Revolutionizing Amide Bond Formation: A Dual Iridium-Cobalt Photocatalytic Strategy for Commercial Scale-Up
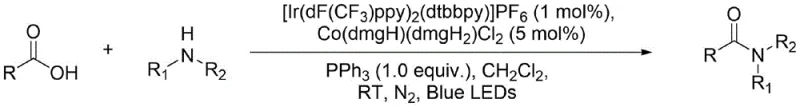
The landscape of amide bond construction is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies in the pharmaceutical and fine chemical sectors. Patent CN112778238A introduces a groundbreaking approach that leverages the synergistic power of dual metal catalysis under visible light irradiation to directly couple carboxylic acids with amines. This technology utilizes a sophisticated catalytic system comprising an Iridium-based photocatalyst, specifically [Ir(dF(CF3)ppy)2(dtbbpy)]PF6, and a Cobalt complex, Co(dmgH)(dmgH2)Cl2. By employing triphenylphosphine as a deoxygenating agent in dichloromethane under an inert atmosphere, this method bypasses the traditional limitations of direct amidation. The innovation lies in its ability to activate stable carboxylic acids under mild room temperature conditions using blue LEDs, offering a robust alternative to harsh thermal methods or waste-generating coupling protocols. For R&D directors and process chemists, this represents a pivotal shift towards high-atom-economy transformations that align with modern green chemistry principles while maintaining the rigorous purity standards required for API intermediate synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the formation of amide bonds from carboxylic acids and amines has been plagued by significant thermodynamic and kinetic barriers. The direct condensation reaction is often unfavorable due to the competing acid-base interaction between the reactants, which leads to the formation of unreactive ammonium carboxylate salts rather than the desired amide. To overcome this high activation energy, conventional industrial practices rely heavily on the use of stoichiometric activating agents such as carbodiimides (e.g., EDC, DCC) or phosphonium/uronium salts (e.g., HATU, PyBOP). While effective, these reagents introduce severe drawbacks for large-scale manufacturing, primarily concerning atom economy and waste generation. For every mole of amide produced, at least one mole of stoichiometric by-product, such as urea derivatives, is generated, necessitating complex and costly purification steps to remove these impurities. Furthermore, many coupling agents are moisture-sensitive, expensive, and can pose safety hazards during handling and storage. The removal of these by-products often requires extensive chromatographic separation or recrystallization, which drastically reduces overall yield and increases the environmental footprint of the process through excessive solvent consumption.
The Novel Approach
In stark contrast to these legacy methods, the photocatalytic strategy disclosed in the patent offers a streamlined pathway that fundamentally alters the reaction mechanism. By harnessing the energy of blue light photons, the Iridium photocatalyst is excited to a high-energy state capable of engaging in single-electron transfer processes. This photo-induced activation, coupled with the Cobalt complex which likely facilitates radical generation or hydrogen atom transfer, allows for the direct dehydration of the acid-amine pair without the need for pre-activation of the carboxylic acid. The use of triphenylphosphine serves as a crucial oxygen scavenger, driving the equilibrium forward by sequestering the oxygen atom from the carboxylic acid. This approach not only operates under exceptionally mild conditions (room temperature) but also significantly reduces the E-factor of the reaction by eliminating stoichiometric coupling waste. The result is a cleaner reaction profile that simplifies downstream processing, making it an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing where purity and efficiency are paramount.
Mechanistic Insights into Iridium-Cobalt Dual Photocatalysis
The efficacy of this transformation relies on the intricate interplay between the photoredox cycle of the Iridium complex and the catalytic activity of the Cobalt species. Upon irradiation with blue LEDs, the ground state Iridium(III) catalyst absorbs a photon to reach an excited triplet state, *[Ir(III)], which possesses strong oxidizing or reducing potential depending on the specific ligand environment. In this system, the excited photocatalyst likely interacts with the triphenylphosphine or the substrate to generate radical intermediates. The Cobalt complex, featuring a dimethylglyoxime (dmg) ligand framework, acts as a co-catalyst that may stabilize radical species or facilitate the cleavage of the O-H bond in the carboxylic acid. This dual catalytic manifold enables the generation of an acyl radical or a similar activated species that is highly susceptible to nucleophilic attack by the amine. The precise tuning of the redox potentials ensures that the reaction proceeds selectively without degrading sensitive functional groups present on the aromatic rings or aliphatic chains of the substrates.
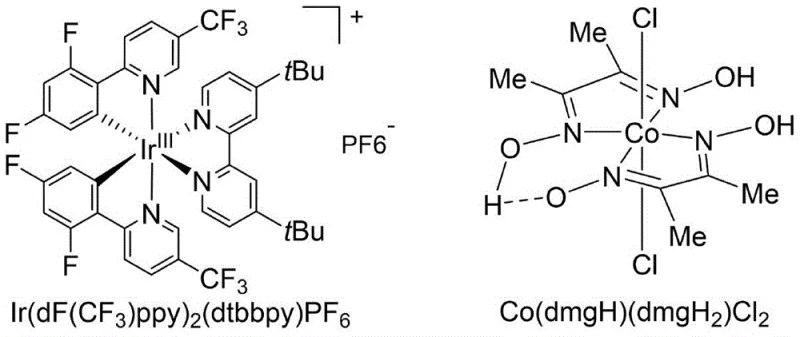
Furthermore, the mechanism inherently controls impurity profiles by avoiding the formation of stable side-products typical of coupling reagents. The radical nature of the intermediate species allows for rapid recombination to form the amide bond, while the catalytic turnover ensures that only minute quantities of the expensive metal complexes are required (typically 1 mol% for Ir and 5 mol% for Co). This mechanistic pathway is particularly advantageous for substrates containing base-sensitive moieties, as the reaction does not require strong bases often needed in traditional activation protocols. The robustness of the catalytic cycle is evidenced by the broad substrate scope, tolerating electron-withdrawing and electron-donating groups alike, which suggests a highly versatile electronic environment within the catalytic pocket. Understanding this mechanism is critical for process optimization, as factors such as light intensity, oxygen exclusion, and stirring speed directly influence the efficiency of the photon absorption and subsequent radical propagation steps.
