Advanced Manufacturing Strategy for Gefitinib Intermediates via Direct Cyclization
Introduction to Advanced Gefitinib Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of critical oncology therapeutics, specifically targeting the epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors. Patent CN1300118C discloses a highly optimized preparation method for 4-(3-chloro-4-fluorophenylamino)-7-methoxy-6-(3-morpholinopropoxy)quinazoline, widely known as Gefitinib. This specific intellectual property represents a significant departure from legacy synthetic pathways by addressing the inherent inefficiencies associated with traditional demethylation and protection strategies. The disclosed technology leverages 3,4-dimethoxybenzoic acid as a cost-effective starting material to construct the quinazoline scaffold through a streamlined sequence that minimizes waste and maximizes atom economy. For procurement leaders and R&D directors evaluating supply chain resilience, understanding the mechanistic advantages of this route is paramount for securing a reliable API intermediate supplier capable of meeting stringent global quality standards.
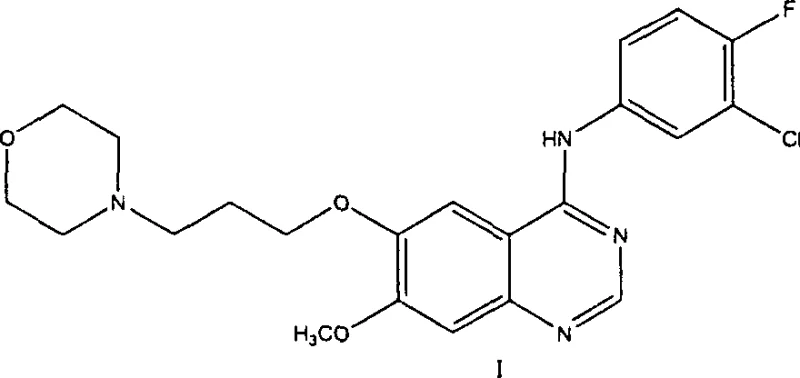
Furthermore, the strategic elimination of hazardous reagents and complex purification steps positions this methodology as a superior candidate for green chemistry initiatives within fine chemical intermediates manufacturing. The ability to synthesize the key intermediate 6-hydroxy-7-methoxy-3,4-dihydroquinazoline-4-ketone directly, without the need for prior protection of the phenolic hydroxyl group, drastically simplifies the operational workflow. This technical breakthrough not only enhances the purity profile of the final active pharmaceutical ingredient but also reduces the environmental footprint associated with solvent usage and waste disposal. As we delve deeper into the comparative analysis of synthetic routes, it becomes evident that this patent offers a viable solution for cost reduction in pharmaceutical manufacturing while maintaining the high purity specifications required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Gefitinib has relied heavily on routes exemplified by early filings such as WO9633980, which utilize 6,7-dimethoxy-3,4-dihydroquinazoline-4-one as the primary precursor. A critical bottleneck in this conventional approach is the selective demethylation of the 6-position methoxy group, a transformation that traditionally necessitates the use of large quantities of methanesulfonic acid and L-methionine. This specific chemical step is fraught with operational challenges, including the difficulty in recovering and recycling the expensive methanesulfonic acid and L-methionine reagents, leading to substantial increases in raw material costs. Moreover, the reaction yield for this demethylation step is notoriously poor, often reported to be less than 50%, which creates a significant burden on the overall mass balance and throughput of the manufacturing plant. The generation of acidic waste streams and the complexity of downstream processing further exacerbate the environmental compliance issues, making this legacy route increasingly unsustainable for modern large-scale production facilities aiming for efficiency.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN1300118C introduces a paradigm shift by initiating the synthesis from 3,4-dimethoxybenzoic acid and employing a novel sequence that bypasses the problematic demethylation of the dimethoxy quinazolinone entirely. Instead of forming the dimethoxy intermediate first, this innovative route synthesizes the precursor 2-amino-4-methoxy-5-hydroxybenzoic acid directly through a concurrent demethylation and reduction process on the nitro-benzoic acid derivative. This strategic modification allows for the direct cyclization to form the 6-hydroxy-7-methoxy-3,4-dihydroquinazoline-4-one core, effectively skipping the need for the low-yielding methanesulfonic acid step. By avoiding the acetylation protection and subsequent deprotection of the hydroxyl group, the total number of synthetic steps is reduced, leading to a more concise and practical process flow. This streamlined approach not only mitigates the environmental hazards associated with strong acids but also significantly improves the overall yield and economic viability of producing this high-value anticancer agent.
