Optimizing Cephalosporin Antibiotic Production: A Novel Activated Carbon Purification Strategy for High-Purity Z-Isomers
Introduction to Advanced Cephalosporin Manufacturing
The global demand for third-generation cephalosporin antibiotics continues to drive innovation in pharmaceutical intermediate synthesis, particularly for compounds requiring high stereochemical purity. Patent CN102725297A introduces a transformative approach to manufacturing cephalosporin derivatives, specifically targeting the production of Cefditoren and its precursors with superior Z-isomer content. This technology addresses a critical bottleneck in antibiotic production: the separation of Z and E isomers, where only the Z-configuration exhibits the desired potent antibacterial activity. By leveraging a specialized activated carbon treatment followed by a streamlined acylation reaction, this process offers a robust pathway for producing high-purity pharmaceutical intermediates suitable for large-scale commercialization.
The significance of this patent lies in its ability to bypass traditional, cumbersome protection-deprotection sequences. Conventional methods often rely on protecting the amino group at the 7-position, which necessitates additional synthetic steps and reagents to remove the protecting group later. In contrast, the disclosed method utilizes a direct purification strategy on the 7-amino-3-cephem-4-carboxylic acid scaffold. This not only simplifies the operational workflow but also enhances the overall yield and purity profile of the final active pharmaceutical ingredient (API). For reliable pharmaceutical intermediates suppliers, adopting such efficient purification technologies is essential to maintain competitiveness in a market that increasingly demands cost-effective and environmentally sustainable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cephalosporin antibiotics like Cefditoren has been plagued by the challenge of isomeric contamination. Traditional synthetic routes often involve the use of protected amino intermediates to control regioselectivity during the acylation step. However, this approach introduces significant inefficiencies. The installation and subsequent removal of amino protecting groups add at least two extra unit operations to the manufacturing process, increasing solvent consumption, waste generation, and overall production time. Furthermore, standard purification techniques such as recrystallization or chromatography often struggle to effectively separate the Z and E isomers due to their similar physicochemical properties, leading to final products with suboptimal isomeric purity that may fail stringent regulatory specifications.
Another major drawback in existing processes is the management of byproducts generated during enzymatic deprotection steps. When synthesizing the 7-amino cephem core via enzymatic cleavage of a 7-acylamino precursor, phenylacetic acid is inevitably produced as a stoichiometric byproduct. In conventional workflows, removing this acidic impurity requires multiple solvent extraction steps or complex pH adjustments, which can lead to product degradation or loss. The inability to efficiently remove phenylacetic acid and the E-isomer simultaneously forces manufacturers to accept lower yields or invest in expensive downstream processing equipment, creating a significant barrier to cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
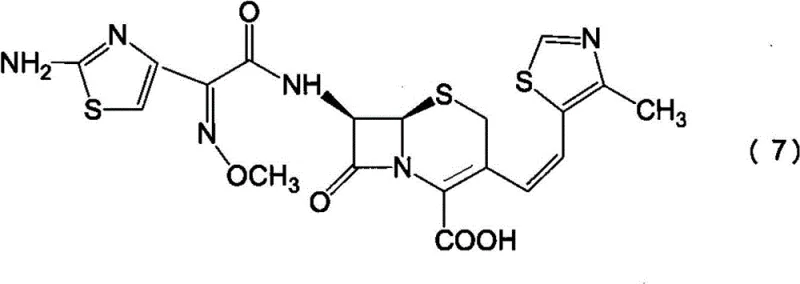
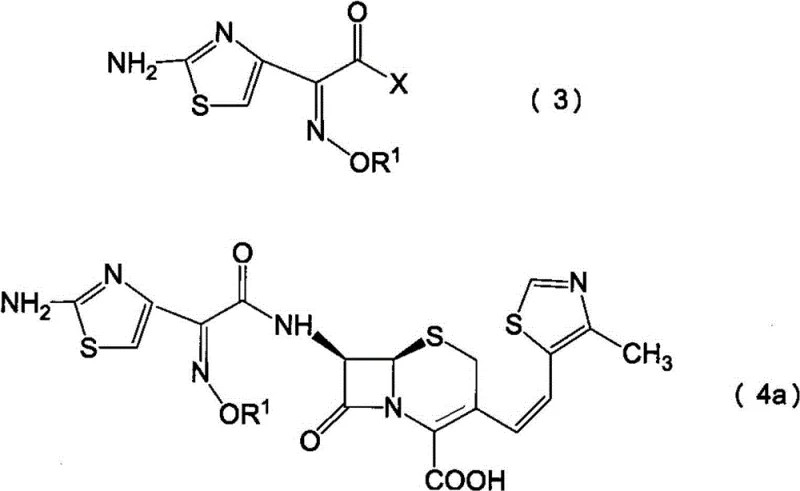
The patented process revolutionizes this landscape by introducing a highly selective adsorption step using specifically characterized activated carbon. Instead of relying on chemical protection strategies, the method treats a mixture of Z and E isomers of 7-amino-3-[(E/Z)-2-(4-methylthiazol-5-yl)vinyl]-3-cephem-4-carboxylic acid with activated carbon possessing defined physical properties. Specifically, the carbon must exhibit an iodine adsorption capacity of at least 1200 mg/g and a methylene blue adsorption capacity of at least 250 ml/g. These parameters indicate a unique pore size distribution capable of discriminating between the molecular geometries of the Z and E isomers.
This novel approach allows for the simultaneous removal of the unwanted E-isomer and phenylacetic acid impurities in a single unit operation. By contacting the aqueous solution of the cephem compound with this specialized carbon, the E-isomer is selectively adsorbed onto the carbon surface, while the desired Z-isomer remains in the solution. This breakthrough eliminates the need for amino protecting groups entirely, streamlining the synthesis from the 7-amino intermediate to the final antibiotic. The result is a significantly simplified process flow that enhances the commercial scale-up of complex pharmaceutical intermediates by reducing the number of reaction vessels and purification stages required.
