Scalable Synthesis of Cefepime Hydrochloride: Advanced 7-ACA Route for Global Pharma Supply
The pharmaceutical landscape for fourth-generation cephalosporins demands robust, scalable, and high-purity synthesis routes to ensure consistent supply of critical antibiotics. Patent CN101337971A introduces a refined methodology for synthesizing Cefepime Hydrochloride, addressing the inherent instability of the free base form by converting it into a stable hydrochloride salt suitable for injection. This technical breakthrough leverages 7-aminocephalosporanic acid (7-ACA) as the foundational starting material, utilizing a sophisticated silylation protection strategy followed by a streamlined one-pot quaternization to generate the key intermediate, 7-MPCA. The structural integrity and chemical complexity of the final active pharmaceutical ingredient are depicted below, highlighting the specific stereochemistry required for broad-spectrum efficacy against gram-positive and gram-negative microorganisms.
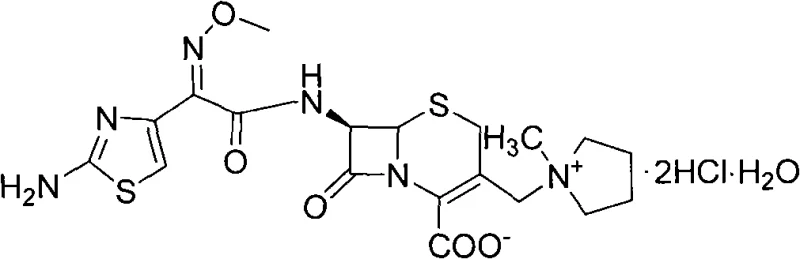
As a reliable cefepime intermediate supplier, understanding the transition from unstable free bases to stable salt forms is paramount for maintaining shelf-life and potency in global supply chains. The invention specifically targets the resolution of prior art limitations where product quality was inconsistent and storage stability was compromised. By optimizing the reaction conditions—specifically the interplay between hexamethyldisilazane (HMDS), iodotrimethylsilane (TMSI), and N-methylpyrrolidine—the process achieves a high-yield conversion that is economically viable for commercial scale-up of complex antibiotic intermediates. This approach not only enhances the chemical stability of the molecule but also simplifies the downstream purification requirements, offering a distinct competitive edge in the manufacturing of high-purity cefepime hydrochloride.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fourth-generation cephalosporins like Cefepime has been plagued by significant technical hurdles that impede efficient cost reduction in antibiotic manufacturing. Traditional routes often rely on starting materials such as GCLE (7-phenylacetamide-3-chloromethyl-4-cephalosporanic acid p-methoxybenzyl ester), which are not only expensive but also difficult to source in bulk quantities with consistent quality. Furthermore, conventional methodologies frequently involve harsh reaction conditions, including extreme temperatures and prolonged reaction times, which degrade the sensitive beta-lactam ring and lead to the formation of complex impurity profiles. These factors contribute to low overall yields and necessitate extensive, costly purification steps to meet regulatory standards for injectable products. The difficulty in controlling the quaternization step at the C-3 position often results in variable product quality, making continuous large-scale industrial production challenging and risky for supply chain managers.
The Novel Approach
In stark contrast, the novel approach detailed in CN101337971A revolutionizes the synthetic pathway by employing a direct, one-pot transformation of protected 7-ACA into the quaternary ammonium intermediate, 7-MPCA. This method eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and operational time significantly. The process utilizes readily available reagents like HMDS and TMSI to protect the amino and carboxyl groups of 7-ACA in situ, creating a reactive environment where N-methylpyrrolidine can efficiently introduce the requisite quaternary ammonium side chain. This streamlined sequence ensures that the reaction proceeds under milder conditions, preserving the integrity of the cephalosporin nucleus while maximizing yield. The subsequent acylation with AE active ester is conducted in a mixed solvent system that facilitates easy separation of byproducts, ultimately delivering a product with superior stability and purity compared to legacy methods.
