Advanced Catalytic Hydrogenation Strategy for Scalable Mirabegron Intermediate Production
Introduction to Next-Generation Mirabegron Synthesis
The global pharmaceutical landscape for treating Overactive Bladder (OAB) syndrome has been significantly transformed by the advent of beta-3 adrenergic receptor agonists, with Mirabegron standing out as a pivotal therapeutic agent. As demand for this active pharmaceutical ingredient surges across European, Japanese, and American markets, the imperative for developing robust, cost-effective, and environmentally sustainable manufacturing processes has never been more critical. Patent CN103387500A introduces a groundbreaking methodology that addresses the longstanding inefficiencies associated with traditional Mirabegron synthesis, specifically targeting the preparation of its key chiral intermediate. This intellectual property outlines a sophisticated yet operationally simple route that leverages heterogeneous catalysis to achieve high purity and yield, marking a substantial departure from the hazardous and complex protocols previously established in the industry.
The core innovation lies in the strategic replacement of aggressive reducing agents with a catalytic hydrogenation system that operates under remarkably mild conditions. By utilizing palladium on carbon (Pd/C) in an alcohol solvent environment, the process facilitates the selective reduction of the nitro group in Compound I to the corresponding amine, Compound II, without compromising the stereochemical integrity of the molecule. This approach not only enhances the safety profile of the manufacturing facility by eliminating pyrophoric reagents but also streamlines the downstream processing requirements. The subsequent acylation step further exemplifies green chemistry principles by employing water as the reaction medium, thereby minimizing the reliance on volatile organic solvents and reducing the overall environmental footprint of the production cycle.
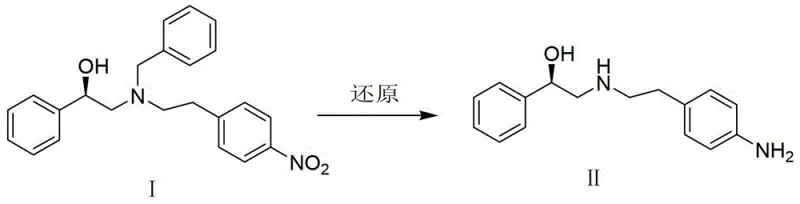
For procurement specialists and supply chain directors, the implications of this technological shift are profound. The transition to a hydrogenation-based protocol implies a decoupling from the volatile supply chains associated with specialized reducing agents like borane-THF complexes, which often require stringent storage and handling procedures. Instead, the reliance on hydrogen gas and recoverable heterogeneous catalysts offers a pathway to significant cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the mild reaction temperatures ranging from 10°C to 30°C suggest that existing standard reactor infrastructure can be utilized without the need for expensive cryogenic cooling systems or specialized high-pressure vessels beyond standard hydrogenation ratings, facilitating a smoother scale-up process from pilot plant to commercial tonnage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in earlier patents like CN100406011C, have historically relied on the use of borane-tetrahydrofuran (borane-THF) complexes for the critical reduction step in Mirabegron synthesis. This conventional approach presents a myriad of operational challenges that severely hinder its viability for large-scale industrial application. The use of borane necessitates strictly anhydrous conditions and a protective nitrogen atmosphere throughout the reaction, imposing rigorous demands on equipment integrity and operator training. Any breach in moisture control can lead to catastrophic decomposition of the reagent, posing significant safety hazards including fire and explosion risks, which in turn drives up insurance and facility maintenance costs exponentially.
Moreover, the workup procedures associated with borane reductions are notoriously cumbersome and waste-intensive. Quenching excess borane requires careful addition of protic solvents, often generating large volumes of boron-containing waste streams that are difficult and costly to treat in compliance with modern environmental regulations. The requirement for tetrahydrofuran (THF) as a solvent further exacerbates these issues, as THF is a volatile organic compound (VOC) that requires extensive recovery systems to prevent atmospheric release. Additionally, the sensitivity of the borane reagent often leads to variability in reaction outcomes, potentially impacting the consistency of the intermediate's purity and necessitating additional purification steps that erode overall process yield and throughput efficiency.
