Streamlined Synthesis of Pyrrolidinylthio Carbapenem Derivatives for Industrial Scale-up
Introduction to Advanced Carbapenem Synthesis
The pharmaceutical industry continuously seeks more efficient pathways for producing broad-spectrum antibiotics, particularly within the carbapenem class which includes critical drugs like doripenem. Patent CN100582106C, published in January 2010, introduces a groundbreaking preparation method for pyrrolidinylthio carbapenem derivatives that addresses longstanding inefficiencies in side-chain functionalization. This technology focuses on the strategic introduction of a sulfamoylaminomethyl group at the 5-position of the pyrrolidine side chain, a structural motif essential for the biological activity of doripenem. Unlike traditional approaches that rely on cumbersome multi-step sequences involving heavy metal reagents, this invention leverages a direct Mitsunobu condensation reaction. This shift represents a significant technological leap, offering a robust framework for reliable pharmaceutical intermediate supplier operations aiming to optimize their manufacturing portfolios. The core innovation lies in reacting a pyrrolidinylthio carbapenem derivative bearing a hydroxymethyl group directly with a sulfonamide derivative, thereby streamlining the entire synthetic architecture.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies, such as those disclosed in US Patent 5317016 and Chinese Patent CN1071428A, illustrate the historical complexity associated with synthesizing these critical antibiotic intermediates. The conventional route typically necessitates a minimum of four distinct reaction steps merely to construct the requisite side chain functionality before it can even be coupled to the carbapenem nucleus. This legacy process relies heavily on specialized and often hazardous reagents, including sodium triphenylmethanethiolate, potassium phthalimide, and p-methoxybenzyloxycarbonylsulfonyl chloride. Furthermore, the removal of protecting groups in these older methods frequently requires the use of silver nitrate and hydrogen sulfide, introducing significant environmental and safety burdens. Perhaps most critically for industrial operations, these multi-step sequences often mandate purification via macroporous adsorption resin chromatography or column chromatography, which severely limits throughput and increases production costs. 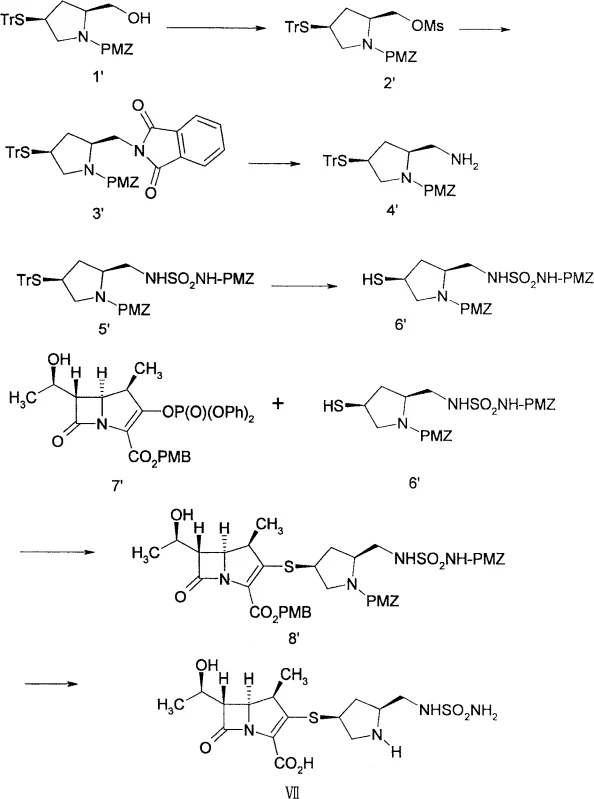
The Novel Approach
In stark contrast, the novel approach detailed in CN100582106C fundamentally reimagines the synthetic strategy by condensing a pre-formed carbapenem nucleus with a pyrrolidine derivative followed by a direct Mitsunobu coupling. This method bypasses the need for the intricate phthalimide protection-deprotection cycle entirely. Instead, it utilizes a pyrrolidinylthio carbapenem derivative with a free hydroxymethyl group at the 5-position, which reacts efficiently with sulfonamide derivatives in the presence of a trivalent phosphorus compound and an azodicarboxylic acid derivative. This consolidation of steps not only accelerates the timeline for cost reduction in pharmaceutical intermediate manufacturing but also drastically improves the impurity profile by avoiding heavy metal contaminants like silver. The resulting process is far more amenable to large-scale operations, replacing difficult chromatographic separations with simpler crystallization and filtration techniques. 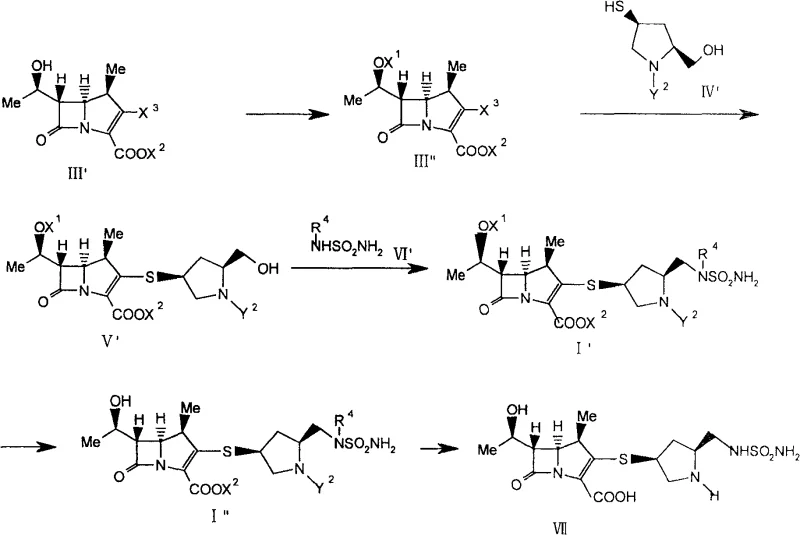
