Advanced Carbazole-Based Emissive Materials for Next-Generation OLED Displays
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that balance high efficiency with exceptional operational stability. Patent CN106967060B introduces a groundbreaking series of carbazole derivatives designed to address the critical bottlenecks in current display technologies. These compounds, characterized by a unique central benzene core flanked by carbazole units and various heterocyclic substituents, represent a significant leap forward in fluorescent emitter design. Unlike traditional phosphorescent materials that rely on scarce and costly heavy metals, these organic structures leverage thermally activated delayed fluorescence (TADF) mechanisms or highly efficient fluorescence to achieve superior performance. The patent details a comprehensive library of over one hundred specific embodiments, demonstrating the versatility of this chemical scaffold in tuning emission colors from orange-yellow to red while maintaining robust thermal properties. For R&D directors seeking reliable OLED material supplier partnerships, this technology offers a pathway to devices with significantly improved power efficiency and external quantum efficiency (EQE). The structural integrity of these molecules ensures that they can withstand the rigorous thermal evaporation processes required for device fabrication without degradation.
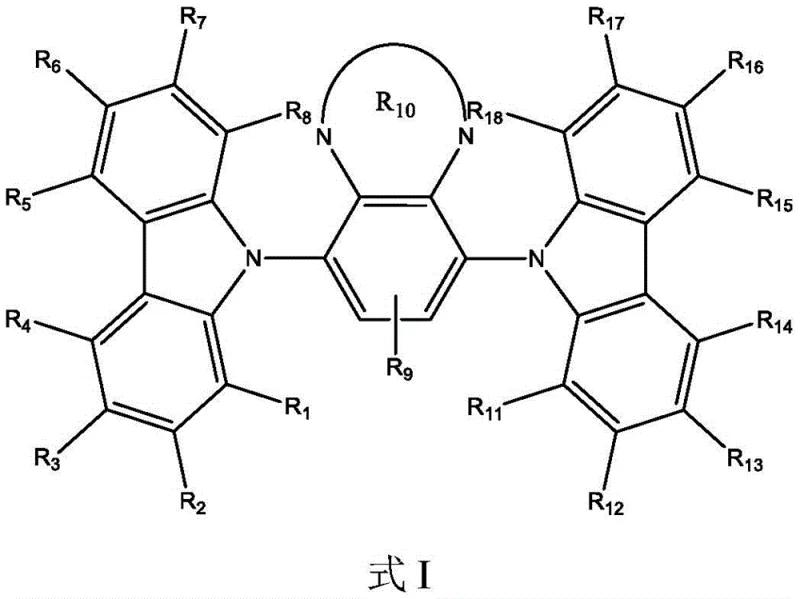
Beyond mere efficiency, the commercial viability of these materials is underpinned by their synthesis scalability. The patent outlines straightforward synthetic routes that avoid exotic reagents, making them ideal candidates for cost reduction in electronic chemical manufacturing. By utilizing readily available starting materials such as carbazole, diamines, and simple aldehydes, the production process minimizes supply chain risks associated with specialized precursors. This accessibility is crucial for supply chain heads who prioritize continuity and lead time management. Furthermore, the purification methods described, often involving simple recrystallization or column chromatography, are easily adaptable to kilogram-scale operations. This ensures that the transition from laboratory discovery to commercial production is seamless, allowing manufacturers to meet the growing demand for high-resolution displays in smartphones, televisions, and wearable devices. The ability to fine-tune the energy levels through substituent modification means that these materials can be precisely matched with various host systems, optimizing charge balance within the emissive layer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
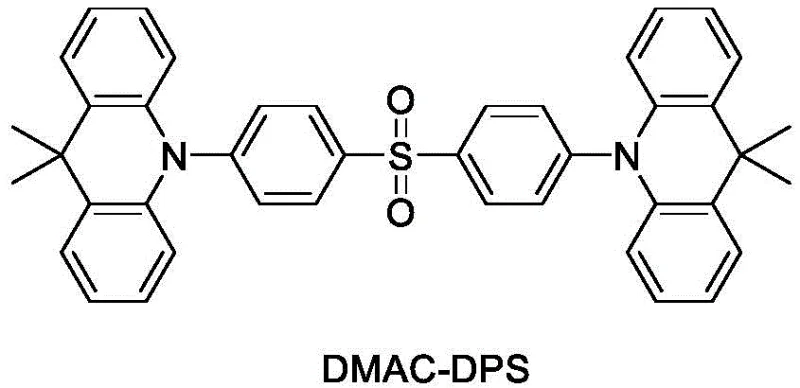
Traditional approaches to achieving high-efficiency OLEDs have largely depended on phosphorescent emitters containing iridium or platinum complexes. While these materials boast high internal quantum efficiencies, they suffer from significant drawbacks, including the prohibitive cost of noble metals and stability issues, particularly in the blue and deep-red regions of the spectrum. Moreover, the synthesis of these metal complexes often involves complex multi-step procedures with low overall yields and difficult purification requirements to remove trace metal impurities that can quench luminescence. Even among fluorescent materials, conventional options often struggle with aggregation-caused quenching (ACQ) in the solid state, leading to reduced efficiency in actual devices. Comparative materials like DMAC-DPS, while useful, have shown limitations in terms of operational lifetime and efficiency when subjected to high brightness conditions. The reliance on specific host-guest interactions that are not universally compatible further restricts the flexibility of device architects. These factors collectively contribute to higher manufacturing costs and limited device longevity, creating a pressing need for alternative molecular designs that can overcome these inherent physical and economic constraints.