The Novel Approach
In stark contrast, the novel approach detailed in the subject patent revolutionizes the synthesis landscape by introducing a catalytic hydrogenation strategy that is inherently safer and more economically viable. By substituting the hazardous borane reagent with hydrogen gas and a solid Pd/C catalyst, the process eliminates the need for stringent anhydrous conditions and inert gas blanketing, thereby simplifying the operational protocol significantly. The reaction proceeds smoothly in common alcohol solvents such as methanol or ethanol, which are not only cheaper and more readily available than THF but also possess favorable toxicity profiles and are easier to recover and recycle through standard distillation techniques.
The robustness of this new method is further evidenced by its tolerance to a wide range of reaction parameters, including hydrogen pressures from 0.5MPa to 10MPa and temperatures between 15°C and 30°C. This flexibility allows process engineers to optimize the reaction kinetics based on available equipment capabilities without compromising product quality. The elimination of moisture-sensitive reagents also translates to a dramatic reduction in raw material costs and waste disposal fees. Furthermore, the subsequent acylation step performed in an aqueous medium represents a paradigm shift towards green manufacturing, effectively removing the need for chlorinated or aromatic solvents typically used in amide coupling reactions, thus aligning the production process with increasingly stringent global environmental compliance standards.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation
The mechanistic foundation of this improved synthesis rests on the principles of heterogeneous catalysis, where the palladium metal dispersed on the carbon support acts as a surface for the adsorption and activation of molecular hydrogen. In this system, hydrogen molecules dissociate upon contact with the palladium surface to form reactive atomic hydrogen species, which are then transferred to the nitro group of Compound I. This surface-mediated reduction is highly chemoselective, preferentially targeting the nitro functionality while leaving other sensitive groups, such as the chiral hydroxyl moiety and the benzyl ether linkage, intact. This selectivity is crucial for maintaining the optical purity of the intermediate, which is a critical quality attribute for the biological activity of the final Mirabegron API.
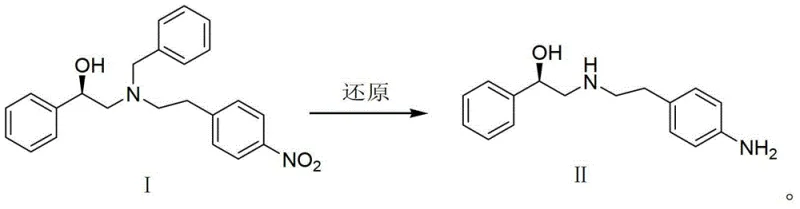
From an impurity control perspective, the use of Pd/C catalysis offers distinct advantages over stoichiometric chemical reductions. Traditional methods often generate side products resulting from over-reduction or incomplete reaction due to reagent degradation. In the catalytic system, the reaction progress can be precisely monitored via HPLC or TLC, allowing for termination exactly at the point of maximum conversion to the desired amine. The heterogeneous nature of the catalyst also facilitates its removal; a simple filtration step effectively separates the palladium carbon from the reaction mixture, preventing metal contamination in the final product. This ease of separation is a significant advantage for meeting the strict residual metal specifications required by pharmacopoeial standards for pharmaceutical intermediates, thereby reducing the burden on downstream purification units.
Furthermore, the aqueous acylation mechanism employed in the second step leverages the solubility characteristics of the reactants to drive the reaction forward. By adjusting the pH of the aqueous medium to a range of 1 to 7, preferably between 4 and 6, the amine group of Intermediate II is maintained in a state that balances nucleophilicity with solubility. The addition of carbodiimide coupling agents like EDC or DCC activates the carboxylic acid of the thiazole component, forming an reactive O-acylisourea intermediate that is rapidly attacked by the amine. Performing this in water minimizes the formation of organic-solvent-soluble impurities and simplifies the isolation of the final product through pH-adjusted precipitation, ensuring a high-purity profile suitable for direct crystallization.
