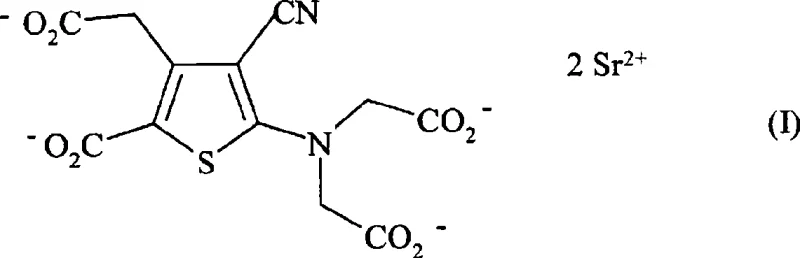
Optimizing Strontium Ranelate Production For Commercial Scale And Purity Standards
The pharmaceutical industry continuously seeks robust manufacturing pathways for active ingredients that treat chronic conditions such as osteoporosis. Patent CN101397292A introduces a significant advancement in the synthesis of strontium ranelate, a compound widely recognized for its therapeutic efficacy in bone disease management. This technical disclosure outlines a method that overcomes historical limitations in yield and purity, offering a viable route for industrial scale-up. By leveraging specific solvent systems and controlled reaction parameters, the process ensures the production of high-purity strontium ranelate suitable for stringent regulatory requirements. As a reliable strontium ranelate supplier, understanding these mechanistic improvements is crucial for securing a stable supply chain. The following analysis details how this innovation transforms the production landscape for this critical pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of strontium ranelate has been plagued by inefficiencies that hinder commercial viability and cost-effectiveness. Prior art, specifically referenced in European patent specification EP 0 415 850, describes methods involving the heating of tetraethyl esters with sodium hydroxide in aqueous alcoholic media under reflux conditions. This conventional approach necessitates the distillation of ethanol and water to isolate intermediate tetrasodium salts before reacting them with strontium chloride. Such multi-step isolation procedures are not only energy-intensive but also introduce significant opportunities for product loss and impurity accumulation. Data from previous attempts indicate that yields often remain stubbornly below 70%, which is economically unsustainable for large-scale manufacturing. Furthermore, the harsh thermal conditions required for reflux can lead to the degradation of sensitive functional groups, complicating downstream purification and increasing the burden on quality control laboratories to remove trace impurities.
The Novel Approach
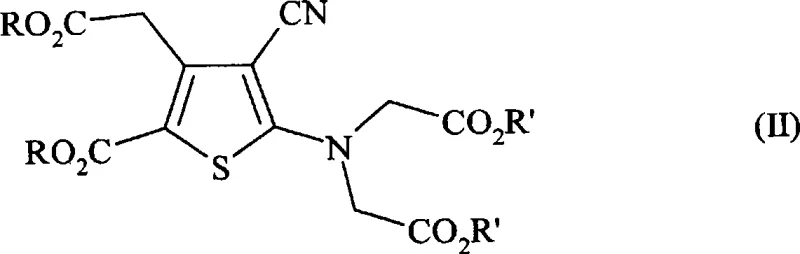
In stark contrast, the methodology disclosed in CN101397292A presents a streamlined and highly efficient alternative that fundamentally alters the reaction environment. The novel approach utilizes a tetra-ester precursor, preferably the tetramethyl ester, which undergoes saponification in a mixture of water and organic solvents at moderate temperatures ranging from 0 to 100°C. A critical innovation lies in the salt conversion step, where the intermediate alkali metal salt reacts with strontium chloride in the continued presence of organic solvents such as tetrahydrofuran, acetone, or alcohols. This strategic inclusion of organic co-solvents during the metathesis reaction surprisingly enhances the precipitation kinetics and crystal quality of the final strontium salt. By eliminating the need for rigorous distillation steps between saponification and salt formation, the process reduces operational complexity and significantly boosts overall recovery rates to levels exceeding 90%, marking a substantial improvement over legacy techniques.
Mechanistic Insights into Optimized Salt Formation and Saponification
The core of this technological breakthrough resides in the precise control of solubility and reactivity during the transition from the organic ester to the inorganic salt. The process begins with the hydrolysis of the tetra-ester functionality using strong bases like sodium hydroxide or potassium hydroxide. Unlike traditional methods that might rely on单一 aqueous systems, this protocol employs a biphasic or homogeneous mixture involving water and polar aprotic or protic organic solvents. This solvent engineering ensures that the intermediate carboxylate species remain sufficiently solubilized to react completely while preventing the formation of intractable oils that often trap impurities. The reaction temperature is carefully maintained between 20 and 70°C, a range that balances reaction kinetics with thermal stability, ensuring that the cyano and thiophene moieties within the molecular scaffold remain intact throughout the transformation.