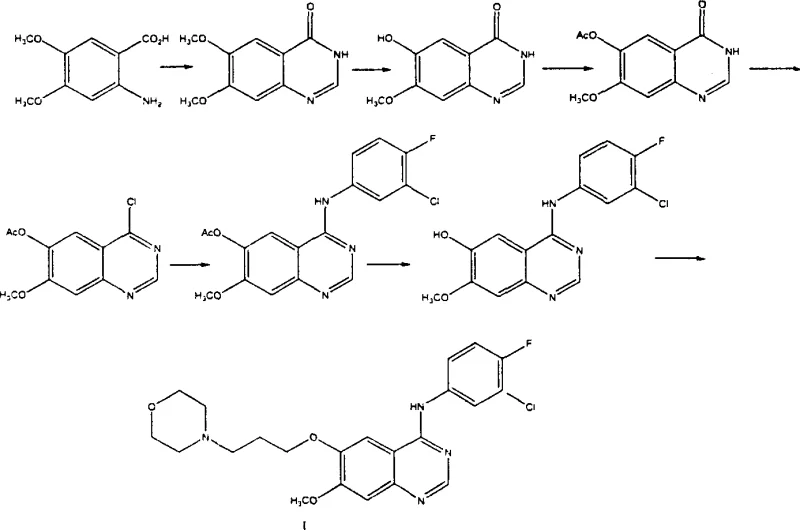
Mechanistic Insights into Selective Demethylation and Direct Cyclization
The core chemical innovation of this patent lies in the sophisticated manipulation of substituent effects on the benzene ring to achieve selective functionalization. The process begins with the nitration of 3,4-dimethoxybenzoic acid to yield 3,4-dimethoxy-6-nitrobenzoic acid, where the nitro group exerts a strong electron-withdrawing effect that activates the adjacent methoxy group at the 4-position (para to the nitro group) towards nucleophilic attack. In the subsequent step, the substrate is subjected to alkaline aqueous conditions in the presence of a reducing agent such as iron powder or sodium sulfide. Under these specific conditions, the activated methoxy group undergoes selective hydrolysis to form a phenolic hydroxyl group, while the nitro group is simultaneously reduced to an amino group. This one-pot tandem reaction is a masterpiece of process chemistry, as it accomplishes two distinct transformations—demethylation and reduction—in a single operational unit, thereby saving time, energy, and solvent resources compared to performing these steps sequentially. The result is the efficient formation of 2-amino-4-methoxy-5-hydroxybenzoic acid, a crucial building block that sets the stage for the construction of the heterocyclic core.
Following the formation of the amino-hydroxy acid intermediate, the synthesis proceeds to the cyclization step, which is critical for establishing the quinazoline ring system. Unlike traditional methods that might require harsh conditions or protecting groups, this patent utilizes formamidine salts, such as formamidine acetate or hydrochloride, as the cyclization reagent. The reaction is conducted at elevated temperatures ranging from 60°C to 210°C, facilitating the condensation between the carboxylic acid and the ortho-amino group with the formamidine source to close the ring. The presence of the free hydroxyl group at the 6-position during this cyclization is tolerated and does not interfere with the ring closure, which is a significant advantage over routes that require prior acetylation. Subsequent chlorination using reagents like thionyl chloride or phosphorus oxychloride converts the ketone functionality into a chloro group, creating a highly reactive electrophile. This chloro-intermediate then undergoes nucleophilic aromatic substitution with 3-chloro-4-fluoroaniline to install the anilino side chain, followed by final alkylation with morpholinyl chloropropane to complete the synthesis of Gefitinib with high structural fidelity.
How to Synthesize Gefitinib Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize the benefits of the tandem demethylation-reduction and direct cyclization steps. Operators must carefully manage the temperature profiles during the nitration and reduction phases to ensure selectivity and prevent over-reaction or degradation of the sensitive amino-hydroxy intermediate. The choice of reducing agent and base concentration plays a pivotal role in determining the success of the one-pot transformation, with iron powder and potassium hydroxide being highlighted as effective reagents in the patent examples. Furthermore, the cyclization step demands rigorous temperature control to drive the reaction to completion while minimizing the formation of polymeric byproducts. For technical teams looking to adopt this methodology, it is essential to establish robust in-process controls to monitor the conversion of the nitro precursor to the amino acid and subsequently to the quinazolinone core. The detailed standardized synthesis steps see the guide below for specific operational parameters and stoichiometry derived from the patent embodiments.
- Nitration of 3,4-dimethoxybenzoic acid to form 3,4-dimethoxy-6-nitrobenzoic acid.
