Industrial Scale Production of High-Purity Tebipenem via Optimized Hydrogenation
The pharmaceutical landscape for oral carbapenems has been significantly advanced by the development of tebipenem, a potent prodrug known commercially as L-084. As detailed in patent CN102304129B, a groundbreaking method has been established that addresses the critical bottlenecks in synthesizing the active parent drug, tebipenem. This innovation is particularly vital for a reliable pharmaceutical intermediates supplier aiming to meet the stringent quality demands of modern antibiotic production. The traditional pathways often suffered from excessive solvent consumption and suboptimal purity profiles, which complicated downstream processing. In contrast, this novel technique leverages a refined catalytic hydrogenation strategy followed by an aqueous recrystallization process, achieving a final product purity exceeding 99.9 percent. By optimizing the reaction conditions and eliminating the need for organic solvents during the refining stage, this method not only enhances the chemical integrity of the intermediate but also aligns with green chemistry principles essential for sustainable manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing tebipenem, such as those referenced in J. Antibiot. 59(4): 241-247, 2006, relied heavily on inefficient precipitation techniques that hindered industrial scalability. Specifically, the conventional process involved catalytic hydrogenation followed by pH adjustment and precipitation using cold acetone. A major drawback of this legacy approach was the enormous volume of acetone required, with a mass-to-volume ratio of approximately 100:1 relative to the starting material. This excessive solvent usage not only inflated operational costs but also created significant challenges in solvent recovery and waste management. Furthermore, the yield of the crude product was disappointingly low, hovering around 30 percent, and the resulting purity was merely about 98 percent. Such purity levels are insufficient for direct progression to the subsequent esterification step required to produce L-084, as impurities greater than 1 percent can compromise the safety and efficacy of the final oral medication.
The Novel Approach
The innovative method disclosed in the patent fundamentally reengineers the isolation and purification stages to overcome these historical inefficiencies. Instead of relying on acetone precipitation, the new protocol utilizes a tetrahydrofuran (THF) and water system for the initial crystallization of the crude product. This shift allows for a much more controlled crystallization environment where the solvent ratio can be optimized between 1:8 and 1:20. More importantly, the refining step completely abandons organic solvents in favor of pure water for recrystallization. By dissolving the crude tebipenem in hot water at temperatures between 40 and 80 degrees Celsius and subsequently cooling the filtrate, manufacturers can obtain white crystals with exceptional purity. This transition from organic precipitation to aqueous recrystallization represents a paradigm shift in cost reduction in API manufacturing, drastically simplifying the workflow while simultaneously boosting the quality of the output to meet rigorous pharmacopeial standards.
Mechanistic Insights into Pd-C Catalyzed Hydrogenation and Crystallization
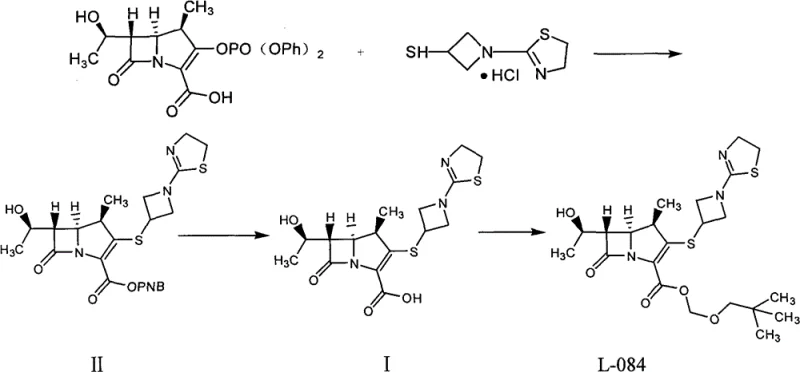
The core of this synthesis lies in the precise execution of the catalytic hydrogenation of Compound II, specifically the (1R, 5S, 6S)-6-[(1R)-hydroxyethyl]-1-methyl-2-[1-(2-thiazoline-2-yl)azetidine-3-yl]thio-1-carba-2-penem-3-carboxylic acid p-nitrobenzyl ester. The reaction employs 10 percent palladium on charcoal (Pd-C) as the heterogeneous catalyst within a mixed solvent system of THF and water. The presence of an alkali base, such as sodium bicarbonate or 2,6-lutidine, is critical to neutralize the carboxylic acid formed during hydrogenolysis, ensuring the stability of the sensitive beta-lactam ring. The reaction is typically conducted at mild temperatures ranging from 20 to 50 degrees Celsius under hydrogen pressures up to 5 MPa. Following the completion of hydrogenation, the removal of the catalyst via filtration is a crucial step to prevent metal contamination. The subsequent pH adjustment to 5.6 plus or minus 0.2 using dilute hydrochloric acid is the trigger for the zwitterionic form of tebipenem to precipitate or crystallize upon the addition of extra THF, leveraging the solubility differences in the mixed solvent system to isolate the product efficiently.