The Novel Approach
The novel approach presented in this patent utilizes a rigidified carbazole-based architecture that inherently suppresses non-radiative decay pathways. By linking carbazole units to a central benzene ring through nitrogen atoms and incorporating additional heterocyclic moieties, the resulting molecules exhibit high glass transition temperatures and excellent film-forming properties. This structural rigidity prevents the molecular motion that typically leads to energy loss as heat, thereby enhancing the radiative decay rate. The patent demonstrates that devices fabricated using these materials, such as OLED-2 employing compound A82, achieve an EQE of 18.6% and a lifetime (T95) of 5000 hours, outperforming the DMAC-DPS reference device which showed an EQE of 17.0% and a lifetime of only 3000 hours. This improvement is not merely incremental but represents a substantial enhancement in device reliability. The synthetic strategy focuses on forming robust C-N bonds via copper-catalyzed coupling, a reaction known for its scalability and tolerance to various functional groups. This allows for the easy introduction of diverse substituents to modulate the HOMO-LUMO gap, enabling precise color tuning without compromising the core stability of the emitter.
Mechanistic Insights into Cu-Catalyzed Ullmann Coupling and Cyclization
The core synthetic mechanism driving the formation of these advanced materials revolves around the copper-catalyzed Ullmann-type C-N coupling reaction. In this process, a halogenated aromatic core, such as 4,7-dibromobenzothiadiazole, reacts with carbazole derivatives in the presence of a copper catalyst like cuprous iodide (CuI) and a base such as potassium carbonate. The reaction typically proceeds in high-boiling solvents like xylene or DMF at elevated temperatures, facilitating the oxidative addition of the copper species into the carbon-halogen bond. This is followed by coordination with the nitrogen lone pair of the carbazole and subsequent reductive elimination to form the stable C-N bond. The patent highlights the use of ligands like L-proline to accelerate this transformation and improve yields, which is critical for minimizing waste and maximizing throughput in a commercial setting. Following the coupling, a condensation step with dialdehydes or quinones creates the final fused ring system. This cyclization locks the molecular conformation, reducing vibrational freedom and enhancing fluorescence quantum yield. The careful control of reaction stoichiometry and temperature during these steps ensures high purity, which is paramount for preventing trap states in the final OLED device that could degrade performance.
Impurity control is another critical aspect of the mechanistic design, particularly given the sensitivity of OLED materials to trace contaminants. The synthetic route is designed to produce byproducts that are chemically distinct from the target molecule, facilitating their removal via standard purification techniques like recrystallization or silica gel chromatography. For instance, the use of excess amine or specific workup procedures involving acid-base extraction helps to remove unreacted starting materials and copper residues effectively. The patent emphasizes that the final products exhibit high thermal stability, with decomposition temperatures well above the processing temperatures used in vacuum deposition. This thermal robustness is a direct result of the extended conjugation and rigid backbone formed during the cyclization steps. By minimizing the presence of flexible alkyl chains that could introduce disorder, the materials maintain a uniform amorphous state in the thin film. This homogeneity is essential for ensuring consistent charge transport across the device area, preventing local hot spots that could lead to premature device failure. The mechanistic understanding of these structure-property relationships allows chemists to predictively design new analogs with even better performance characteristics.