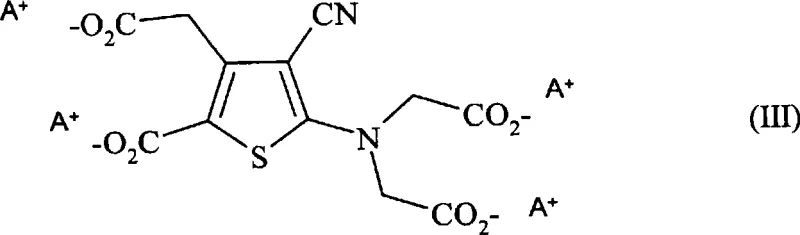
Following the generation of the intermediate alkali metal salt, typically represented as the tetrasodium or tetrapotassium species, the system proceeds directly to the strontium exchange without mandatory isolation. The addition of strontium chloride in an aqueous solution, combined with the existing organic solvent matrix, triggers the selective precipitation of the strontium ranelate complex. The presence of the organic component modifies the dielectric constant of the medium, thereby reducing the solubility of the target strontium salt and promoting rapid, clean crystallization. This mechanism effectively minimizes the occlusion of mother liquor and inorganic byproducts within the crystal lattice, leading to a product with superior purity profiles. The ability to clarify the intermediate solution prior to strontium addition further enhances the removal of insoluble particulates, ensuring that the final filtration step yields a chemically pure solid ready for drying and packaging.
How to Synthesize Strontium Ranelate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent selection to replicate the high yields reported in the patent examples. The procedure generally involves dissolving the tetra-ester precursor in a reactor equipped with temperature control and agitation, followed by the gradual addition of the base solution. Maintaining the reaction mixture within the specified thermal window is essential to drive the saponification to completion without side reactions. Once the intermediate salt is formed, the introduction of the strontium source must be managed to control the rate of precipitation, ensuring the formation of filterable crystals rather than fine amorphous powders. Detailed standardized operating procedures regarding mixing speeds, addition rates, and washing protocols are critical for transferring this laboratory success to commercial production environments. For a comprehensive breakdown of the specific operational parameters, please refer to the technical guide below.
- Perform saponification of the tetra-ester precursor using sodium or potassium hydroxide in a water-organic solvent mixture at 20-70°C.
- React the resulting intermediate salt solution directly with strontium chloride in the presence of organic solvents like THF or ethanol.
- Isolate the final strontium ranelate product through rapid filtration and drying to ensure high purity and yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this optimized synthesis method offers tangible benefits that extend beyond mere chemical yield. For procurement managers and supply chain directors, the primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced reliability. By eliminating energy-intensive distillation steps and reducing the number of unit operations, the overall production cycle time is compressed, allowing for faster turnaround on orders. This efficiency gain is particularly vital in the context of cost reduction in pharmaceutical intermediates manufacturing, where margin pressures are constant. The robust nature of the process also implies a lower risk of batch failures, ensuring a more consistent supply of material to downstream formulation partners.
- Cost Reduction in Manufacturing: The elimination of complex isolation steps for the intermediate sodium or potassium salts significantly lowers utility consumption and labor costs associated with extended processing times. By achieving yields that are substantially higher than the industry standard of 70%, the amount of raw material required per kilogram of final product is drastically reduced, leading to direct savings on input costs. Furthermore, the use of common, commercially available solvents like ethanol and acetone avoids the need for specialized or expensive reagents, keeping the bill of materials lean and predictable. This economic efficiency allows suppliers to offer more competitive pricing structures without compromising on quality standards.
- Enhanced Supply Chain Reliability: The mild reaction conditions, operating primarily between 20 and 70°C, reduce the thermal stress on equipment and minimize the risk of safety incidents related to high-pressure or high-temperature operations. This operational safety profile contributes to greater plant uptime and fewer unplanned shutdowns, which are critical factors for maintaining continuous supply to global markets. Additionally, the flexibility to use various organic solvents provides a buffer against supply chain disruptions for specific chemicals, as alternative solvents can be substituted without fundamentally altering the reaction outcome. This adaptability ensures that production schedules remain resilient even in the face of raw material volatility.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with filtration steps described as very fast, indicating excellent physical properties of the precipitated solid that facilitate handling in large-scale reactors. The reduction in solvent usage and the avoidance of extensive distillation also contribute to a lower environmental footprint, aligning with increasingly strict global regulations on industrial emissions and waste disposal. Efficient solid-liquid separation reduces the volume of wastewater generated, simplifying effluent treatment requirements and lowering compliance costs. These factors collectively make the technology highly attractive for long-term commercial partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and implications of this synthesis technology, we have compiled answers based on the specific data points and embodiments found within the patent literature. These questions focus on the practical aspects of yield improvement, reaction conditions, and intermediate handling that are most relevant to technical decision-makers. Understanding these nuances helps in evaluating the feasibility of integrating this method into existing production lines or when qualifying new vendors for critical supply contracts. The following responses provide clarity on how the theoretical advantages translate into practical manufacturing outcomes.
Q: How does the new synthesis method improve yield compared to prior art?
A: The patented method achieves yields exceeding 90% by utilizing organic solvents during the strontium salt conversion step, whereas conventional methods often result in yields below 70% due to inefficient precipitation.
Q: What are the preferred reaction conditions for this process?
A: The process operates under mild conditions, with saponification occurring between 20-70°C and salt conversion between 20-50°C, reducing energy consumption and thermal degradation risks.
Q: Is the intermediate salt isolated before strontium addition?
A: The process allows flexibility; the intermediate salt solution can be clarified and used directly for reaction with strontium chloride, streamlining the workflow and reducing processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Strontium Ranelate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in laboratory settings are faithfully reproduced at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch of strontium ranelate meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that clients can rely on us for consistent material performance, minimizing the risk of delays in their own drug development or manufacturing timelines.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this high-efficiency method. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to secure a stable, cost-effective, and high-quality supply of this essential therapeutic intermediate for your global operations.
