Revolutionizing Antiviral Intermediate Production: Scalable Supercritical Synthesis of 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane
The pharmaceutical industry's relentless pursuit of efficient antiviral therapies has placed significant demand on the supply chain for critical bicyclic amine intermediates. Patent CN114702431B introduces a groundbreaking preparation method for 6, 6-dimethyl-3-azabicyclo [3.1.0] hexane, a pivotal building block for hepatitis C protease inhibitors like Boceprevir and novel coronavirus treatments such as PF-07321332. This technology leverages the unique physicochemical properties of supercritical fluids to overcome the limitations of traditional batch synthesis. By transitioning from solvent-heavy conventional routes to a streamlined supercritical ammonia process, manufacturers can achieve drastic improvements in reaction kinetics and overall material throughput. The following analysis details how this innovation addresses the triad of purity, cost, and scalability for global procurement leaders.
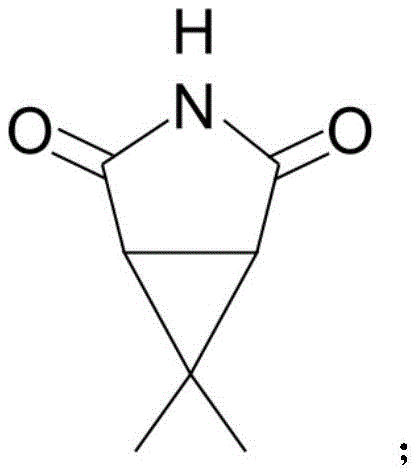
![Chemical structure of the final product 6,6-dimethyl-3-azabicyclo[3.1.0]hexane](/insights/img/6-6-dimethyl-3-azabicyclo-hexane-supercritical-synthesis-pharma-supplier-20260305223028-02.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6, 6-dimethyl-3-azabicyclo [3.1.0] hexane has relied on multi-step sequences involving harsh reagents and inefficient solvent systems. Prior art, such as that described in WO2008082508 and WO2007075790, typically employs lithium aluminum hydride for reduction followed by hydrogenation to remove benzyl protecting groups, or utilizes hazardous HCl gas at cryogenic temperatures (-23 to -20°C) to form salts. These legacy methods suffer from inherent drawbacks, including the generation of substantial chemical waste due to excessive solvent usage and the safety risks associated with handling pyrophoric reagents and corrosive gases. Furthermore, the overall yields in these conventional pathways are often suboptimal, frequently failing to exceed 78% in the final reduction steps due to side reactions and incomplete conversions. The reliance on ambient or cryogenic conditions often results in sluggish reaction rates, necessitating longer processing times and larger reactor footprints, which directly inflates the cost of goods sold (COGS) for the final API intermediate.
The Novel Approach
The patented methodology fundamentally reengineers the synthetic route by introducing supercritical fluid technology at two critical junctures: anhydride formation and ammonolysis. Instead of traditional thermal dehydration, the process utilizes supercritical carbon dioxide to facilitate the conversion of cis-caronic dicarboxylic acid to caronic anhydride, achieving yields upwards of 87%. More critically, the subsequent ammonolysis step replaces liquid ammonia or ammonia-alcohol mixtures with supercritical ammonia maintained at 140-165°C and 5-25 MPa. In this supercritical state, ammonia exhibits gas-like diffusivity and liquid-like density, allowing it to penetrate the reactant matrix with exceptional efficiency without the need for auxiliary organic solvents. This solvent-free environment not only aligns with green chemistry principles by eliminating VOC emissions but also drives the equilibrium towards the formation of the imide intermediate (Compound A) with remarkable yields of 91-92%, significantly outperforming the 70% yield observed in non-supercritical comparative examples.
Mechanistic Insights into Supercritical Ammonolysis and Reduction
The core mechanistic advantage of this process lies in the manipulation of phase behavior to enhance nucleophilic attack. In the supercritical ammonolysis step, the caronic anhydride is subjected to ammonia under conditions where the distinction between liquid and gas phases disappears. This state creates a homogeneous reaction medium with zero surface tension, allowing ammonia molecules to access the electrophilic carbonyl carbons of the anhydride with minimal steric hindrance. The high temperature (140-165°C) provides the necessary activation energy to overcome the ring strain of the bicyclic system, facilitating the rapid opening of the anhydride ring and subsequent cyclization to form the stable imide structure of Compound A. The absence of competing solvent molecules ensures that the concentration of the nucleophile (ammonia) remains effectively high throughout the reaction volume, thereby suppressing hydrolysis side reactions that typically plague aqueous or alcoholic ammonolysis processes. This precise control over the reaction environment is the primary driver for the observed purity enhancements and yield maximization.
