Revolutionizing Quinazolinone Manufacturing: A Green One-Pot Catalytic Approach for Commercial Scale
Introduction to Advanced Quinazolinone Synthesis
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and environmentally benign pathways for constructing nitrogen-containing heterocycles, particularly the quinazolinone scaffold which is ubiquitous in bioactive molecules. Addressing these critical needs, the methodology disclosed in patent CN112645887B introduces a transformative approach to preparing quinazolinone derivatives. This innovation leverages a copper-catalyzed cascade reaction that operates effectively in an aqueous medium, marking a significant departure from traditional organic synthesis protocols that rely heavily on toxic solvents and harsh reagents. By utilizing readily available o-aminobenzonitriles and aldehydes as starting materials, this process achieves high atom economy and operational simplicity.
The core breakthrough lies in the ability to perform an in-situ hydrolysis of the nitrile group followed by cyclization in a single pot, thereby eliminating the need for isolating sensitive amide intermediates. This not only streamlines the synthetic route but also enhances the overall safety profile of the manufacturing process. For R&D teams focused on process intensification, this method offers a robust platform for generating diverse libraries of quinazolinone analogs with high purity profiles. The following reaction scheme illustrates the general transformation facilitated by this novel catalytic system.
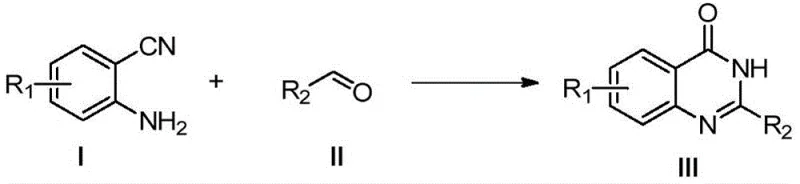
Furthermore, the compatibility of this method with a wide range of substituents ensures its versatility across different drug discovery programs. Whether targeting anticancer, anti-inflammatory, or antihypertensive agents, the ability to rapidly access these core structures with minimal environmental footprint is a strategic asset. As we delve deeper into the technical specifics, it becomes evident that this protocol represents a substantial upgrade over legacy manufacturing techniques, offering a compelling value proposition for both technical and commercial stakeholders in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone ring system has relied on classical condensation reactions involving anthranilic acid derivatives, acid chlorides, or anhydrides. These traditional pathways are fraught with significant logistical and environmental challenges that hinder their efficiency in modern manufacturing settings. A primary drawback is the frequent requirement for stoichiometric amounts of strong oxidants such as potassium permanganate, manganese dioxide, or hypervalent iodine reagents to drive the oxidative cyclization steps. The use of such reagents generates substantial quantities of heavy metal waste and toxic by-products, creating a severe burden on waste treatment facilities and escalating the overall cost of goods sold.
Moreover, conventional syntheses often necessitate the use of volatile organic solvents like dichloromethane, DMF, or toluene, which pose serious health risks to operators and require complex recovery systems to meet regulatory emission standards. The multi-step nature of many older routes, which often involve the separate preparation and purification of anthranilamide intermediates, further compounds these issues by increasing material handling time and reducing overall throughput. These inefficiencies result in lower cumulative yields and higher energy consumption, making traditional methods increasingly unsustainable in the face of tightening global environmental regulations and the industry's push towards green chemistry principles.
The Novel Approach
In stark contrast, the methodology described in the patent data utilizes a streamlined one-pot strategy that fundamentally reimagines the synthetic sequence. By employing o-aminobenzonitriles as the nitrogen source, the process bypasses the need for pre-formed amides, leveraging the nitrile group's susceptibility to catalytic hydrolysis under mild basic conditions. The integration of a copper chloride catalyst paired with a phenanthroline ligand enables this transformation to proceed smoothly in water, a solvent that is non-toxic, non-flammable, and economically negligible in cost. This shift to an aqueous system eliminates the hazards associated with organic solvents and simplifies the downstream processing significantly.
The reaction conditions are remarkably mild, typically operating at temperatures between 60°C and 80°C, which reduces energy expenditure compared to high-temperature reflux methods. The use of cesium carbonate as a base provides the necessary alkalinity to facilitate the hydrolysis without degrading sensitive functional groups on the aromatic rings. This gentle yet effective approach allows for the direct conversion of starting materials into the final quinazolinone-4(3H)-one derivatives with impressive efficiency. The elimination of isolation steps for intermediates not only saves time but also minimizes material loss, resulting in a cleaner reaction profile that is easier to control and scale up for commercial production requirements.
Mechanistic Insights into Copper-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring consistent quality in a production environment. The proposed mechanism initiates with the coordination of the copper(II) species to the nitrile nitrogen of the o-aminobenzonitrile substrate, activating the triple bond towards nucleophilic attack by water molecules present in the solvent. Under the influence of the cesium carbonate base, this hydration step proceeds to generate an o-aminobenzamide intermediate in situ. This transient species is highly reactive and does not require isolation, as it immediately engages in the subsequent condensation step with the aldehyde component present in the reaction mixture.
