Advanced Manufacturing of Loxoprofen Sodium: A Scalable Route for Global API Production
Introduction to Patent CN1294115A and Loxoprofen Sodium
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), with Loxoprofen Sodium standing out as a critical therapeutic agent for pain management and inflammation control. Patent CN1294115A discloses a sophisticated and highly optimized synthetic methodology that addresses the historical challenges of complexity and high production costs associated with earlier literature methods. This proprietary process leverages readily available industrial feedstocks, such as 2-chloropropionyl chloride and diethyl adipate, to construct the complex molecular architecture of Loxoprofen Sodium through a series of high-yielding transformations. By integrating classical organic reactions like Friedel-Crafts acylation with specialized catalytic rearrangements, the technology offers a streamlined approach that is particularly attractive for commercial scale-up.
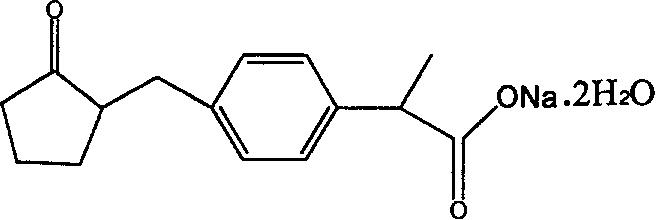
The strategic design of this synthesis not only ensures a reliable supply of high-purity active pharmaceutical ingredients but also incorporates rigorous purification protocols at intermediate stages to minimize impurity carryover. For R&D directors and process chemists, understanding the nuances of this route provides valuable insights into managing stereochemistry and regioselectivity during the construction of the chiral center adjacent to the aromatic ring. Furthermore, the emphasis on solvent recovery and the avoidance of exotic reagents positions this method as a cornerstone for sustainable manufacturing practices in the competitive generic drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Loxoprofen Sodium has been plagued by methodologies that rely on expensive starting materials or involve hazardous reaction conditions that complicate safety protocols and waste management. Traditional routes often necessitate the use of precious metal catalysts or require extremely low temperatures that are energy-intensive to maintain on a multi-ton scale, leading to inflated operational expenditures. Additionally, many legacy processes suffer from poor atom economy, generating significant quantities of saline waste during neutralization steps or requiring chromatographic purification which is impractical for bulk manufacturing. These inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher volatility in pricing for the final API, which directly impacts the profitability of downstream pharmaceutical formulations.
The Novel Approach
In stark contrast, the methodology outlined in CN1294115A introduces a pragmatic and cost-effective strategy that begins with the Friedel-Crafts acylation of toluene, a commodity chemical, to establish the carbon skeleton efficiently. A standout feature of this novel approach is the utilization of a zinc oxide and cuprous oxide catalytic system to facilitate a 1,2-aryl migration, a transformation that proceeds under milder conditions than traditional acid-catalyzed rearrangements. This innovation eliminates the need for corrosive strong acids in the migration step, thereby reducing equipment corrosion and extending the lifespan of reactor vessels. The subsequent coupling with a cyclopentanone derivative derived from diethyl adipate via Dieckmann condensation ensures that the core ring structure is built from inexpensive dicarboxylic esters.
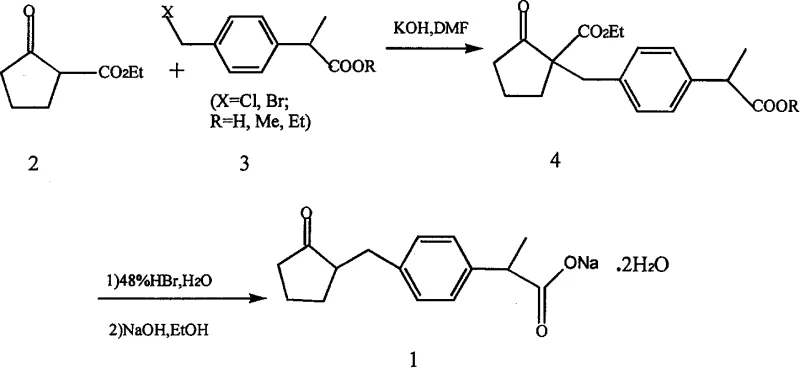
Furthermore, the process is designed with scalability in mind, utilizing standard unit operations such as reflux, distillation, and crystallization that are easily transferable from pilot plant to full commercial production. The integration of a ketal protection strategy prior to the rearrangement step demonstrates a high level of chemical foresight, preventing unwanted side reactions at the carbonyl group and ensuring high regioselectivity. This comprehensive optimization results in a process that is not only chemically elegant but also commercially superior, offering a distinct advantage for manufacturers seeking to reduce their cost of goods sold while maintaining exceptional product quality.
