Revolutionizing TAF Intermediate Production: A Deep Dive into Crown Ether Catalysis
Revolutionizing TAF Intermediate Production: A Deep Dive into Crown Ether Catalysis
The global demand for potent antiretroviral therapies continues to drive innovation in the synthesis of nucleotide analogs, particularly for the treatment of HIV and Hepatitis B. At the forefront of this chemical landscape is Tenofovir Alafenamide (TAF), a prodrug that offers improved plasma stability and intracellular delivery compared to its predecessors. 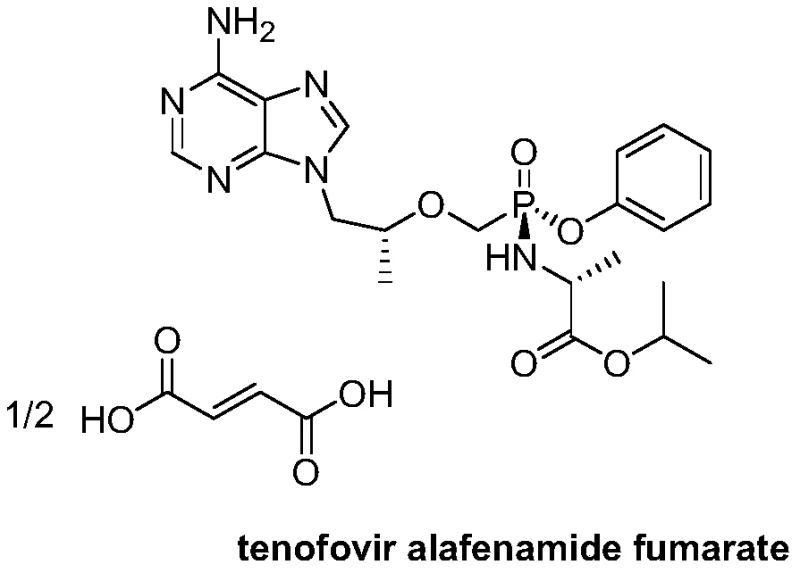 Central to the efficient manufacturing of this critical medication is the availability of high-purity key intermediates, specifically (R)-9-(2-phenoxyphosphomethoxypropyl)adenine. Recent intellectual property developments, notably detailed in patent CN107098936B, have unveiled a transformative preparation method that addresses long-standing bottlenecks in yield, purity, and environmental impact. This technical insight report analyzes the strategic advantages of this novel crown ether-catalyzed hydrolysis pathway, providing R&D directors and supply chain leaders with a clear roadmap for optimizing their antiviral intermediate sourcing strategies.
Central to the efficient manufacturing of this critical medication is the availability of high-purity key intermediates, specifically (R)-9-(2-phenoxyphosphomethoxypropyl)adenine. Recent intellectual property developments, notably detailed in patent CN107098936B, have unveiled a transformative preparation method that addresses long-standing bottlenecks in yield, purity, and environmental impact. This technical insight report analyzes the strategic advantages of this novel crown ether-catalyzed hydrolysis pathway, providing R&D directors and supply chain leaders with a clear roadmap for optimizing their antiviral intermediate sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical mono-phenyl ester intermediate has been plagued by inefficient coupling strategies and cumbersome protection group manipulations. One prevalent prior art method involves the direct coupling of tenofovir with phenol using DCC (dicyclohexylcarbodiimide) as a dehydrating agent in solvents like NMP. 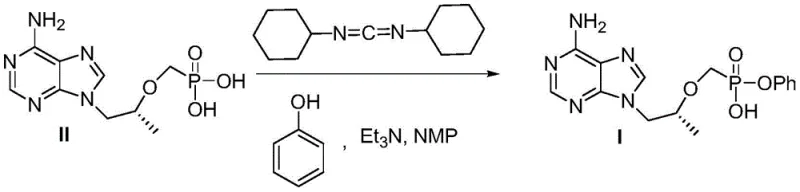 While chemically feasible, this approach generates stoichiometric amounts of dicyclohexylurea, a solid by-product that is exceptionally difficult to separate from the desired product, often necessitating extensive chromatographic purification that drastically reduces overall throughput. Furthermore, alternative routes utilizing triphenyl phosphite introduce significant environmental liabilities due to the generation of phosphorus-containing wastewater, while Boc-protection strategies extend the synthetic timeline by requiring additional steps for amine protection and subsequent high-pressure hydrolysis to remove the protecting group.