Following the formation of the imine intermediate through the condensation of the amine and aldehyde, the copper catalyst facilitates an intramolecular cyclization. This step involves the nucleophilic attack of the amide nitrogen onto the imine carbon, closing the heterocyclic ring to form the dihydroquinazolinone skeleton. Finally, an oxidative aromatization or tautomerization step, potentially mediated by the copper species or ambient oxygen, yields the stable quinazolinone product. The ligand, specifically 1,10-phenanthroline, plays a pivotal role in stabilizing the copper center and modulating its electronic properties, ensuring that the catalytic cycle turns over efficiently without precipitating inactive metal species.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction occurs in a homogeneous aqueous phase, side reactions typical of heterogeneous conditions are minimized. The specificity of the copper-ligand complex helps suppress the formation of polymeric by-products or over-oxidized species that often plague radical-based oxidations. Furthermore, the mild pH conditions prevent the hydrolysis of other sensitive esters or amides that might be present on the substrate, ensuring high chemoselectivity. This precise control over the reaction pathway translates directly into a cleaner crude product, reducing the burden on purification columns and enhancing the final purity of the API intermediate.
How to Synthesize Quinazolinone Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The process begins by charging the reactor with the o-aminobenzonitrile and the chosen aldehyde in a molar ratio ranging from 1:1 to 1:1.5, ensuring a slight excess of the aldehyde to drive the equilibrium forward. The catalyst system, comprising copper(II) chloride and 1,10-phenanthroline, is added at a loading of approximately 10 mol% each, while cesium carbonate is introduced as the base in equimolar amounts relative to the nitrile substrate. Water serves as the exclusive solvent, providing a green medium for the reaction to proceed.
- Dissolve o-aminobenzonitrile compounds and aldehydes in water solvent within a reaction vessel.
- Add copper chloride catalyst, 1,10-phenanthroline ligand, and cesium carbonate base to the mixture.
- Heat the reaction mixture to 60-80°C for 10-12 hours, then isolate and purify the crude product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous copper-catalyzed method presents a compelling opportunity to optimize the cost structure and reliability of the supply chain for quinazolinone intermediates. The most immediate impact is seen in the raw material costs; o-aminobenzonitriles and substituted benzaldehydes are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. Unlike specialized reagents required in traditional methods, these starting materials benefit from established, high-volume production networks that ensure price stability and consistent availability even during market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive organic solvents and the reduction in waste disposal fees. By switching to water, the facility avoids the capital and operational expenditures associated with solvent recovery distillation units and VOC abatement systems. Furthermore, the use of base metal copper instead of precious metals like palladium or rhodium drastically lowers the catalyst cost per kilogram of product. The simplified one-pot workflow also reduces labor hours and utility consumption, as there are fewer unit operations such as filtration, drying, and transfer of intermediates, leading to substantial overall cost savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply chain resilience. The tolerance of the reaction to various functional groups means that a single standardized protocol can be used to produce a wide array of derivatives, simplifying inventory management and production scheduling. The mild reaction conditions reduce the risk of thermal runaways or equipment corrosion, minimizing unplanned downtime and maintenance requirements. Additionally, the high yields reported across diverse substrates ensure that less starting material is needed to meet production targets, buffering the supply chain against potential shortages of key precursors and enabling more accurate forecasting of lead times for customers.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is straightforward due to the use of water as a heat transfer medium, which offers superior thermal capacity compared to organic solvents. This inherent safety feature facilitates easier temperature control in large reactors, reducing the engineering complexity required for scale-up. From a regulatory standpoint, the green nature of the process aligns perfectly with increasingly stringent environmental, health, and safety (EHS) mandates. The reduction in hazardous waste generation simplifies permitting processes and enhances the company's sustainability profile, which is becoming a critical factor in vendor selection for major pharmaceutical clients committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: What are the primary advantages of this aqueous synthesis method over traditional organic solvent methods?
A: The primary advantage is the elimination of volatile organic compounds (VOCs) by using water as the solvent, which significantly reduces environmental impact and waste disposal costs. Additionally, the one-pot procedure simplifies the workflow by avoiding the isolation of unstable intermediates like anthranilamide.
Q: Can this method tolerate diverse functional groups on the aldehyde substrate?
A: Yes, the protocol demonstrates excellent substrate scope, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl moieties without significant loss in yield.
Q: Is the copper catalyst system cost-effective for large-scale production?
A: Absolutely. The use of inexpensive copper chloride compared to precious metal catalysts like palladium, combined with the recyclability potential of the aqueous phase, offers substantial cost reductions suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in maintaining a competitive edge in the global pharmaceutical market. Our technical team has thoroughly evaluated the copper-catalyzed aqueous synthesis method and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of quinazolinone derivative delivered meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing capability can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need detailed technical dossiers including specific COA data and route feasibility assessments, we are prepared to provide comprehensive support. By collaborating with us, you gain access to a reliable supply of high-quality intermediates produced via sustainable and cost-effective technologies, positioning your organization for success in a rapidly evolving industry landscape.
