Advanced Ferrocene Production: Overcoming Yield Barriers with Crown Ether Catalysis for Commercial Scale-up
The landscape of organometallic synthesis is undergoing a significant transformation driven by the need for safer, more efficient, and higher-yielding production methods for critical intermediates like ferrocene. Patent CN101341164B introduces a groundbreaking approach to the preparation of metallocenes, specifically focusing on dicyclopentadienyl iron, commonly known as ferrocene. This technology leverages the unique properties of crown ether complexes to facilitate the reaction between cyclopentadiene and ferrous chloride under mild conditions. Unlike traditional methodologies that rely on hazardous alkali metals or toxic solvents, this invention utilizes a catalytic system involving a Group 1A metal and a crown ether, such as dibenzo-18-crown-6, in the presence of diethylamine. The result is a robust process that not only enhances reaction kinetics but also simplifies the downstream purification workflow, making it an attractive option for industrial scale-up.
Ferrocene itself is a compound of immense strategic value, serving as a gasoline anti-knock additive, a combustion catalyst for rocket propellants, and a precursor in the manufacturing of integrated circuits and pharmaceuticals. The ability to produce this material with high purity and consistency is paramount for suppliers serving the fine chemical intermediates market. By adopting the techniques outlined in this patent, manufacturers can achieve a reliable supply of high-purity organometallics while adhering to increasingly stringent environmental and safety regulations. This report analyzes the technical merits of this innovation and its implications for procurement and supply chain strategies in the global chemical sector.
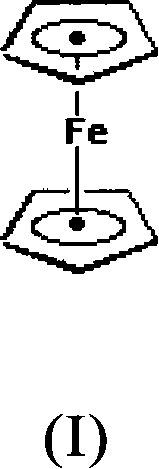
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ferrocene has been plagued by inefficiencies and safety concerns inherent to early organometallic protocols. One prominent prior art method, described in U.S. Patent No. 3,217,022, involves the dissolution of ferric chloride in methanol followed by the addition of iron powder, sodium methoxide, and cyclopentadiene. This multi-step procedure is not only cumbersome but also suffers from significant drawbacks, including the use of large volumes of methanol which complicates the purification process. Consequently, reported yields for this method are often suboptimal, typically hovering between 65% and 70%, which represents a substantial loss of valuable raw materials in a commercial setting. Furthermore, alternative methods found in literature, such as those utilizing tetrahydrofuran (THF) and sodium metal, require rigorous preparatory treatments like peroxide removal and extensive drying to prevent side reactions. These requirements add layers of complexity and cost, rendering the processes economically unviable for large-scale commercial scale-up of complex organometallics.
The Novel Approach
In stark contrast, the method disclosed in CN101341164B offers a streamlined and economically feasible alternative that addresses the core deficiencies of the prior art. By employing a catalytic system based on the interaction between a crown ether (specifically dibenzo-18-crown-6) and a Group 1A metal like potassium, the process generates a highly reactive species capable of deprotonating cyclopentadiene efficiently. This novel approach eliminates the need for dangerous reagents such as metallic sodium or volatile ethers like THF, replacing them with safer and more manageable components like diethylamine and petroleum ether for extraction. The reaction proceeds vigorously at room temperature, significantly reducing the energy input required compared to heated reflux conditions often needed in older methods. Most importantly, this technique consistently delivers yields in the range of 77% to 86%, representing a marked improvement in atom economy and process efficiency that directly translates to cost reduction in fine chemical manufacturing.
Mechanistic Insights into Crown Ether-Catalyzed Metallocene Synthesis
The efficacy of this synthesis route lies in the sophisticated mechanistic role played by the crown ether catalyst. The process initiates with the formation of a stable complex between the potassium cation and the dibenzo-18-crown-6 ether, which effectively solubilizes the metal ion in the organic phase. This interaction creates a "naked" anion environment that enhances the nucleophilicity of the subsequent species. As illustrated in the reaction scheme below, the potassium-crown ether complex interacts with diethylamine to form a potassium anion-diethylamine-crown ether-cation complex. This intermediate is crucial as it acts as a potent base, capable of abstracting a proton from cyclopentadiene to generate the cyclopentadienyl anion, the key building block for the metallocene structure.
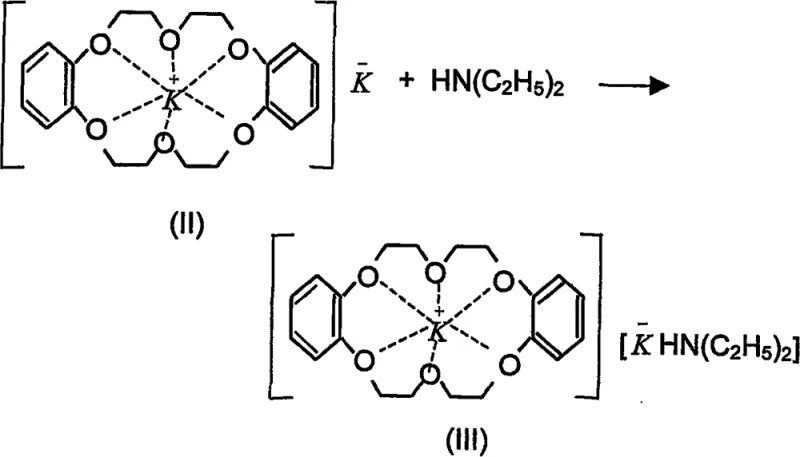
Following the generation of the active catalytic species, the cyclopentadienyl anion is formed in situ within the reaction mixture. This anionic species is then poised to react with the ferrous chloride source. The presence of the crown ether ensures that the ionic species remain sufficiently soluble and reactive throughout the process, preventing premature precipitation or decomposition that often plagues heterogeneous reactions. The subsequent step involves the coordination of two cyclopentadienyl anions to the iron center, displacing the chloride ligands and forming the characteristic sandwich structure of ferrocene. The entire catalytic cycle is designed to minimize side reactions and maximize the conversion of the iron salt into the desired organometallic product, ensuring a clean impurity profile that is essential for downstream applications in sensitive fields like electronics and pharmaceuticals.
