Advanced Manufacturing of Beta-Nitrostyrene Intermediates via Safe Acetic Acid Condensation
The pharmaceutical and fine chemical industries are constantly seeking robust manufacturing pathways that balance high efficiency with uncompromising safety standards, particularly when handling energetic nitro compounds. Patent CN101765579A introduces a transformative methodology for the production of beta-nitrostyrene compounds, which serve as critical intermediates in the synthesis of therapeutic agents such as baclofen for reflux esophagitis and various antispasmodics. This innovation addresses the longstanding thermal hazards associated with traditional nitro-condensation reactions by shifting the reaction paradigm from hazardous high-temperature reflux to a controlled, moderate-temperature regime. By leveraging a primary amine catalyst within an acetic acid solvent system, this technology enables manufacturers to achieve exceptional yields while maintaining an industrially safe reaction window, thereby mitigating the risks of thermal runaway that have historically plagued the scale-up of nitro-styrene derivatives.
For R&D directors and process chemists evaluating reliable pharmaceutical intermediate supplier options, the distinction between legacy protocols and this novel approach is stark. Conventional methods, such as those disclosed in earlier Japanese patents, typically rely on ammonium acetate as a base and necessitate heating the reaction mixture to near-boiling temperatures to drive the condensation of benzaldehyde derivatives with nitromethane. These harsh conditions not only consume excessive energy but also create a precarious safety environment; Accelerating Rate Calorimeter (ARC) data indicates that under traditional operating temperatures of 95°C, the time to maximum self-reaction rate (thermal runaway) is merely 7.9 hours, which is dangerously close to typical batch residence times. In contrast, the novel approach utilizes primary amines like benzylamine or n-propylamine in acetic acid, allowing the reaction to proceed efficiently at significantly lower temperatures of 70-80°C, drastically extending the safety margin and preventing uncontrolled exothermic events.
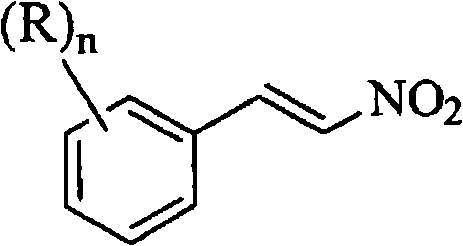
The mechanistic advantages of this primary amine-catalyzed condensation extend beyond mere safety metrics to encompass superior reaction kinetics and impurity control. The use of acetic acid as the solvent plays a dual role: it acts as a polar medium that stabilizes the transition state of the Henry reaction (nitroaldol condensation) and facilitates a unique downstream processing strategy. Unlike non-polar solvents like toluene or nitroalkanes used in prior art, acetic acid allows for the direct crystallization of the product upon the addition of water during the workup phase. This eliminates the need for complex distillation steps to remove high-boiling solvents, thereby reducing the thermal load on the final product and minimizing the formation of degradation byproducts. Furthermore, the specific selection of primary amines, particularly benzylamine, optimizes the basicity of the system to promote rapid enolate formation from nitromethane without inducing excessive side reactions, ensuring a clean conversion to the target beta-nitrostyrene structure.
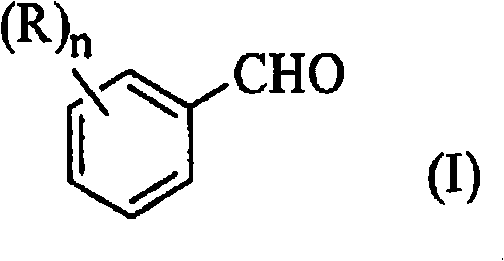
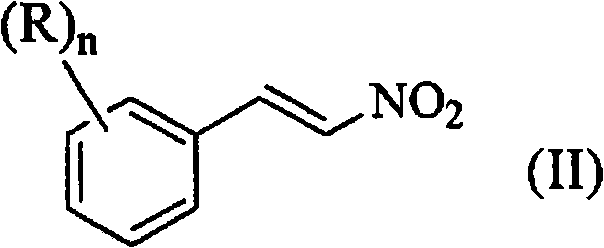
From a structural perspective, the versatility of this synthesis allows for the accommodation of diverse substituents on the aromatic ring, as defined by Formula (I) in the patent documentation. Whether the starting material is 4-chlorobenzaldehyde or derivatives containing alkyl, alkoxy, or haloalkyl groups, the reaction conditions remain robust, delivering consistent performance across a broad substrate scope. The resulting beta-nitrostyrene compounds, represented by Formula (II), are obtained with purities that meet the rigorous demands of downstream API synthesis. The ability to operate at an Adiabatic Decomposition Temperature for 24 hours (ADT24) of 78°C means that even in the event of cooling failure, the process remains stable for a full day, providing supply chain heads with the confidence needed for commercial scale-up of complex pharmaceutical intermediates without the fear of catastrophic reactor incidents.