Mechanistic Insights into Mitsunobu-Mediated Side Chain Functionalization
The heart of this technological advancement is the application of the Mitsunobu reaction to install the sulfamoylaminomethyl moiety. Mechanistically, this transformation involves the activation of the primary alcohol on the pyrrolidine side chain through the formation of an alkoxyphosphonium salt intermediate, generated in situ from the reaction between the alcohol, a trivalent phosphorus compound such as triphenylphosphine, and an azodicarboxylate like diisopropyl azodicarboxylate (DIAD). This activated species is then susceptible to nucleophilic attack by the nitrogen of the sulfonamide derivative. The reaction proceeds with high efficiency under mild conditions, typically ranging from 0°C to 40°C, which is crucial for preserving the sensitive beta-lactam ring of the carbapenem core. The choice of solvent plays a pivotal role, with tetrahydrofuran, dichloromethane, and ethyl acetate identified as optimal media to balance solubility and reaction kinetics while maintaining anhydrous conditions to prevent hydrolysis of the activated intermediates.
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic substitution methods that might require harsh leaving groups or basic conditions detrimental to the beta-lactam stability. By avoiding the use of silver salts for deprotection, the process eliminates the risk of heavy metal residues, a critical quality attribute for API intermediates. Furthermore, the stereocenters on the carbapenem nucleus and the pyrrolidine ring remain unaffected during this transformation, ensuring the preservation of the desired (4R, 5S, 6S) and (3S, 5S) configurations essential for antibacterial potency. The ability to tune the protecting groups on the nitrogen atoms (such as PNZ, BOC, or Alloc) allows for flexible downstream processing, facilitating the final deprotection steps to yield the active pharmaceutical ingredient with high purity specifications. 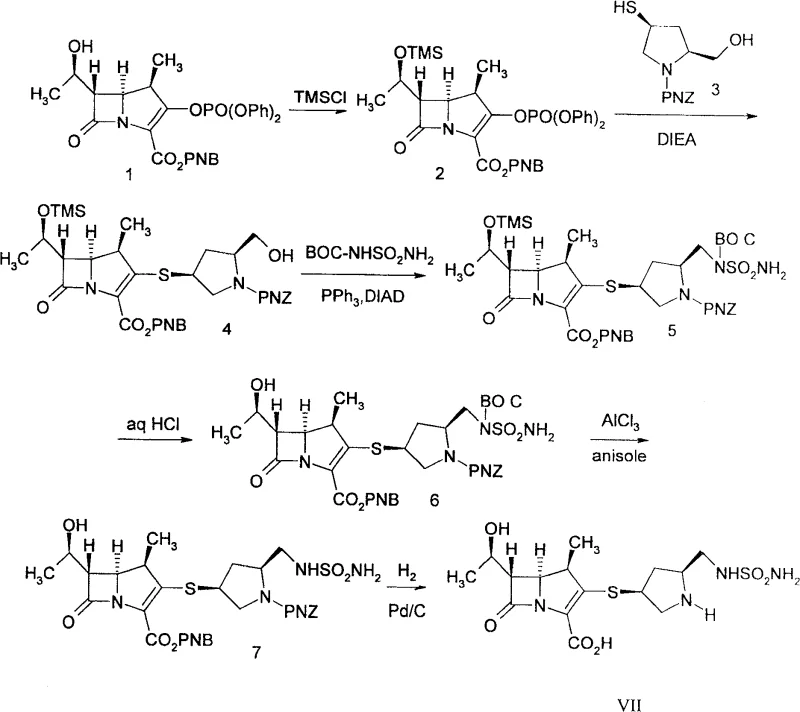
How to Synthesize Doripenem Intermediate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and minimize degradation of the sensitive beta-lactam scaffold. The process begins with the protection of the hydroxyl group on the carbapenem nucleus, typically using silyl protecting groups like trimethylsilyl (TMS) or tert-butyldimethylsilyl (TBDMS), followed by coupling with the pyrrolidine thiol. Once the key thioether linkage is established, the Mitsunobu reaction is executed under inert atmosphere to introduce the sulfonamide side chain. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that ensure reproducibility.