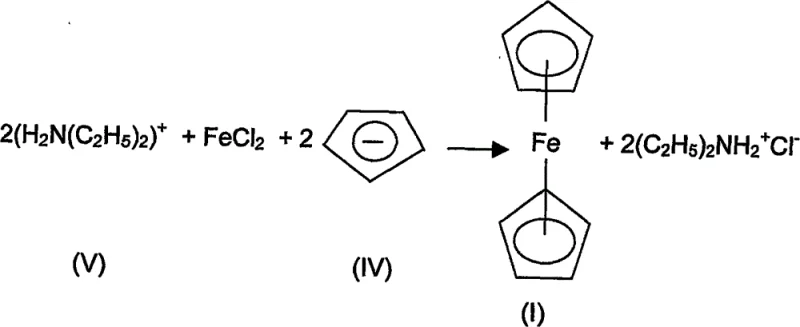
How to Synthesize Ferrocene Efficiently
Implementing this advanced synthesis protocol requires careful attention to reaction conditions, particularly regarding moisture control and reagent stoichiometry. The patent outlines a specific procedure where dry ferrous chloride is added to a cooled mixture of cyclopentadiene, diethylamine, potassium, and the crown ether catalyst under a dry nitrogen atmosphere. This inert environment is critical to prevent the oxidation of the ferrous species and the hydrolysis of the reactive intermediates. The mixture is then stirred vigorously at room temperature for a period of approximately 4 to 6 hours, allowing the catalytic cycle to reach completion. Detailed standard operating procedures regarding exact molar ratios, temperature gradients, and workup techniques are essential for reproducibility and safety in a plant environment.
- Prepare a mixture of cyclopentadiene, diethylamine, a Group 1A metal (such as potassium), and a crown ether (like dibenzo-18-crown-6) in a cooled flask under dry nitrogen.
- Add dry ferrous chloride to the solution and stir vigorously at room temperature for several hours (typically 4-6 hours) to allow the catalytic reaction to proceed.
- Evaporate excess diethylamine under vacuum, extract the residue with petroleum ether, filter hot, and remove the solvent to isolate the orange-yellow ferrocene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this crown ether-catalyzed process presents a compelling value proposition centered on risk mitigation and operational efficiency. The shift away from hazardous reagents like sodium metal and pyrophoric substances significantly lowers the safety risks associated with storage and handling, thereby reducing insurance premiums and compliance costs. Moreover, the ability to recover and recycle a substantial portion of the diethylamine solvent (approximately 40-47% as noted in the examples) contributes to a more sustainable and cost-effective operation. This closed-loop potential minimizes waste disposal fees and aligns with modern green chemistry initiatives, enhancing the corporate sustainability profile of the manufacturer.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-handle reagents such as tetrahydrofuran and metallic sodium drastically simplifies the raw material procurement strategy. By achieving higher yields (up to 86% compared to 65% in legacy methods), the process inherently reduces the cost per kilogram of the final product through better atom economy. The reduction in reaction time by approximately 50% also implies higher throughput capacity for existing reactor vessels, allowing facilities to produce more volume without capital expenditure on new equipment. These factors combined create a robust economic model that supports competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials like ferrous chloride and cyclopentadiene ensures a resilient supply chain less susceptible to the volatility often seen with specialized organometallic reagents. The simplified workup procedure, which involves straightforward extraction with petroleum ether and hot filtration, reduces the dependency on complex purification infrastructure. This simplicity translates to fewer bottlenecks in production scheduling and a more predictable lead time for fulfilling customer orders, which is critical for maintaining long-term contracts with major pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at room temperature and atmospheric pressure, removing the engineering challenges associated with high-pressure or cryogenic reactions. The ability to recover the crown ether catalyst via vacuum distillation after hot water treatment further demonstrates the process's suitability for continuous or semi-continuous manufacturing modes. From an environmental perspective, the reduction in solvent usage and the avoidance of heavy metal contaminants in the waste stream facilitate easier regulatory compliance, ensuring uninterrupted operations in jurisdictions with strict environmental protection laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrocene synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the primary advantages of using crown ether catalysis for ferrocene synthesis compared to traditional methods?
A: The crown ether catalyzed process significantly reduces reaction time by approximately 50% compared to conventional methods and eliminates the need for hazardous reagents like sodium metal or tetrahydrofuran (THF). Furthermore, it achieves superior yields ranging from 77% to 86%, whereas older methods often struggled to exceed 65-70%.
Q: Can this synthesis method be adapted for other metallocenes besides ferrocene?
A: Yes, the patent explicitly states that this methodology is versatile and can be applied to synthesize cyclopentadiene compounds of various metals including titanium, zirconium, hafnium, vanadium, chromium, molybdenum, tungsten, manganese, ruthenium, osmium, cobalt, rhodium, and nickel by simply substituting the metal salt.
Q: How is the catalyst recovered or managed in this process to ensure cost efficiency?
A: The process allows for the recovery of valuable components; specifically, approximately 40-47% of the initial diethylamine can be recovered under vacuum. Additionally, the crown ether can be separated from the residue via hot water treatment and vacuum distillation, facilitating reuse and minimizing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality organometallic compounds play in the advancement of modern industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether your application requires ferrocene for polymer stabilization, fuel additives, or as a precursor for advanced materials, our capability to implement the improved crown ether catalysis method ensures a consistent and reliable supply.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your operations.