Mechanistic Insights into Silylation Protection and Quaternization
The core chemical innovation lies in the precise orchestration of silylating agents to manipulate the reactivity of the 7-ACA nucleus. Initially, 7-ACA reacts with hexamethyldisilazane (HMDS) and a catalytic amount of iodotrimethylsilane (TMSI) under reflux conditions. This step serves a dual purpose: it protects the nucleophilic amino group at the C-7 position and the carboxylic acid at the C-4 position as trimethylsilyl derivatives. This protection is crucial because it prevents unwanted side reactions during the subsequent quaternization step. Once the protective groups are established, the reaction mixture is cooled, and additional TMSI along with N-methylpyrrolidine is introduced. The TMSI acts as a potent iodinating agent, converting the C-3 acetoxymethyl group into a highly reactive iodomethyl intermediate. This activated species immediately undergoes nucleophilic attack by the nitrogen of N-methylpyrrolidine, forming the quaternary ammonium salt characteristic of Cefepime. The entire sequence occurs in a single vessel, minimizing material transfer losses and exposure to atmospheric moisture which could hydrolyze the sensitive beta-lactam ring.
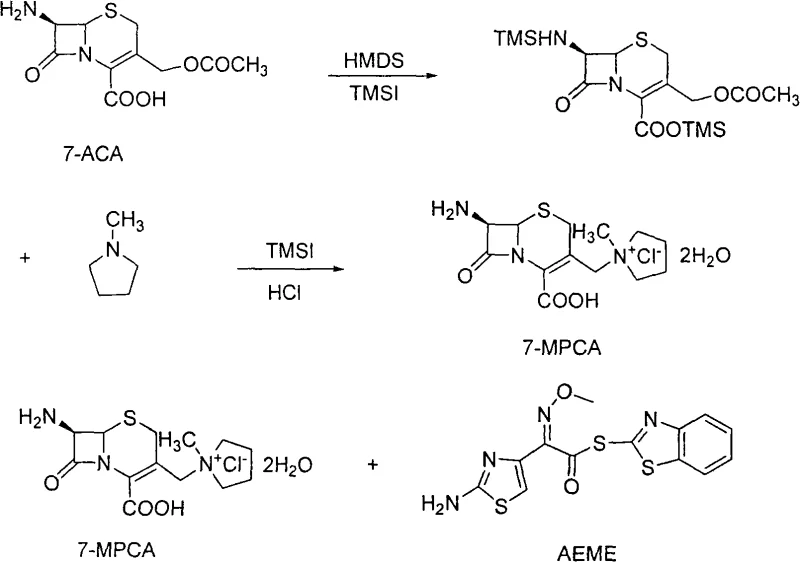
Following the formation of 7-MPCA, the acylation step involves reacting the intermediate with AE active ester (AEEME) in a carefully controlled pH environment (7.0 to 9.5). The use of a mixed solvent system comprising organic solvents like DMF or DMAC and water allows for the dissolution of both the hydrophilic 7-MPCA salt and the lipophilic active ester. A critical aspect of this mechanism is the purification strategy employed post-acylation. The patent describes a unique refinement treatment where the crude cefepime solution undergoes activated carbon decolorization followed by passage through a gamma-alumina column. This specific chromatographic step is engineered to adsorb and remove mercaptobenzothiazole, a thiol-containing byproduct generated from the leaving group of the active ester, which is notoriously difficult to remove and often causes discoloration. This rigorous impurity control mechanism ensures that the final crystallized hydrochloride salt meets stringent visual and chemical purity specifications required for parenteral administration.