While chemically feasible, this approach generates stoichiometric amounts of dicyclohexylurea, a solid by-product that is exceptionally difficult to separate from the desired product, often necessitating extensive chromatographic purification that drastically reduces overall throughput. Furthermore, alternative routes utilizing triphenyl phosphite introduce significant environmental liabilities due to the generation of phosphorus-containing wastewater, while Boc-protection strategies extend the synthetic timeline by requiring additional steps for amine protection and subsequent high-pressure hydrolysis to remove the protecting group.
The Novel Approach
In stark contrast to these legacy methodologies, the invention disclosed in CN107098936B introduces a highly elegant and direct hydrolysis strategy. This novel approach bypasses the need for coupling reagents or protecting groups entirely by starting from the readily accessible diphenyl ester precursor (Compound II). 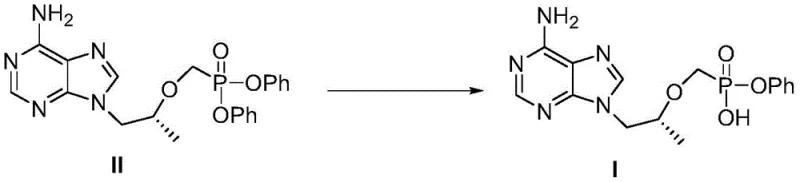 By employing a phase transfer catalysis system comprising an alkali metal hydroxide and a specific crown ether catalyst, the process achieves selective cleavage of one phenyl ester bond under remarkably mild conditions. This shift from constructive coupling to selective deconstruction not only simplifies the reaction workflow but also fundamentally alters the impurity profile, allowing for purification via simple crystallization rather than resource-intensive chromatography, thereby representing a paradigm shift in process chemistry for this class of nucleotides.
By employing a phase transfer catalysis system comprising an alkali metal hydroxide and a specific crown ether catalyst, the process achieves selective cleavage of one phenyl ester bond under remarkably mild conditions. This shift from constructive coupling to selective deconstruction not only simplifies the reaction workflow but also fundamentally alters the impurity profile, allowing for purification via simple crystallization rather than resource-intensive chromatography, thereby representing a paradigm shift in process chemistry for this class of nucleotides.
Mechanistic Insights into Crown Ether-Catalyzed Selective Hydrolysis
The efficacy of this novel synthesis lies in the sophisticated application of phase transfer catalysis (PTC) to control regioselectivity during ester hydrolysis. In a typical biphasic system involving an organic solvent (such as tetrahydrofuran or 1,4-dioxane) and an aqueous base, the hydroxide ions are largely confined to the aqueous phase, limiting their reactivity with the organic-soluble diphenyl ester substrate. The introduction of a crown ether, such as 18-crown-6, 15-crown-5, or 12-crown-4, acts as a molecular host that selectively complexes with the alkali metal cation (K+, Na+, or Li+). This complexation effectively solubilizes the hydroxide anion into the organic phase as a naked, highly nucleophilic species. This enhanced nucleophilicity allows for the precise attack on the phosphorus center, facilitating the cleavage of the P-O-Ph bond. Crucially, the steric and electronic environment created by the crown ether-cation complex helps moderate the reactivity, preventing over-hydrolysis to the free phosphonic acid and ensuring the reaction stops selectively at the mono-phenyl ester stage, which is the desired intermediate.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over carbodiimide-mediated couplings. Traditional methods often suffer from side reactions such as N-acylurea formation or racemization at the chiral center adjacent to the phosphate group due to harsh activation conditions. The mild basic conditions (0-80°C, preferably 20-60°C) employed in the crown ether method preserve the stereochemical integrity of the (R)-configuration at the propyl chain. Furthermore, because the reaction does not generate insoluble urea by-products or toxic phosphine oxides, the crude reaction mixture is significantly cleaner. The final product can be isolated by simply adjusting the pH of the aqueous phase to precipitate the target compound, leaving soluble inorganic salts and excess phenol in the mother liquor, thus achieving high purity specifications essential for downstream API synthesis without the need for silica gel column chromatography.
How to Synthesize (R)-9-(2-phenoxyphosphomethoxypropyl)adenine Efficiently
The operational simplicity of this patented process makes it highly attractive for technology transfer and scale-up. The procedure generally involves dissolving the diphenyl ester precursor in a water-miscible organic solvent, followed by the addition of an aqueous hydroxide solution and a catalytic loading of the appropriate crown ether. The reaction proceeds smoothly at ambient or slightly elevated temperatures, monitored conveniently by TLC or HPLC. Upon completion, the workup is straightforward: extraction to remove organic impurities, followed by acidification to precipitate the product. For a comprehensive, step-by-step breakdown of the exact molar ratios, solvent volumes, and specific workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve the diphenyl ester precursor (Compound II) in a suitable organic solvent such as tetrahydrofuran, 1,4-dioxane, or methanol.