Following the formation of the imide intermediate, the process proceeds to a robust reduction phase using lithium aluminum hydride (LiAlH4) in tetrahydrofuran. Mechanistically, the hydride ions attack the carbonyl groups of the imide, sequentially reducing them to methylene groups to generate the target tertiary amine. The protocol specifies a controlled addition at 0°C followed by heating to 40-50°C, a thermal profile designed to manage the exothermic nature of the reduction while ensuring complete conversion of the recalcitrant amide bonds. The subsequent quenching with dilute sodium hydroxide and careful phase separation allows for the efficient removal of aluminum salts, resulting in a light yellow liquid that requires only vacuum distillation for final purification. This streamlined workup avoids the need for column chromatography, a critical factor for maintaining cost-efficiency and throughput in a commercial manufacturing setting.
How to Synthesize 6, 6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
The implementation of this synthesis route requires precise control over high-pressure parameters to fully realize the benefits of supercritical fluid dynamics. Operators must ensure that the reactor systems are rated for the specified pressures of up to 25 MPa and temperatures exceeding 150°C to maintain the ammonia in its supercritical state. The process begins with the dehydration of the dicarboxylic acid precursor, followed by the critical high-pressure ammonolysis step which dictates the overall success of the campaign. Detailed standard operating procedures regarding pressure ramping, dwell times, and safe depressurization protocols are essential for reproducibility. For a comprehensive breakdown of the specific operational parameters and safety guidelines required for execution, please refer to the standardized synthesis guide below.
- Preparation of Caronic Anhydride: React cis-caronic dicarboxylic acid with acetic anhydride in a supercritical carbon dioxide reactor at 35-40°C and 10-15 MPa.
- Supercritical Ammonolysis: React caronic anhydride with ammonia in a high-pressure kettle at 140-165°C and 5-25 MPa to form the imide intermediate (Compound A).
- Reduction and Purification: Reduce Compound A using lithium aluminum hydride in tetrahydrofuran, followed by quenching, extraction, and vacuum distillation to obtain the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this supercritical synthesis route offers tangible strategic benefits beyond mere technical novelty. The elimination of organic solvents in the ammonolysis step represents a direct reduction in raw material procurement costs and, more significantly, a drastic decrease in waste disposal expenses associated with solvent recovery and treatment. By simplifying the reaction mixture to essentially reactants and products, the downstream purification burden is lightened, leading to shorter cycle times and increased asset utilization rates. This efficiency gain translates into a more resilient supply chain capable of responding rapidly to fluctuations in demand for antiviral medications without the bottlenecks typically caused by complex purification trains.
- Cost Reduction in Manufacturing: The transition to a solvent-free supercritical ammonia process removes the financial burden of purchasing, storing, and disposing of large volumes of organic solvents. Furthermore, the substantial increase in reaction yield—from approximately 70% in conventional methods to over 90% in this patented route—means that less starting material is required to produce the same amount of final product. This improvement in atom economy directly lowers the variable cost per kilogram of the intermediate, providing a competitive pricing advantage in the global market for pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: The robustness of the supercritical method reduces the risk of batch failures caused by incomplete reactions or difficult-to-remove impurities. Since the process avoids the use of sensitive reagents like HCl gas at cryogenic temperatures, the operational complexity is lowered, minimizing the potential for human error or equipment malfunction. This reliability ensures a consistent flow of high-quality intermediate to downstream API manufacturers, securing the continuity of supply for critical antiviral drug production lines.
- Scalability and Environmental Compliance: High-pressure reactor technology is well-established in the fine chemical industry, allowing for straightforward scale-up from pilot to commercial production scales without fundamental changes to the chemistry. Additionally, the "green" nature of using supercritical CO2 and ammonia aligns with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste generation. This compliance future-proofs the manufacturing process against regulatory tightening, avoiding potential shutdowns or fines that could disrupt supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this supercritical synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is crucial for R&D teams assessing technology transfer and procurement teams negotiating supply agreements.
Q: What are the advantages of using supercritical ammonia over traditional ammonolysis?
A: Supercritical ammonia eliminates the need for additional organic solvents, significantly improving the ammonolysis effect and reaction yield while simplifying downstream purification and reducing environmental waste.
Q: What is the typical yield improvement compared to conventional methods?
A: The patented supercritical method achieves yields of approximately 91-92% for the intermediate step, compared to roughly 70% in conventional room-temperature processes, representing a substantial increase in material efficiency.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard high-pressure reactor technology and avoids complex chromatographic separations, making it highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent CN114702431B can be reliably replicated on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex bicyclic amines, guaranteeing that every batch of 6, 6-dimethyl-3-azabicyclo [3.1.0] hexane meets the exacting standards required for GMP pharmaceutical manufacturing.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this solvent-free methodology for your specific volume requirements. Contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines.