Mechanistic Insights into ZnO/Cu2O Catalyzed Rearrangement and Cyclization
The heart of this synthetic innovation lies in the mechanistic intricacies of the catalytic rearrangement step, where a ketal-protected intermediate undergoes a 1,2-aryl migration to form the crucial propionic acid backbone. The use of zinc oxide and cuprous oxide as a dual-catalyst system suggests a Lewis acid-mediated pathway that activates the ketal functionality, facilitating the migration of the aryl group with high fidelity. This specific catalytic environment is critical for suppressing competing elimination reactions that could lead to olefinic impurities, which are notoriously difficult to remove in later stages. By carefully controlling the stoichiometry and reaction temperature, the process ensures that the migration occurs exclusively to the desired position, preserving the integrity of the chiral center that will eventually define the biological activity of the drug.
Following the construction of the aromatic side chain, the synthesis employs a Dieckmann condensation to generate the 2-ethoxycarbonylcyclopentanone moiety, a key building block that provides the five-membered ring characteristic of Loxoprofen. This intramolecular Claisen condensation is driven by the formation of a stable enolate species in the presence of sodium metal, followed by a precise alkylation with the brominated aromatic intermediate. The mechanistic precision required here is paramount, as over-alkylation or self-condensation of the cyclopentanone derivative must be strictly avoided to maintain high yields. The subsequent hydrolysis and decarboxylation steps are managed through controlled acidic conditions that cleave the ester and remove the carboxyl group without affecting the sensitive ketone functionality.
Impurity control is woven into the very fabric of this mechanism, with each step designed to produce intermediates that can be purified via crystallization rather than complex chromatography. For instance, the bromination step utilizes benzoyl peroxide as a radical initiator under UV irradiation, a condition that selectively targets the benzylic position while leaving the aromatic ring and the carboxylic acid group intact. This selectivity minimizes the formation of poly-brominated byproducts, simplifying the downstream workup. The final salt formation with sodium hydroxethoxide in ethanol further acts as a purification checkpoint, where the solubility differences between the sodium salt and organic impurities are exploited to deliver a product of pharmaceutical grade purity suitable for direct tableting.
How to Synthesize Loxoprofen Sodium Efficiently
The synthesis of Loxoprofen Sodium described in this patent represents a benchmark for efficiency in NSAID manufacturing, combining robust chemistry with practical engineering solutions. To execute this process successfully, one must adhere to strict anhydrous conditions during the initial acylation and Dieckmann condensation steps to prevent hydrolysis of sensitive intermediates. The detailed standardized synthetic steps provided below outline the precise molar ratios, temperature profiles, and workup procedures necessary to achieve the high yields reported in the patent data. Operators should pay particular attention to the catalyst loading in the rearrangement step and the timing of the bromine addition to ensure optimal conversion rates.
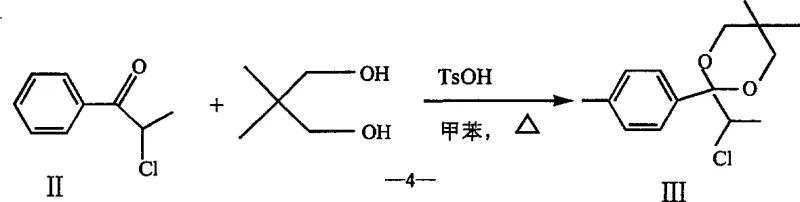
- Perform Friedel-Crafts acylation of toluene with 2-chloropropionyl chloride using AlCl3 catalyst to form the chloro-ketone intermediate.
