Advanced Manufacturing of Prasugrel Intermediates for Global Pharmaceutical Supply Chains
The global demand for effective antiplatelet therapies continues to drive innovation in the synthesis of cardiovascular pharmaceuticals, with Prasugrel standing out as a critical second-generation thienopyridine. Patent CN102030761A introduces a transformative approach to manufacturing the key intermediates required for this potent medication, addressing long-standing inefficiencies in legacy production methods. This technical disclosure outlines a streamlined three-step synthetic pathway that begins with the phase-transfer catalyzed cyanation of o-fluorobenzyl bromide, followed by selective alpha-bromination and a final nucleophilic coupling with a substituted thienopyridine scaffold. 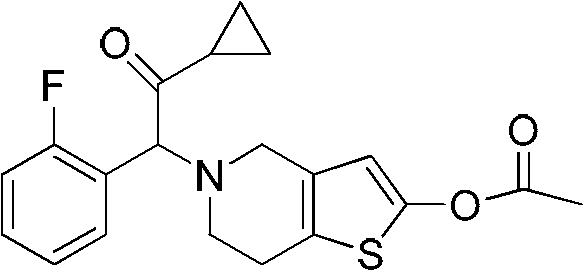 . For R&D directors and procurement specialists, this methodology represents a significant leap forward, offering a route that is not only chemically robust but also economically viable for multi-ton production. By focusing on readily available starting materials and avoiding complex catalytic systems, this process ensures a stable supply of high-purity precursors essential for the final drug substance.
. For R&D directors and procurement specialists, this methodology represents a significant leap forward, offering a route that is not only chemically robust but also economically viable for multi-ton production. By focusing on readily available starting materials and avoiding complex catalytic systems, this process ensures a stable supply of high-purity precursors essential for the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prasugrel and its precursors has been plagued by significant technical hurdles that impede efficient commercial manufacturing. As illustrated in prior art such as patent EP542411, traditional routes often suffer from sluggish reaction kinetics and a propensity for generating complex mixtures of side products. 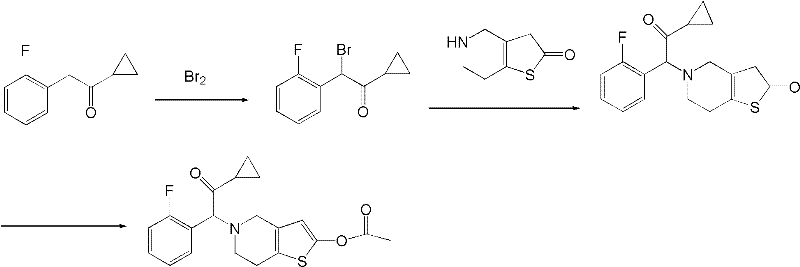 . These inefficiencies result in lower overall yields and necessitate rigorous, costly purification protocols to meet the stringent purity specifications required for pharmaceutical intermediates. Furthermore, older methods frequently rely on reagents that impose heavy environmental burdens, creating substantial waste disposal challenges and increasing the total cost of ownership for manufacturers. The difficulty in separating the desired isomer from by-products in these conventional pathways often leads to extended production cycles, thereby compromising supply chain reliability and increasing lead times for downstream API producers.
. These inefficiencies result in lower overall yields and necessitate rigorous, costly purification protocols to meet the stringent purity specifications required for pharmaceutical intermediates. Furthermore, older methods frequently rely on reagents that impose heavy environmental burdens, creating substantial waste disposal challenges and increasing the total cost of ownership for manufacturers. The difficulty in separating the desired isomer from by-products in these conventional pathways often leads to extended production cycles, thereby compromising supply chain reliability and increasing lead times for downstream API producers.
The Novel Approach
In stark contrast, the methodology disclosed in CN102030761A offers a refined and highly efficient alternative that directly addresses these historical pain points. The core innovation lies in the strategic construction of the carbon-nitrogen bond through a controlled coupling reaction between an alpha-bromo nitrile and a protected thienopyridine derivative. This novel approach utilizes a phase-transfer catalyst system that dramatically accelerates the initial cyanation step, ensuring high conversion rates under mild thermal conditions. The subsequent bromination is carefully managed to prevent over-halogenation, preserving the integrity of the fluorophenyl ring while activating the alpha-position for nucleophilic attack. By optimizing solvent systems and base selection, this route minimizes side reactions, resulting in a crude product profile that is significantly cleaner than that of prior art. This structural simplicity translates directly into operational excellence, allowing for easier isolation of the target intermediate and reducing the reliance on resource-intensive chromatographic separations.
Mechanistic Insights into Phase-Transfer Catalyzed Cyanation and Coupling
The chemical elegance of this synthesis is rooted in the precise manipulation of nucleophilicity and electrophilicity across three distinct stages. The process initiates with the conversion of o-fluorobenzyl bromide to o-fluorophenylacetonitrile, a transformation facilitated by a quaternary ammonium or phosphonium salt phase-transfer catalyst. This catalyst shuttles the cyanide anion from the aqueous phase into the organic phase, where it reacts rapidly with the benzyl bromide, overcoming the solubility barriers that typically limit such biphasic reactions. Following this, the nitrile undergoes alpha-bromination using reagents such as molecular bromine or N-bromosuccinimide. This step generates a highly reactive electrophile, the alpha-bromo nitrile, which is primed for the subsequent displacement reaction. The final and most critical stage involves the coupling of this electrophile with the nitrogen atom of the tetrahydrothieno[3,2-c]pyridine ring. 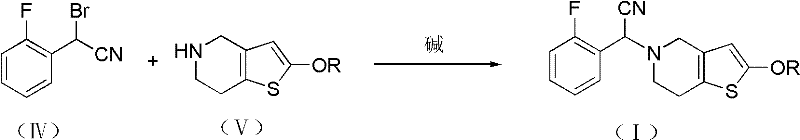 . The use of a non-nucleophilic base ensures that the deprotonation of the thienopyridine occurs selectively, driving the formation of the C-N bond without degrading the sensitive ester or ether protecting groups present on the heterocyclic ring.