- Add an aqueous solution of an alkali metal hydroxide (LiOH, NaOH, or KOH) along with a catalytic amount of a crown ether (e.g., 18-crown-6) to the reaction mixture.
- Stir the mixture at a mild temperature between 20°C and 60°C until completion, then acidify the aqueous phase to precipitate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this crown ether-catalyzed route translates directly into tangible operational efficiencies and risk mitigation. The elimination of expensive and hazardous coupling reagents like DCC or toxic phosphites removes a significant variable cost from the bill of materials. Moreover, the avoidance of complex purification steps such as column chromatography drastically reduces solvent consumption and processing time, leading to a leaner manufacturing footprint. The ability to purify the intermediate via crystallization not only lowers waste disposal costs associated with silica gel and eluent mixtures but also ensures a more consistent and robust supply of material that meets rigorous quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the reduction of auxiliary material costs. By removing the need for stoichiometric amounts of coupling agents and the subsequent removal of their by-products, the process significantly lowers the raw material intensity per kilogram of output. Additionally, the use of common, commodity-grade bases like potassium hydroxide and recyclable crown ether catalysts replaces specialized, high-cost reagents. The simplified workup, which relies on precipitation and filtration rather than energy-intensive distillation or chromatography, further reduces utility consumption and labor hours, resulting in substantial overall cost savings for the production of this high-value pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely available starting materials and reagents that are not subject to the same regulatory restrictions or supply volatility as specialized coupling agents or protected amino acid derivatives. The robustness of the reaction conditions—operating effectively at atmospheric pressure and moderate temperatures—means that production is not dependent on specialized high-pressure reactors or cryogenic cooling systems, allowing for manufacturing across a broader range of facility types. This flexibility reduces the risk of production bottlenecks and ensures that suppliers can maintain consistent lead times even during periods of high market demand for antiviral medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology represents a significant advancement towards green chemistry principles. The process avoids the generation of large volumes of solid urea waste or toxic phosphorus-containing effluents, which are major compliance hurdles in traditional synthesis. The primary waste streams consist of aqueous salts and recovered solvents, which are far easier to treat and manage. This reduced environmental burden simplifies the permitting process for scale-up and aligns with the increasingly stringent sustainability goals of global pharmaceutical companies, making it a future-proof choice for long-term commercial manufacturing.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their supply chains, we have compiled answers to common inquiries regarding the process capabilities and quality attributes. These responses are derived directly from the experimental data and comparative analysis provided in the patent literature, addressing key concerns about yield, purity, and operational safety.
Q: Why is the traditional DCC coupling method problematic for industrial scale-up?
A: Traditional methods utilizing DCC (dicyclohexylcarbodiimide) generate substantial amounts of dicyclohexylurea by-products which are notoriously difficult to remove, leading to complex purification processes and significant solid waste generation that hinders large-scale manufacturing.
Q: How does the crown ether catalytic method improve upon Boc-protection strategies?
A: Unlike Boc-protection routes that require additional synthetic steps for protection and deprotection, often involving high-pressure equipment for ammonia hydrolysis, the crown ether method allows for direct, selective hydrolysis under atmospheric pressure and mild temperatures, significantly shortening the production cycle.
Q: What are the purity advantages of this new hydrolysis technique?
A: The novel process facilitates high-purity isolation through simple crystallization rather than complex chromatography, effectively minimizing impurities and ensuring the intermediate meets stringent specifications required for antiviral API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-9-(2-phenoxyphosphomethoxypropyl)adenine Supplier
As the pharmaceutical industry continues to prioritize efficient and sustainable manufacturing pathways for critical antiviral therapies, the ability to execute complex chemistries like crown ether-catalyzed hydrolysis at scale becomes a key differentiator. NINGBO INNO PHARMCHEM stands ready to support your development and commercial needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and crystallization protocols required for this intermediate, ensuring that we deliver material with stringent purity specifications and rigorous QC labs verification to support your regulatory filings.
We invite you to engage with our technical team to discuss how this advanced synthesis route can optimize your cost structure and supply security. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic potential of switching to this greener, more efficient methodology. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more reliable supply of high-quality TAF intermediates.