- Protect the hydroxyl group of the carbapenem nucleus using silyl protecting groups like TMS or TBDMS to ensure stability during subsequent coupling.
- Condense the protected carbapenem nucleus with a pyrrolidine thiol derivative to form the key thioether linkage at the C3 position.
- Perform a Mitsunobu reaction between the hydroxymethyl side chain of the pyrrolidine ring and a sulfonamide derivative to introduce the sulfamoylaminomethyl group directly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational improvements and risk mitigation. The elimination of expensive and hazardous reagents such as silver nitrate and potassium phthalimide directly impacts the bill of materials, leading to substantial cost savings without compromising quality. Moreover, the removal of multi-step column chromatography purification significantly reduces solvent consumption and waste generation, aligning with modern green chemistry initiatives and reducing the environmental footprint of the manufacturing process. This streamlined workflow enhances supply chain reliability by shortening the overall production cycle time and reducing the dependency on specialized purification equipment that often creates bottlenecks in facility utilization.
- Cost Reduction in Manufacturing: The novel process achieves cost optimization primarily by consolidating multiple synthetic steps into fewer, higher-yielding transformations. By eliminating the need for silver-mediated deprotection and phthalimide chemistry, the method removes some of the most cost-intensive reagents from the supply chain. Additionally, the shift from chromatographic purification to crystallization-based isolation reduces the consumption of large volumes of organic solvents and silica gel, which are significant cost drivers in fine chemical production. These efficiencies collectively lower the cost of goods sold (COGS), making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The reagents required for this Mitsunobu-based approach, such as triphenylphosphine and DIAD, are commodity chemicals with robust global supply chains, unlike some of the specialized reagents used in conventional methods. This availability ensures consistent raw material sourcing and reduces the risk of production delays due to supply shortages. Furthermore, the simplified process flow reduces the number of intermediate handlings and storage requirements, minimizing the potential for quality deviations and ensuring a more predictable delivery schedule for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scalability is inherently improved by designing a process that avoids complex chromatographic separations, which are difficult to scale beyond pilot plant levels. The reliance on crystallization and filtration allows for seamless transition from kilogram to ton-scale production using standard reactor infrastructure. From an environmental compliance standpoint, the reduction in heavy metal usage and solvent waste simplifies effluent treatment processes, facilitating easier regulatory approval and adherence to strict environmental, health, and safety (EHS) standards required by international regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for R&D teams planning technology transfer and for procurement teams assessing vendor capabilities.
Q: How does the Mitsunobu reaction improve the synthesis of doripenem intermediates?
A: The Mitsunobu reaction allows for the direct coupling of the hydroxymethyl-pyrrolidine derivative with sulfonamide derivatives in a single step. This eliminates the need for multiple protection and deprotection steps involving expensive reagents like silver nitrate and phthalimide, significantly simplifying the process and reducing impurity profiles.
Q: What are the key advantages of this new process over conventional methods?
A: Conventional methods require at least four reaction steps to prepare the side chain, utilizing hazardous and costly reagents such as potassium phthalimide and silver nitrate. The new process described in CN100582106C reduces the step count, avoids column chromatography purification by enabling crystallization, and uses readily available reagents like triphenylphosphine and DIAD.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. By removing multi-step column chromatography and replacing it with crystallization and filtration, the method enhances throughput and safety. The use of standard solvents like tetrahydrofuran and ethyl acetate further facilitates easy scale-up in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doripenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex antibiotic intermediates like those described in CN100582106C. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to monitor every stage of the synthesis. Our expertise in carbapenem chemistry allows us to navigate the complexities of beta-lactam stability and stereochemical control effectively.
We invite you to collaborate with us to optimize your supply chain for carbapenem antibiotics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us help you secure a stable and cost-effective supply of high-quality intermediates for your pharmaceutical formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