Mechanistic Insights into Selective Adsorption and Acylation
The core mechanism driving the success of this process is the physical adsorption phenomenon governed by the pore structure of the activated carbon. The specified iodine and methylene blue adsorption values correspond to the presence of both micropores and mesopores within the carbon matrix. The micropores (indicated by iodine adsorption) are effective at trapping smaller molecules or specific functional groups, while the mesopores (indicated by methylene blue adsorption) accommodate larger molecular structures. The E-isomer of the vinyl-cephem side chain likely presents a steric configuration that fits more readily into these specific pore environments compared to the Z-isomer, or perhaps interacts more strongly with the carbon surface due to dipole alignment. This differential adsorption allows for the enrichment of the Z-isomer in the liquid phase to levels exceeding 90%, a purity threshold difficult to achieve via crystallization alone.
Following the purification step, the enriched Z-isomer undergoes acylation with an active ester, typically a benzothiazolyl thioester derivative. The reaction mechanism involves nucleophilic attack by the 7-amino group of the cephem nucleus on the carbonyl carbon of the active ester. The presence of the electron-withdrawing benzothiazole ring activates the carbonyl group, facilitating the formation of the amide bond under mild conditions. Crucially, because the starting material has been pre-purified to remove phenylacetic acid, the reaction environment is cleaner, minimizing side reactions such as hydrolysis of the beta-lactam ring. This ensures that the final product retains the structural integrity necessary for biological activity, demonstrating a clear advantage in high-purity pharmaceutical intermediates production.
How to Synthesize Cefditoren Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for manufacturing high-quality cephalosporin derivatives. The process begins with the preparation of an aqueous solution of the alkenyl cephem compound, which may exist as a salt depending on the pH adjustment. This solution is then subjected to the critical activated carbon treatment, where temperature and contact time are controlled to maximize adsorption efficiency without degrading the sensitive beta-lactam core. Following filtration to remove the carbon, the purified solution is reacted with the side-chain acid derivative to form the final amide bond. For detailed operational parameters and specific reagent quantities, please refer to the standardized synthesis guide below.
- Prepare an aqueous solution of the alkenyl cephem compound containing both Z and E isomers, optionally adjusting pH to acidic or weakly alkaline ranges depending on salt formation.
- Contact the solution with specific activated carbon having iodine adsorption >1200mg/g and methylene blue adsorption >250ml/g to selectively remove E-isomers and phenylacetic acid byproducts.
- React the purified Z-enriched intermediate with an active ester compound (such as a benzothiazolyl thioester) in the presence of an organic base to form the final cephalosporin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits beyond mere technical superiority. The elimination of amino protecting groups translates directly into a reduction in raw material costs, as expensive protecting reagents and the solvents required for their removal are no longer needed. Furthermore, the simplified workflow reduces the overall cycle time for production batches, allowing facilities to increase throughput without expanding physical infrastructure. This efficiency gain is crucial for maintaining a competitive edge in the global supply of antibiotic intermediates, where margin pressures are constant.
- Cost Reduction in Manufacturing: The process significantly lowers operational expenditures by consolidating purification steps. By using activated carbon to remove both isomeric impurities and phenylacetic acid byproducts simultaneously, the need for multiple solvent extraction cycles is reduced or eliminated. This consolidation leads to substantial savings in solvent procurement and waste disposal costs. Additionally, the avoidance of protection-deprotection chemistry removes the cost associated with specialized reagents and the energy-intensive steps required to drive those reactions to completion, resulting in a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on standard, commercially available activated carbon with defined specifications ensures a stable supply of critical processing aids. Unlike proprietary catalysts or enzymes that may have long lead times or single-source risks, high-performance activated carbon is a commodity chemical available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the adsorption process against variations in feedstock quality ensures consistent output, reinforcing the reliability of the supply chain for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the elimination of hazardous deprotection reagents align with green chemistry principles. The process generates less chemical waste, simplifying compliance with increasingly stringent environmental regulations. The scalability of the adsorption step is well-understood in the chemical industry, allowing for seamless transition from pilot scale to multi-ton commercial production. This ease of scale-up ensures that manufacturers can rapidly respond to market demand surges for cephalosporin antibiotics without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cephalosporin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and mechanistic details for potential partners and licensees. Understanding these nuances is key to evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why is specific activated carbon critical for Z-isomer enrichment?
A: Standard activated carbon lacks the specific pore size distribution required to differentiate between Z and E isomers. The patented process utilizes carbon with high iodine and methylene blue adsorption values, indicating a bimodal pore distribution that selectively traps the E-isomer and phenylacetic acid impurities while allowing the Z-isomer to pass through.
Q: How does this process handle phenylacetic acid byproducts?
A: Phenylacetic acid is a common byproduct of enzymatic deprotection in cephalosporin synthesis. This method leverages the strong adsorption affinity of the specific activated carbon for phenylacetic acid, removing it simultaneously with the E-isomer, thereby simplifying the purification workflow and eliminating the need for complex solvent extraction steps in some embodiments.
Q: What are the industrial advantages of this synthesis route?
A: The process avoids the use of amino protecting groups that require harsh deprotection conditions. By starting with a mixed isomer feed and purifying via adsorption, manufacturers can utilize cheaper starting materials and achieve high Z-isomer content directly, reducing overall processing time and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefditoren Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and efficiency in the production of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN102725297A can be successfully implemented at an industrial level. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to optimize your cephalosporin supply chain. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