How to Synthesize N-Benzylbenzamide Efficiently
To implement this advanced methodology in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding light exposure and atmospheric control. The synthesis generally begins with the precise weighing of the carboxylic acid starting material, followed by the addition of the catalytic system and the deoxygenating agent. The reaction mixture is then subjected to multiple vacuum-nitrogen cycles to ensure a completely inert environment, which is crucial for preventing the quenching of the excited photocatalyst by molecular oxygen. Once the amine is introduced, the vessel is placed in a photoreactor equipped with blue LEDs, where it is stirred vigorously to maximize photon flux penetration throughout the solution. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction vessel by weighing the carboxylic acid substrate, triphenylphosphine (PPh3), and the dual catalyst system consisting of the Iridium photocatalyst and Cobalt complex.
- Seal the vessel, evacuate the atmosphere three times to ensure an inert environment, and add dichloromethane (DCM) as the solvent under nitrogen protection.
- Slowly add the amine substrate to the mixture and place the reaction vessel under blue LED irradiation at room temperature with vigorous stirring for 3 to 18 hours.
- Monitor reaction progress via TLC, remove the solvent by rotary evaporation upon completion, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers compelling strategic advantages that extend beyond mere technical novelty. The primary value proposition lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on specialized, often imported, stoichiometric coupling reagents, manufacturers can mitigate risks associated with supply disruptions and price volatility of these high-cost additives. Instead, the process relies on bulk commodity chemicals like carboxylic acids and amines, alongside catalytic amounts of metal complexes that, while precious, are used in such low quantities that their impact on the Bill of Materials is minimal. This shift translates directly into substantial cost savings in amide manufacturing, as the expense of purchasing, storing, and disposing of tons of coupling waste is effectively removed from the operational budget.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of stoichiometric waste streams. Traditional amidation generates equimolar amounts of by-products that require expensive disposal and complicate purification. By switching to a catalytic deoxygenative approach, the process mass intensity (PMI) is significantly lowered. This reduction in waste handling costs, combined with the decreased solvent usage required for purification (since there are no urea by-products to separate), leads to a leaner, more cost-effective production model. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Reliance on complex coupling agents often introduces bottlenecks, as these reagents can have long lead times and strict storage requirements. This new method utilizes widely available carboxylic acids and amines, which are staple commodities in the chemical industry with robust and diversified supply chains. The catalysts, though specialized, are used in such small loadings that a single batch can last for extended production runs, minimizing the frequency of reordering. This stability ensures consistent production schedules and reduces the risk of downtime caused by missing critical reagents, thereby improving the reliability of delivery to downstream customers.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this method aligns perfectly with increasingly stringent environmental standards. The absence of toxic coupling by-products simplifies wastewater treatment and reduces the burden on effluent management systems. The reaction has been demonstrated to scale effectively from milligram to gram levels with consistent yields, indicating a clear path to kilogram and ton-scale production. The use of blue LED lights is energy-efficient and easily scalable in flow chemistry setups, allowing for continuous manufacturing processes that offer superior heat and mass transfer compared to batch reactors. This scalability ensures that the technology can meet the demands of commercial production without compromising on quality or safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dual catalytic system. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology for industrial partners.
Q: What are the key advantages of this photocatalytic amidation method over traditional coupling agents?
A: This method eliminates the need for stoichiometric coupling reagents like EDC or HATU, which generate significant chemical waste. By utilizing a catalytic amount of Iridium and Cobalt complexes with triphenylphosphine as a deoxygenator, the process achieves superior atom economy and simplifies downstream purification by avoiding urea byproducts.
Q: What is the substrate scope for this dual catalytic system?
A: The protocol demonstrates excellent functional group tolerance, accommodating a wide range of carboxylic acids including aryl, heteroaryl, and protected amino acids. It is also compatible with various amines such as primary alkyl amines, benzyl amines, and cyclic amines, making it highly versatile for complex molecule synthesis.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction operates under mild conditions (room temperature) and has been successfully demonstrated on gram scales. The use of commercially available catalysts and standard blue LED equipment facilitates scalability, while the absence of hazardous coupling agents enhances safety profiles for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Iridium-Cobalt dual catalytic technology for the synthesis of high-value amide intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our facility is equipped with state-of-the-art photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical clients. We understand that transitioning to a new catalytic platform requires confidence in both the chemistry and the supply capability, and our team is dedicated to de-risking this adoption for your projects.
We invite you to engage with our technical procurement team to discuss how this methodology can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching from traditional coupling methods to this photocatalytic route. We encourage you to contact us today to obtain specific COA data for similar amide structures and to receive comprehensive route feasibility assessments that demonstrate our commitment to delivering high-quality, cost-effective chemical solutions.
