allow PdCl2 to perform this step efficiently without the electron-donating assistance of phosphine ligands. Following oxidative addition, the coordination and migratory insertion of the alkene moiety from Compound 3 into the palladium-carbon bond occurs, setting the stage for the final cyclization.
Crucially, the choice of solvent and base plays a pivotal role in stabilizing the catalytic intermediates and promoting the final beta-hydride elimination and re-aromatization steps. The use of polar aprotic solvents like NMP helps to solvate the ionic intermediates and the base, ensuring that the deprotonation steps necessary for cyclization proceed rapidly. This mechanistic efficiency translates directly into a cleaner impurity profile, as the rapid turnover of the catalyst minimizes the residence time of reactive intermediates that could otherwise undergo side reactions such as homocoupling or hydrodehalogenation. For quality control teams, this means that the crude product obtained after the reaction contains significantly lower levels of genotoxic impurities and metal residues, simplifying the subsequent crystallization and washing steps required to meet stringent pharmaceutical specifications.
How to Synthesize Pyrido[2,3-d]pyrimidine Compound 1 Efficiently
To implement this high-yielding synthesis in a laboratory or pilot plant setting, strict adherence to the optimized molar ratios and temperature profiles is essential. The process begins with the dissolution of the starting materials in the selected solvent, followed by the careful addition of the catalyst under an inert atmosphere to prevent premature oxidation. The reaction is then heated to the target temperature, where it is monitored via TLC or HPLC to ensure complete conversion of the starting material before proceeding to the workup phase. The post-reaction treatment involves acetylation and controlled crystallization, which are critical for locking in the high purity achieved during the reaction step.
- Dissolve the halogenated precursor (Compound 2) and the acrylate coupling partner (Compound 3) in a water-soluble organic solvent such as NMP or THF.
- Add an organic base like triethylamine and the PdCl2 catalyst under an inert nitrogen atmosphere, ensuring the catalyst loading is optimized between 0.03 to 0.05 equivalents.
- Heat the reaction mixture to 65°C-75°C for approximately 6 hours, followed by acetic anhydride treatment and crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this PdCl2-catalyzed methodology offers substantial strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized phosphine ligands, which are often sourced from limited suppliers and subject to price volatility, manufacturers can secure a more stable and cost-effective supply of catalysts. PdCl2 is a commodity chemical with a well-established global supply network, ensuring continuity of supply even during market fluctuations. Furthermore, the reduced complexity of the reaction mixture lowers the burden on waste management systems, as there are fewer organic byproducts to treat, aligning with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The elimination of expensive ligands and the ability to use lower catalyst loadings (0.03 to 0.05 equivalents) directly decreases the bill of materials for each batch. Additionally, the milder reaction temperatures reduce energy consumption for heating and cooling cycles, contributing to lower utility costs per kilogram of product. The high crude purity achieved reduces the volume of solvents required for recrystallization and chromatography, further driving down processing costs and increasing overall throughput capacity.
- Enhanced Supply Chain Reliability: The robustness of the PdCl2 system against minor variations in moisture or oxygen levels makes the process more forgiving in large-scale reactors, reducing the risk of batch failures and ensuring consistent delivery schedules. The use of common solvents like NMP and THF, which are widely available in bulk quantities, mitigates the risk of solvent shortages that can plague specialized chemical syntheses. This reliability is crucial for maintaining the continuous flow of intermediates required for just-in-time API manufacturing.
- Scalability and Environmental Compliance: The simplified workup procedure, which relies on standard filtration and washing techniques rather than complex column chromatography, is inherently easier to scale from kilograms to tons. The reduction in hazardous waste generation, particularly phosphine oxides, simplifies effluent treatment and lowers disposal costs. This environmental efficiency not only reduces the carbon footprint of the manufacturing process but also ensures compliance with green chemistry initiatives demanded by major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making.
Q: What are the advantages of using PdCl2 over Pd(OAc)2 with phosphine ligands?
A: Using simple PdCl2 eliminates the need for expensive and air-sensitive phosphine ligands, significantly reducing raw material costs and simplifying the handling requirements for large-scale manufacturing.
Q: How does this method improve impurity profiles compared to prior art?
A: The optimized temperature range of 60°C to 85°C minimizes thermal decomposition and side reactions often seen at higher temperatures (90°C+), resulting in crude purities exceeding 98% before final recrystallization.
Q: Is this process suitable for multi-ton scale production?
A: Yes, the use of common solvents like NMP and THF, combined with a robust catalyst system that tolerates standard industrial equipment constraints, makes this route highly scalable for commercial API supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrido[2,3-d]pyrimidine Intermediate Supplier
As the pharmaceutical industry continues to evolve, the need for reliable partners who can translate innovative patent technologies into commercial reality has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging deep expertise in palladium-catalyzed transformations to deliver high-quality intermediates for oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory are faithfully reproduced in our state-of-the-art manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Compound 1 meets the exacting standards required for GMP API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this efficient PdCl2-catalyzed process. We encourage you to request specific COA data and route feasibility assessments to verify the superiority of our manufacturing capabilities and secure a sustainable supply of this critical pharmaceutical intermediate.