How to Synthesize Cefepime Hydrochloride Efficiently
The synthesis of Cefepime Hydrochloride via this patented route requires precise control over stoichiometry, temperature, and pH to ensure optimal conversion and minimal degradation. The process begins with the silylation of 7-ACA in an anhydrous organic solvent such as dichloromethane or acetonitrile, followed by the sequential addition of quaternizing agents at low temperatures to manage exotherms. Detailed standardized operating procedures regarding specific molar ratios, addition rates, and crystallization parameters are essential for reproducibility. For a comprehensive breakdown of the exact experimental conditions and step-by-step protocols validated in the patent examples, please refer to the technical guide below.
- Protect 7-ACA amino and carboxyl groups using HMDS and TMSI in organic solvent under reflux.
- Perform one-pot quaternization with N-methylpyrrolidine and TMSI to synthesize 7-MPCA hydrochloride.
- React 7-MPCA with AE active ester followed by acidification and crystallization to obtain Cefepime Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial cost savings derived from process intensification; by combining protection and quaternization into a one-pot operation, the number of unit operations is drastically reduced. This consolidation eliminates the need for intermediate isolation, drying, and re-dissolution steps, which are typically labor-intensive and solvent-heavy. Consequently, the overall consumption of raw materials and utilities is significantly lowered, directly impacting the cost of goods sold (COGS). Furthermore, the use of common, commodity-grade solvents like acetone, dichloromethane, and DMF ensures that the supply chain remains resilient against fluctuations in specialty chemical availability, securing a more stable production schedule for high-purity cefepime hydrochloride.
- Cost Reduction in Manufacturing: The elimination of complex isolation steps and the ability to recycle solvents such as acetone mother liquors create a leaner manufacturing profile. By avoiding the use of expensive starting materials like GCLE and instead relying on the more abundant 7-ACA, the raw material cost base is fundamentally optimized. Additionally, the regeneration capability of the gamma-alumina column means that purification media costs are amortized over multiple batches, further driving down operational expenses without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical process translates directly into supply chain security. The reaction conditions are forgiving enough to allow for large batch sizes without the risk of thermal runaway or significant yield loss, which is a common bottleneck in beta-lactam synthesis. The stability of the final hydrochloride salt form ensures that the product can be stored and transported under standard conditions without rapid degradation, reducing the risk of inventory write-offs. This reliability is critical for maintaining continuity of supply for essential antibiotic medications in the global market.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for industrial throughput. The waste streams are manageable, primarily consisting of aqueous phases and recoverable organic solvents that can be distilled and reused. The avoidance of heavy metal catalysts or exotic reagents simplifies waste treatment and disposal compliance. The crystallization step using acetone is highly scalable, allowing manufacturers to easily transition from pilot plant quantities to multi-ton commercial production campaigns to meet surging demand for fourth-generation cephalosporins.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefepime Hydrochloride using this specific patented methodology. These insights are derived directly from the experimental data and process descriptions found in the intellectual property documentation, providing clarity on impurity profiles, scalability, and quality control measures. Understanding these nuances is vital for technical teams evaluating potential manufacturing partners or licensing opportunities for this advanced synthetic route.
Q: What is the key advantage of the 7-MPCA intermediate route?
A: The route utilizes a one-pot method to convert protected 7-ACA directly into 7-MPCA, significantly simplifying the process flow and reducing solvent usage compared to multi-step isolation methods.
Q: How is product color and purity controlled in this synthesis?
A: The process employs a specialized purification step using activated carbon decolorization followed by gamma-alumina column chromatography, which effectively removes mercaptobenzothiazole byproducts and ensures high visual quality.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method features simple processing conditions, convenient operation, and high yields, making it highly suitable for continuity and large-scale industrial manufacturing of fourth-generation cephalosporins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality antibiotic intermediates that meet the rigorous demands of the global pharmaceutical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of Cefepime Hydrochloride conforms to the highest international standards, minimizing impurities such as mercaptobenzothiazole through our advanced purification protocols.
We invite you to collaborate with us to leverage this innovative synthesis technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your mission to deliver life-saving antibiotics to patients worldwide with unmatched reliability and quality.