- Protect the ketone as a ketal using neopentyl glycol, followed by a unique ZnO/Cu2O catalyzed 1,2-aryl migration and hydrolysis to yield the propionic acid derivative.
- Execute radical bromination to activate the benzylic position, then couple with 2-ethoxycarbonylcyclopentanone via alkylation, followed by hydrolysis and decarboxylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible strategic benefits that extend far beyond simple chemical yield. The reliance on commodity chemicals like toluene, 2-chloropropionyl chloride, and diethyl adipate insulates the manufacturing process from the volatility associated with specialized or scarce reagents. This abundance of raw materials ensures a stable supply chain that is less susceptible to geopolitical disruptions or sudden price spikes, providing a predictable cost structure for long-term planning. Moreover, the process design inherently supports cost reduction in NSAID manufacturing by eliminating the need for expensive transition metal catalysts such as palladium or rhodium, which often require complex and costly removal steps to meet residual metal specifications.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the extensive implementation of solvent recovery loops, particularly for toluene and ethanol, which constitute the bulk of the liquid waste stream in traditional batch processes. By distilling and reusing these solvents multiple times, the facility drastically reduces both raw material procurement costs and waste disposal fees, leading to substantial overall savings. Additionally, the avoidance of chromatographic purification in favor of crystallization-based purification lowers the consumption of silica gel and eluents, further driving down the variable costs per kilogram of produced API. The high atom economy of the Dieckmann condensation and the efficient coupling steps mean that less raw material is wasted as byproduct, maximizing the output from every ton of input.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without catastrophic failure, makes this process highly reliable for continuous or large-batch production. This operational stability reduces the risk of batch failures and reprocessing, ensuring that delivery schedules to downstream formulation partners are met consistently. The use of standard glass-lined or stainless steel reactors, rather than specialized Hastelloy equipment required for highly corrosive alternatives, means that the process can be manufactured in a wider range of existing facilities, increasing the available capacity in the market. This flexibility allows for rapid scaling of production volumes to meet surges in demand without the need for significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with modern green chemistry initiatives by minimizing the generation of hazardous waste and reducing the overall E-factor of the synthesis. The catalytic rearrangement step operates under relatively mild conditions, lowering the energy consumption required for heating and cooling compared to high-temperature pyrolysis methods. The final product isolation involves simple filtration and drying, avoiding the use of large volumes of halogenated solvents that pose regulatory challenges in many jurisdictions. This environmental friendliness not only simplifies permitting and compliance but also enhances the brand reputation of the manufacturer as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Loxoprofen Sodium using the patented methodology. These answers are derived directly from the experimental data and process descriptions found in the source documentation, providing clarity on the feasibility and advantages of this specific route. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or contract manufacturing agreements.
Q: What are the key advantages of the ZnO/Cu2O catalyzed rearrangement step?
A: This specific catalytic step allows for a mild 1,2-aryl migration under relatively neutral conditions compared to traditional strong acid rearrangements, significantly reducing side reactions and improving the overall yield of the propionic acid backbone.
Q: How does this process ensure high purity for pharmaceutical grade Loxoprofen Sodium?
A: The process utilizes multiple crystallization and recrystallization steps, particularly in the final salt formation stage using ethanol and ethyl acetate, which effectively removes organic impurities and residual solvents to meet stringent pharmacopeial standards.
Q: Is the solvent system used in this synthesis environmentally sustainable?
A: Yes, the protocol emphasizes the recovery and reuse of major solvents like toluene and ethanol through distillation, minimizing waste generation and aligning with green chemistry principles for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Loxoprofen Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of any pharmaceutical product hinges on the reliability and quality of its supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Loxoprofen Sodium meets the highest international pharmacopeial standards. We are committed to delivering not just a chemical product, but a comprehensive solution that secures your market position.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this optimized process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your NSAID portfolio.