. The use of a non-nucleophilic base ensures that the deprotonation of the thienopyridine occurs selectively, driving the formation of the C-N bond without degrading the sensitive ester or ether protecting groups present on the heterocyclic ring.
From an impurity control perspective, this mechanism offers superior manageability compared to transition-metal catalyzed cross-couplings. The absence of palladium or copper catalysts eliminates the risk of heavy metal contamination, a critical quality attribute for regulatory compliance. The primary impurities arise from potential over-bromination or hydrolysis of the nitrile group, both of which are effectively suppressed by maintaining strict temperature controls and stoichiometric balance during the reaction. The use of protecting groups, such as the tert-butyldimethylsilyl (TBS) ether or methoxymethyl groups on the thienopyridine oxygen, further enhances selectivity by preventing O-alkylation side reactions. This mechanistic robustness ensures that the final intermediate possesses a consistent impurity profile, simplifying the validation process for GMP manufacturing and reducing the analytical burden on quality control laboratories tasked with releasing batches for further processing.
How to Synthesize Prasugrel Intermediate Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters to maximize yield and purity while maintaining safety standards. The process is designed to be scalable, utilizing standard reactor configurations found in most fine chemical facilities. Operators must monitor the progress of the cyanation and bromination steps closely, typically using thin-layer chromatography (TLC) or HPLC to determine the precise endpoint before proceeding to the next stage. The choice of solvent is also critical; biphasic systems of toluene and water are preferred for the cyanation to facilitate phase transfer, while polar aprotic solvents like acetonitrile or DMF are optimal for the final coupling step to solubilize the ionic intermediates.
- Perform phase-transfer cyanation of o-fluorobenzyl bromide using sodium cyanide and a quaternary ammonium salt catalyst to yield o-fluorophenylacetonitrile.
- Conduct controlled alpha-bromination of the nitrile intermediate using bromine or N-bromosuccinimide in a halogenated solvent to form o-fluoro-alpha-bromophenylacetonitrile.
- Execute the key coupling reaction between the brominated nitrile and a protected thienopyridine derivative in the presence of a base to form the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The reliance on commodity chemicals such as o-fluorobenzyl bromide, sodium cyanide, and common organic solvents creates a resilient supply chain that is less susceptible to the volatility associated with specialized reagents. By eliminating the need for expensive transition metal catalysts and the subsequent scavenging steps required to remove them, the overall cost of goods sold (COGS) is significantly reduced. This cost efficiency is further amplified by the high atom economy of the coupling reaction, which minimizes waste generation and lowers the environmental compliance costs associated with hazardous waste disposal. The simplified workup procedures, often involving basic extraction and crystallization rather than complex chromatography, also reduce the consumption of silica gel and elution solvents, contributing to a more sustainable and cost-effective manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive, bulk-available starting materials drastically lower the raw material expenditure. Furthermore, the high selectivity of the reaction reduces the loss of valuable intermediates to side products, maximizing the output per batch and improving the overall return on investment for production campaigns.
- Enhanced Supply Chain Reliability: Since the raw materials are standard industrial chemicals with multiple global suppliers, the risk of supply disruption is minimized. The robustness of the chemistry allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuations in market demand for cardiovascular medications without the long lead times associated with sourcing exotic reagents.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram to kilogram scales in the patent examples. The reduction in hazardous waste and the avoidance of heavy metals simplify the environmental permitting process and align with modern green chemistry principles, making it an attractive option for facilities operating under strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities.
Q: What are the primary advantages of this synthesis route over prior art methods?
A: This method eliminates the slow reaction rates and low yields associated with conventional routes like EP542411. By utilizing phase-transfer catalysis and mild bromination conditions, it significantly reduces side reactions and simplifies purification, leading to higher overall purity and suitability for industrial scale-up.
Q: How does this process impact environmental compliance and waste management?
A: The process is designed with low environmental pressure in mind. It avoids the use of harsh transition metal catalysts that require complex removal steps and generates fewer hazardous by-products. The use of common solvents like toluene and acetonitrile facilitates easier recycling and waste treatment compared to older methodologies.
Q: Is this intermediate suitable for large-scale commercial production of Prasugrel?
A: Yes, the patent explicitly highlights the method's suitability for industrialized production. The raw materials such as o-fluorobenzyl bromide are commercially available and cost-effective, and the operational steps involve standard unit operations like extraction and crystallization, ensuring reliable supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prasugrel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key pharmaceutical intermediates like those described in CN102030761A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Prasugrel intermediate we deliver adheres to the highest international standards. We understand that consistency is key in the pharmaceutical industry, and our dedicated technical team works closely with clients to optimize process parameters for maximum efficiency and yield.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next production campaign. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global cardiovascular drug market.