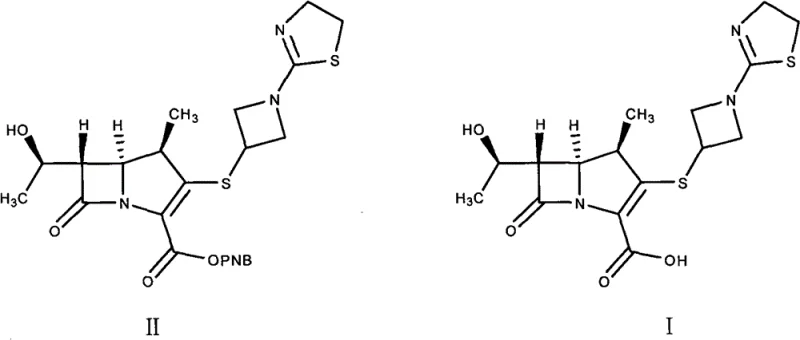
Impurity control is meticulously managed through the thermodynamics of the recrystallization process. The patent highlights that dissolving the crude product in hot water allows for the separation of insoluble mechanical impurities via hot filtration. Upon cooling the filtrate to temperatures between negative 15 and 30 degrees Celsius, the tebipenem crystallizes out in a highly ordered lattice, effectively excluding structurally related impurities. The data indicates that this aqueous recrystallization is capable of reducing the maximum single impurity content to less than 0.1 percent. This level of purity is paramount for a high-purity pharmaceutical intermediate, as it ensures that the subsequent esterification to form L-084 proceeds without the propagation of side products. The mechanism relies on the differential solubility of the target molecule versus its byproducts in water at varying temperatures, providing a robust and scalable purification tool that does not require complex chromatographic separations.
How to Synthesize Tebipenem Efficiently
The synthesis of tebipenem via this optimized route offers a clear pathway for laboratories and pilot plants to replicate the high yields and purity reported in the patent embodiments. The process is characterized by its operational simplicity, utilizing common reagents like THF, water, and standard hydrogenation catalysts. Operators must pay close attention to the pH control during the workup phase, as maintaining the pH at 5.6 is essential for maximizing recovery. The detailed standardized synthesis steps below outline the precise parameters for solvent ratios, temperature controls, and reaction times necessary to achieve the reported 85 to 88 percent yields with over 99.9 percent purity.
- Perform catalytic hydrogenation of Compound II using 10 percent palladium charcoal in a tetrahydrofuran and water mixture with an alkali base.
- Filter off insolubles and adjust the filtrate pH to 5.6 plus or minus 0.2 using hydrochloric acid solution.
- Add tetrahydrofuran to induce crystallization, filter the crude product, and recrystallize from hot pure water to obtain high-purity tebipenem.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible strategic benefits regarding cost structure and operational reliability. The shift away from massive acetone usage to a recyclable THF/water system significantly lowers the raw material expenditure associated with solvent procurement and disposal. Furthermore, the elimination of organic solvents in the recrystallization step simplifies the regulatory compliance burden related to residual solvent limits (ICH Q3C guidelines), thereby reducing the risk of batch rejection. This streamlined process enhances the overall throughput of the manufacturing facility, allowing for faster turnover times and more consistent supply availability for downstream API producers.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven primarily by the drastic reduction in solvent consumption and the simplification of the purification train. By replacing the 100:1 acetone precipitation with a THF crystallization and water recrystallization, the volume of waste solvent requiring treatment is minimized. Additionally, the higher yield (increasing from roughly 30 percent in old methods to over 85 percent in this method) means that less starting material is wasted per kilogram of final product. This efficiency gain directly lowers the cost of goods sold (COGS) and improves the margin profile for the commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the aqueous recrystallization step ensures a more predictable production schedule. Traditional methods prone to low yields and variable purity often result in reprocessing or batch failures, which disrupt supply continuity. With a method that consistently delivers purity above 99.9 percent, manufacturers can guarantee tighter delivery windows to their clients. The use of stable, non-hazardous reagents like water and THF also mitigates supply risks associated with the volatility of specialized organic solvents, ensuring that production can continue uninterrupted even during market fluctuations in chemical availability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is superior. The reduction in volatile organic compound (VOC) emissions due to lower acetone usage aligns with increasingly strict global environmental regulations. The ability to scale this process from laboratory benchtop to multi-ton production is facilitated by the use of standard hydrogenation reactors and crystallizers, without the need for exotic equipment. This scalability ensures that as demand for oral carbapenems grows, the supply chain can expand capacity rapidly without encountering the engineering bottlenecks typical of more complex synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tebipenem synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is critical for making informed decisions about technology transfer and process validation.
Q: What is the purity level achievable with this new tebipenem synthesis method?
A: The patented method achieves a crude product purity of over 99.8 percent. After recrystallization using only pure water, the final tebipenem purity exceeds 99.9 percent with a maximum single impurity content of less than 0.1 percent.
Q: How does this process reduce environmental impact compared to conventional methods?
A: Unlike conventional methods that require massive amounts of acetone for precipitation, this novel approach utilizes a tetrahydrofuran and water system for reaction and pure water for recrystallization. This eliminates the need for organic solvents in the refining stage, significantly reducing waste and resource consumption.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial suitability. It features simple operation, easy solvent recycling, and avoids complex purification steps, making it highly scalable for commercial manufacturing of carbapenem intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tebipenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent methodology to industrial reality is seamless. We are committed to delivering tebipenem and related carbapenem intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the water-based recrystallization technique described in CN102304129B allows us to offer a product that meets the highest international standards for impurity profiles.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, quality, and mutual growth.