How to Synthesize Mirabegron Intermediate Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize yield and purity while ensuring operational safety. The process begins with the suspension of Compound I in a lower alcohol solvent, followed by the addition of the palladium catalyst and the introduction of hydrogen gas under controlled pressure. Maintaining the temperature within the specified ambient range is critical to prevent thermal runaway while ensuring sufficient reaction kinetics. Once the reduction is complete, the catalyst is removed via filtration, and the solvent is evaporated under reduced pressure to yield the crude amine intermediate, which can be used directly in the next step or purified if necessary.
- Perform catalytic hydrogenation of Compound I using 10% Pd/C in an alcohol solvent under 0.5MPa to 10MPa hydrogen pressure at 15°C to 30°C.
- Filter the reaction mixture to remove the catalyst, concentrate the filtrate, and dry to obtain pure Intermediate II.
- Conduct acylation of Intermediate II with 2-amino-4-thiazoleacetic acid in water at pH 1-7 using EDC or DCC as a condensing agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers a compelling value proposition centered around risk mitigation and cost optimization. The shift away from specialized, hazardous reagents like borane-THF to commodity chemicals such as hydrogen, methanol, and palladium carbon fundamentally alters the cost structure of the manufacturing process. Hydrogen is abundantly available and inexpensive, while alcohol solvents are among the most cost-effective organic solvents in the chemical industry. This substitution results in a drastic reduction in raw material expenditure, freeing up capital for other strategic investments within the supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reducing agents directly translates to lower variable costs per kilogram of produced intermediate. Unlike borane complexes which are single-use and generate significant waste, the Pd/C catalyst can potentially be regenerated or recycled, further amortizing the cost of the precious metal. Additionally, the simplified workup procedures reduce labor hours and utility consumption associated with solvent recovery and waste treatment, leading to substantial overall cost savings in the production budget without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on globally available commodities like hydrogen and alcohols insulates the manufacturing process from the supply disruptions often associated with niche fine chemical reagents. The robustness of the reaction conditions means that production can be sustained across different geographical locations without the need for highly specialized infrastructure. This flexibility ensures a continuous supply of high-purity pharmaceutical intermediates, mitigating the risk of stockouts that could delay downstream API synthesis and final drug product availability in the market.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup make this process exceptionally scalable from kilogram to multi-ton scales. The reduction in VOC emissions and hazardous waste generation aligns perfectly with modern environmental, social, and governance (ESG) goals. Facilities adopting this route will face fewer regulatory hurdles and lower compliance costs related to waste disposal and air emissions, ensuring long-term operational sustainability and protecting the company's reputation as a responsible manufacturer in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Mirabegron synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear understanding of the practical benefits and operational requirements for potential licensees or manufacturing partners.
Q: What are the advantages of using Pd/C hydrogenation over borane reduction for Mirabegron synthesis?
A: The Pd/C hydrogenation method operates under mild conditions (10°C-30°C) in alcohol solvents, eliminating the need for hazardous anhydrous borane-THF complexes and strict nitrogen protection, thereby significantly reducing operational complexity and safety risks.
Q: How does the aqueous acylation step improve environmental compliance?
A: By utilizing water as the primary solvent for the acylation step instead of organic solvents, the process drastically reduces volatile organic compound (VOC) emissions and simplifies wastewater treatment protocols compared to traditional organic-phase couplings.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available reagents like 10% Pd/C and EDC, operates at near-ambient temperatures, and avoids cryogenic or high-vacuum conditions, making it highly robust and scalable for multi-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirabegron Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic hydrogenation route for Mirabegron production and are fully equipped to bring this technology to commercial fruition. Our state-of-the-art facilities boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards, providing you with a secure foundation for your API manufacturing operations.
We invite you to collaborate with us to leverage these advanced synthetic strategies for your portfolio. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener, more efficient route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to redefine the standards of efficiency and quality in pharmaceutical intermediate supply.
