Revolutionizing Palbociclib Intermediate Production with Green Silver Catalysis Technology
Revolutionizing Palbociclib Intermediate Production with Green Silver Catalysis Technology
The pharmaceutical industry is constantly seeking more efficient, cost-effective, and environmentally sustainable pathways for the synthesis of complex oncology therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN114539251A, which discloses a novel preparation method for pyrido[2,3-d]pyrimidine derivatives. These compounds serve as critical intermediates in the manufacture of Palbociclib, a potent oral cyclin-dependent kinase (CDK) 4/6 inhibitor used for treating advanced breast cancer. The patent introduces a paradigm shift by utilizing inexpensive silver salts as catalysts to achieve a direct, one-step synthesis of these key scaffolds. This innovation addresses long-standing challenges in the field, such as the reliance on precious metal catalysts, multi-step sequences, and harsh reaction conditions, thereby offering a robust solution for reliable pharmaceutical intermediate supplier networks aiming to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Palbociclib and its precursors has been plagued by convoluted synthetic routes that impose significant burdens on both cost and operational safety. As illustrated in prior art such as WO 2016030439A1, traditional methods typically commence with 5-bromo-2,4-dichloropyrimidine, which undergoes condensation with cyclopentylamine. This is followed by a series of energetically demanding and chemically hazardous steps, including a palladium-catalyzed Heck coupling with crotonic acid in tetrahydrofuran, and subsequent carbonyl alpha-bromination using N-bromosuccinimide (NBS). Furthermore, the convergence of fragments often requires the use of highly reactive and moisture-sensitive organometallic reagents, such as organic magnesium or lithium species, to couple distinct intermediates. These processes not only suffer from low overall yields due to the accumulation of losses at each step but also generate substantial amounts of heavy metal waste and halogenated byproducts, complicating purification and environmental compliance.
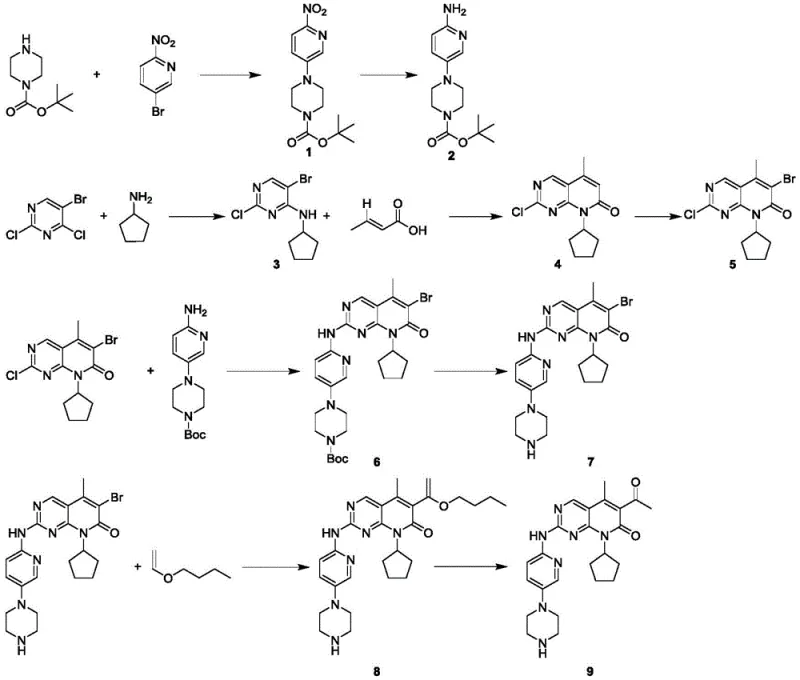
The Novel Approach
In stark contrast to the cumbersome legacy pathways, the technology disclosed in CN114539251A streamlines the production of the target pyrido[2,3-d]pyrimidine core through a direct decarboxylative coupling strategy. This novel approach utilizes a readily available pyrido[2,3-d]pyrimidine substrate and reacts it directly with alpha-keto acids, such as pyruvic acid or benzoylcarboxylic acid, in the presence of a silver catalyst and an oxidant. By bypassing the need for pre-functionalization via bromination and subsequent cross-coupling, this method effectively collapses multiple synthetic operations into a single transformative step. The result is a drastic simplification of the workflow, where the acetyl or benzoyl group is installed directly onto the heterocyclic core under mild conditions. This not only accelerates the timeline for cost reduction in pharmaceutical intermediates manufacturing but also inherently improves the impurity profile by eliminating the side reactions associated with organometallic handling and halogenation.
Mechanistic Insights into Silver-Catalyzed Decarboxylative Coupling
The core of this technological advancement lies in the unique reactivity of silver species in facilitating carbon-carbon bond formation via radical or organometallic pathways. The reaction mechanism likely involves the activation of the alpha-keto acid by the silver catalyst, potentially forming a transient silver-carboxylate species that undergoes oxidative decarboxylation. This process generates a reactive acyl radical or a high-valent silver-acyl intermediate, which subsequently attacks the electron-deficient pyrido[2,3-d]pyrimidine ring system. The use of potassium persulfate (K2S2O8) as a terminal oxidant is crucial, as it serves to regenerate the active silver species and drive the thermodynamic equilibrium towards the desired acylated product. This catalytic cycle operates efficiently at moderate temperatures, typically around 70°C, avoiding the extreme thermal stress that often leads to decomposition in sensitive heterocyclic systems.