- One-pot selective demethylation and reduction to obtain 2-amino-4-methoxy-5-hydroxybenzoic acid.
- Direct cyclization using formamidine salts to form the quinazolinone core without hydroxyl protection.
- Chlorination followed by nucleophilic substitution with 3-chloro-4-fluoroaniline.
- Final etherification with morpholinyl chloropropane to yield Gefitinib.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthetic route described in Patent CN1300118C offers compelling advantages for procurement managers and supply chain heads focused on cost optimization and reliability. The most significant economic driver is the elimination of the methanesulfonic acid and L-methionine demethylation step, which is not only low-yielding but also involves expensive reagents that are difficult to recover. By removing this bottleneck, manufacturers can achieve a substantial reduction in raw material costs and waste treatment expenses, directly impacting the bottom line of API production. Additionally, the reduction in the total number of synthetic steps—from nitration to final etherification—translates to shorter cycle times and higher throughput capacity within existing manufacturing infrastructure. This efficiency gain allows suppliers to respond more agilely to market demand fluctuations, ensuring a steady supply of high-purity pharmaceutical intermediates without the delays associated with complex multi-step purifications.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for costly protecting group chemistry, specifically the acetylation and deacetylation sequences required in older routes. This simplification reduces the consumption of acetic anhydride and other acylating agents, along with the solvents needed for their removal. Furthermore, the avoidance of the low-yield demethylation step significantly improves the overall mass yield of the process, meaning less starting material is required to produce the same amount of final product. These factors combine to lower the cost of goods sold (COGS), providing a competitive pricing advantage for buyers sourcing this critical oncology intermediate from suppliers utilizing this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available and commodity-grade starting materials, such as 3,4-dimethoxybenzoic acid, mitigates the risk of supply disruptions often associated with specialized or custom-synthesized precursors. The robustness of the one-pot demethylation-reduction reaction also reduces the likelihood of batch failures due to intermediate isolation issues, thereby enhancing the consistency of supply. For supply chain planners, this translates to more predictable lead times and a reduced need for safety stock, as the manufacturing process is less prone to the variability inherent in longer, more complex synthetic sequences. This stability is crucial for maintaining continuous production schedules for life-saving medications like Gefitinib.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common solvents and reagents that are easily handled in large-scale reactors. The reduction in hazardous waste generation, particularly the avoidance of large volumes of spent methanesulfonic acid, simplifies environmental compliance and lowers the cost of waste disposal. This aligns with the growing global emphasis on sustainable manufacturing practices and green chemistry principles. Facilities adopting this route can operate with a smaller environmental footprint, reducing regulatory risks and enhancing the corporate social responsibility profile of the supply chain. The ability to scale from kilogram to multi-ton production without significant process redesign makes this an attractive option for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Gefitinib. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational benefits and chemical rationale behind the new methodology. Understanding these nuances is essential for technical evaluators and procurement specialists who are assessing the feasibility of integrating this supply source into their existing vendor networks. The answers reflect the specific improvements in yield, step count, and reagent usage that distinguish this approach from conventional methods.
Q: Why is the methanesulfonic acid demethylation method considered disadvantageous?
A: The conventional method utilizing methanesulfonic acid and L-methionine for demethylation suffers from significant environmental pollution due to difficult recovery of reagents and typically yields less than 50%, making it economically inefficient for large-scale production.
Q: What is the key innovation in Patent CN1300118C regarding the quinazolinone core formation?
A: The patent introduces a direct cyclization of 2-amino-4-methoxy-5-hydroxybenzoic acid using formamidine salts, which eliminates the need for acetyl protection and deprotection steps required in previous routes, thereby shortening the synthetic sequence.
Q: How does this new route impact the overall yield and scalability?
A: By removing the low-yielding demethylation step (<50%) and reducing the total number of reaction steps, the new route significantly improves overall throughput and is more suitable for industrial scale-up due to the use of readily available raw materials like 3,4-dimethoxybenzoic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the pharmaceutical sector. Our team of expert chemists has extensively analyzed advanced synthetic routes like the one described in Patent CN1300118C to ensure that our production capabilities align with the highest standards of innovation and quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly transition promising laboratory methodologies into robust industrial processes. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that verify every batch of API intermediate against global pharmacopeial standards, ensuring that our clients receive materials that are ready for immediate formulation.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your supply chain goals. By leveraging our expertise in complex organic synthesis, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, and discover how NINGBO INNO PHARMCHEM can serve as your strategic partner in the reliable supply of high-quality Gefitinib and related pharmaceutical intermediates.