How to Synthesize Beta-Nitrostyrene Efficiently
The implementation of this advanced synthesis route requires precise control over reagent stoichiometry and addition rates to maximize the benefits of the primary amine catalytic system. The process begins with the dissolution of the benzaldehyde derivative and the selected primary amine in glacial acetic acid, followed by the controlled addition of nitromethane. Detailed operational parameters, including specific molar ratios and temperature ramps, are critical for reproducing the high yields reported in the patent data. For technical teams looking to adopt this methodology, the following guide outlines the standardized synthetic steps derived directly from the exemplary embodiments of the invention, ensuring a smooth transition from laboratory bench to pilot plant operations.
- Dissolve the benzaldehyde derivative (e.g., 4-chlorobenzaldehyde) and a primary amine catalyst (e.g., benzylamine) in an acetic acid solvent.
- Heat the solution to a safe reaction temperature range of 70-80°C and slowly add nitromethane dropwise over 1 to 3 hours.
- After reaction completion, add water to the mixture at 30-60°C to induce crystallization of the high-purity beta-nitrostyrene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented manufacturing process translates into tangible operational improvements and risk mitigation. The shift away from high-temperature reflux conditions significantly reduces the energy consumption required for heating and cooling cycles, leading to substantial cost savings in utility expenditures over the lifecycle of the production campaign. Moreover, the enhanced safety profile minimizes the need for expensive specialized containment equipment and reduces insurance premiums associated with handling energetic nitro compounds, effectively lowering the overall cost of goods sold. By eliminating the reliance on hazardous high-temperature operations, facilities can achieve higher throughput rates with reduced downtime for safety inspections and maintenance, ensuring a more reliable supply of critical intermediates to downstream customers.
- Cost Reduction in Manufacturing: The utilization of acetic acid as a solvent offers a distinct economic advantage over traditional organic solvents due to its low cost and ease of recovery. The process design eliminates the need for energy-intensive distillation steps to separate the product from the reaction matrix, as simple water-induced crystallization suffices for isolation. This streamlined workup not only reduces solvent waste disposal costs but also shortens the overall batch cycle time, allowing for increased production capacity within existing infrastructure without the need for significant capital investment in new reactor trains.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain continuity, and the improved thermal stability of this process directly correlates to reduced operational risk. By operating well below the threshold for thermal runaway, manufacturers can guarantee consistent delivery schedules without the interruptions caused by safety incidents or regulatory shutdowns. The robustness of the reaction across various substituted benzaldehyde derivatives ensures that raw material sourcing remains flexible, allowing procurement teams to adapt to market fluctuations in specific aldehyde availability without compromising the integrity of the final beta-nitrostyrene product quality.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, including the use of less toxic solvents and the generation of cleaner waste streams, align perfectly with modern environmental regulations. The aqueous workup generates wastewater that is easier to treat compared to organic solvent-laden effluents from traditional methods, simplifying compliance with discharge limits. Furthermore, the inherent safety of the low-temperature protocol facilitates seamless scale-up from kilogram to multi-ton scales, enabling manufacturers to respond rapidly to surging demand for pharmaceutical intermediates while maintaining strict adherence to global safety and environmental standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its potential impact on their supply chains. The following questions address common inquiries regarding the safety mechanisms, yield improvements, and purification strategies associated with this primary amine-catalyzed process. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the technology's capabilities and limitations for potential partners and licensees.
Q: Why is the acetic acid method safer than traditional ammonium acetate reflux?
A: Traditional methods using ammonium acetate require reflux temperatures near the boiling point, creating a high risk of thermal runaway (ADI24 < 8 hours at 95°C). The new primary amine method operates safely at 70-80°C with an ADT24 of 78°C, preventing runaway reactions for over 24 hours.
Q: What yields can be expected from this beta-nitrostyrene synthesis route?
A: By utilizing primary amines like benzylamine in acetic acid, the process achieves significantly higher yields ranging from 80% to 99%, compared to the roughly 67% yields often seen in older nitroalkane solvent methods.
Q: How does this process impact impurity profiles for pharmaceutical intermediates?
A: The use of acetic acid facilitates a unique workup where water addition induces direct crystallization. This simplifies purification, removes residual amines effectively, and ensures the stringent purity specifications required for API intermediates like baclofen precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nitrostyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient manufacturing processes is vital for the long-term sustainability of the pharmaceutical supply chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the acetic acid condensation method are implemented with the highest regard for quality and safety. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-nitrostyrene intermediate meets the exacting standards required for the synthesis of life-saving medications like baclofen.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these advanced synthetic technologies for their next-generation drug candidates. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the most cost-effective and safe manufacturing solutions available in the global market.