![General reaction scheme showing silver-catalyzed decarboxylative coupling of pyrido[2,3-d]pyrimidine with alpha-keto acids](/insights/img/palbociclib-intermediate-silver-catalysis-pharma-supplier-20260307090626-07.png)
From a quality control perspective, this mechanism offers superior impurity management compared to traditional nucleophilic substitutions. Because the reaction proceeds through a selective radical or electrophilic pathway directed by the catalyst, there is minimal formation of regioisomers or over-alkylated byproducts that are common in non-catalyzed thermal reactions. The high selectivity ensures that the resulting crude material possesses a purity profile that is amenable to simple recrystallization techniques, such as using petroleum ether, rather than requiring complex chromatographic separations. This inherent cleanliness of the reaction is vital for commercial scale-up of complex pharmaceutical intermediates, as it reduces the burden on downstream processing units and ensures consistent batch-to-batch quality essential for regulatory approval.
How to Synthesize 6-Acetyl-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be compatible with standard chemical manufacturing infrastructure. The process involves charging a reaction vessel with the chloro-substituted pyrido[2,3-d]pyrimidine precursor, pyruvic acid, a silver salt catalyst like silver carbonate, and potassium persulfate in a mixed solvent system of acetonitrile and water. The mixture is then heated in an oil bath, allowing the decarboxylative acylation to proceed to completion. Detailed standardized operating procedures regarding stoichiometry, addition rates, and specific workup parameters are critical for maximizing yield and ensuring safety. For the complete technical protocol and precise reaction conditions validated by our R&D team, please refer to the synthesis guide below.
- Charge a reaction vessel with 2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one, pyruvic acid, silver carbonate catalyst, and potassium persulfate oxidant in an acetonitrile-water solvent system.
- Heat the mixture in an oil bath at 70°C for approximately 6 hours under air atmosphere to facilitate the decarboxylative acylation.
- Upon completion, filter the mixture through kieselguhr, extract the organic phase with ethyl acetate, concentrate, and recrystallize from petroleum ether to obtain the high-purity white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silver-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage is the substantial cost savings derived from the replacement of expensive palladium catalysts and complex organometallic reagents with abundant and inexpensive silver salts and alpha-keto acids. Furthermore, the reduction in synthetic steps from a multi-stage sequence to a one-pot reaction significantly lowers the consumption of solvents, energy, and labor hours per kilogram of product. This streamlined process minimizes the potential for supply chain disruptions caused by the sourcing of specialized reagents, thereby enhancing the overall reliability of the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this silver-catalyzed route is profound, primarily driven by the elimination of noble metal palladium, which is subject to volatile market pricing and high procurement costs. Additionally, the avoidance of hazardous reagents like NBS and organolithiums reduces the expenses associated with specialized storage, handling protocols, and hazardous waste disposal. The ability to recover and recycle the silver catalyst further amplifies these savings, as the patent describes a regeneration loop where silver salts can be converted back to active silver carbonate, creating a closed-loop system that minimizes raw material attrition and maximizes resource efficiency.
- Enhanced Supply Chain Reliability: Simplifying the synthesis from a linear multi-step process to a convergent one-step reaction drastically reduces the lead time required for production. In a traditional multi-step route, a failure or delay in any single intermediate step can halt the entire production line; however, this consolidated approach mitigates such risks by reducing the number of critical control points. The use of commercially available and stable starting materials ensures that production schedules are not held hostage by the availability of custom-synthesized building blocks, providing a more resilient and predictable supply continuity for downstream API manufacturers.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, utilizing aqueous acetonitrile mixtures and moderate temperatures, are inherently safer and easier to scale than exothermic organometallic reactions. The absence of heavy metal palladium residues in the final product simplifies the purification process and ensures compliance with stringent ICH guidelines for elemental impurities in drug substances. Moreover, the greener nature of the process, characterized by atom economy and reduced solvent usage, aligns with modern environmental, social, and governance (ESG) goals, making it an attractive option for companies aiming to reduce their carbon footprint and regulatory liability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about technology transfer and process optimization.
Q: How does the silver-catalyzed method improve upon traditional palladium routes?
A: The silver-catalyzed method eliminates the need for expensive palladium catalysts and hazardous organometallic reagents like Grignard or lithium compounds. It simplifies the process from a multi-step sequence involving Heck coupling and bromination to a direct one-step decarboxylative coupling, significantly reducing operational complexity and waste.
Q: Is the silver catalyst recoverable in this process?
A: Yes, the patent explicitly details a recovery mechanism where excess silver salts can be filtered, treated with nitric acid and sodium carbonate, and regenerated into silver carbonate for reuse without significant loss of catalytic activity, enhancing the economic viability of the process.
Q: What represents the key structural advantage of the produced intermediates?
A: The process yields 6-acetyl or 6-benzoyl substituted pyrido[2,3-d]pyrimidine derivatives with high purity (>99%). These structures serve as direct precursors for Palbociclib, bypassing the need for complex functional group transformations required in older synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palbociclib Intermediate Supplier
The technological potential of silver-catalyzed decarboxylative coupling represents a significant leap forward in the manufacturing of oncology intermediates, yet translating patent chemistry into commercial reality requires deep expertise in process engineering and scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of silver catalysis, including catalyst recovery systems and rigorous QC labs that ensure stringent purity specifications are met for every batch. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated technical team is committed to delivering intermediates that seamlessly integrate into your downstream API synthesis.
We invite you to explore how this innovative route can optimize your production costs and enhance your supply chain resilience. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive landscape of cancer therapeutics.