How to Synthesize Carbazole Derivatives Efficiently
The synthesis of these high-performance carbazole derivatives follows a logical, modular sequence that is well-suited for process optimization. The initial steps involve the preparation of key intermediates, such as halogenated benzothiadiazoles or diamino-carbazole precursors, which serve as the building blocks for the final assembly. These intermediates are then coupled under optimized conditions to ensure high conversion rates. The final cyclization step is the most critical, as it defines the electronic properties of the emitter. Detailed protocols in the patent specify precise molar ratios, solvent volumes, and reaction times to maximize yield and purity. For example, the reaction of diamino intermediates with phenanthrenequinone in glacial acetic acid under reflux is a standard procedure that consistently produces high-quality material. The simplicity of these operations means that they can be transferred to pilot plants with minimal re-engineering. For a complete breakdown of the reaction parameters and workup procedures, please refer to the standardized synthesis guide below.
- Preparation of halogenated intermediates via bromination of benzothiadiazole or similar cores under reflux conditions.
- Execution of copper-catalyzed Ullmann coupling between halogenated cores and carbazole derivatives to form C-N bonds.
- Final cyclization or condensation with dialdehydes or quinones in acidic media to form the rigid fused-ring target structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards these metal-free carbazole derivatives offers profound strategic advantages. The elimination of precious metals like iridium from the supply chain removes a significant source of price volatility and geopolitical risk. Traditional phosphorescent dopants are subject to fluctuating market prices driven by mining outputs and refining capacities, whereas the organic precursors for these carbazole materials are derived from petrochemical feedstocks with stable and abundant supplies. This transition enables substantial cost savings in raw material acquisition, allowing manufacturers to improve their margins or offer more competitive pricing to downstream electronics consumers. Furthermore, the simplified synthesis reduces the number of processing steps required, which directly correlates to lower energy consumption and reduced labor costs per kilogram of product. The high yields reported in the patent examples, often exceeding 80% for key steps, minimize waste generation and solvent usage, aligning with modern green chemistry initiatives and reducing disposal costs. These factors combine to create a highly attractive cost structure for large-volume manufacturing.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of expensive transition metal catalysts that require complex removal processes, thereby streamlining the production workflow. By utilizing common reagents like copper iodide and sodium hydroxide, the material costs are kept low while maintaining high reaction efficiency. The high purity achieved through simple recrystallization reduces the need for expensive preparative HPLC purification, further lowering the cost of goods sold. This economic efficiency makes the materials accessible for a wider range of consumer electronics applications, not just premium flagship devices.
- Enhanced Supply Chain Reliability: The starting materials, such as carbazole and various brominated aromatics, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and ensures continuous availability even during market disruptions. The robust nature of the chemical bonds in the final product also implies a longer shelf life, reducing inventory write-offs due to degradation. For supply chain managers, this translates to greater flexibility in planning and a reduced need for safety stock, optimizing working capital utilization.
- Scalability and Environmental Compliance: The reactions described operate under standard atmospheric pressure and use solvents that are easily recoverable and recyclable. The absence of heavy metals simplifies wastewater treatment and waste disposal, ensuring compliance with stringent environmental regulations in major manufacturing hubs. The process is inherently scalable from gram to ton quantities without significant changes in reaction kinetics, facilitating rapid capacity expansion to meet surging market demand. This scalability ensures that the supply can grow in tandem with the adoption of OLED technology in new sectors like automotive lighting and solid-state lighting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these carbazole derivatives in OLED production lines. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating these materials into existing device architectures. The data confirms that these materials are not just theoretical constructs but have been validated in actual device configurations with measurable performance gains.
Q: What are the key performance advantages of these carbazole derivatives over traditional phosphorescent materials?
A: These carbazole derivatives offer high external quantum efficiency (EQE up to 21.6%) and prolonged device lifetime without relying on expensive rare earth metals like iridium or platinum, significantly reducing raw material costs while maintaining high thermal stability.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the synthesis and purification methods are simple and suitable for large-scale production. The reactions utilize common reagents like cuprous iodide and potassium carbonate, avoiding complex transition metal catalysts that require difficult removal steps.
Q: How do these materials improve OLED device longevity?
A: The rigid molecular structure and high glass transition temperature of these derivatives prevent crystallization and aggregation during operation. Experimental data shows device lifetimes (T95) extending up to 6000 hours, significantly outperforming comparative materials like DMAC-DPS.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the carbazole derivatives described in patent CN106967060B for the future of display technology. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring these advanced materials to market. Our facilities are equipped with state-of-the-art reactors capable of handling the high-temperature coupling and condensation reactions safely and efficiently. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards of the OLED industry. Our team of expert chemists is ready to assist in optimizing the synthetic route for your specific volume requirements, ensuring consistent quality and supply continuity.
We invite you to collaborate with us to unlock the full commercial potential of these high-efficiency emissive materials. By leveraging our expertise, you can accelerate your product development cycles and secure a competitive edge in the marketplace. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Let us help you navigate the complexities of OLED material production with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
