Revolutionizing Amide Bond Construction: Scalable Iridium-Cobalt Dual Catalysis for Commercial Pharmaceutical Manufacturing
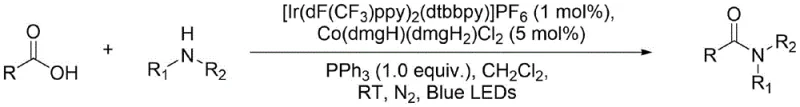
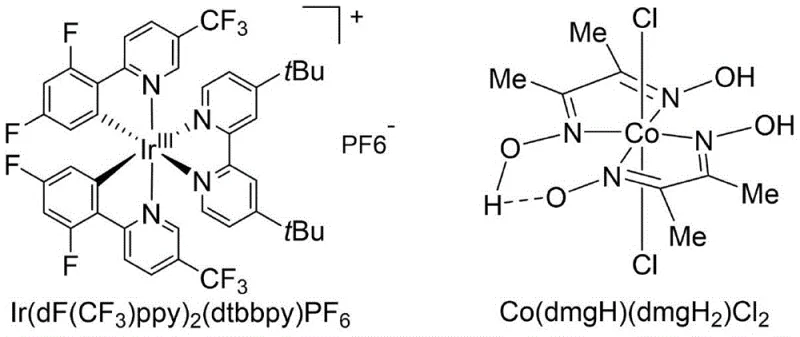
The pharmaceutical and fine chemical industries rely heavily on the formation of amide bonds, a structural motif present in approximately 25% of marketed drugs and a critical linkage in biological systems such as proteins. Patent CN112778238A introduces a groundbreaking methodology for preparing amides from carboxylic acids and amines using a synergistic dual-catalytic system involving iridium and cobalt complexes under blue light irradiation. This innovation addresses the longstanding challenges associated with direct amidation, such as high activation barriers and competing acid-base reactions, by leveraging photoredox chemistry to facilitate the coupling process under exceptionally mild conditions. The method utilizes stable and easily obtained carboxylic acids and amines as raw materials, avoiding the need for pre-activation steps that often complicate synthetic routes. By employing [Ir(dF(CF3)ppy)2(dtbbpy)]PF6 as a photocatalyst and Co(dmgH)(dmgH2)Cl2 as a metal complex catalyst alongside triphenylphosphine as a deoxidizer, this protocol achieves high-efficiency preparation of amide compounds with excellent functional group tolerance. This technological advancement represents a significant leap forward for reliable pharmaceutical intermediate supplier networks seeking greener and more sustainable manufacturing pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the formation of amide bonds from carboxylic acids and amines has been plagued by thermodynamic and kinetic hurdles that necessitate the use of harsh reaction conditions or stoichiometric activating agents. Conventional protocols typically rely on coupling reagents such as carbodiimides (e.g., EDC, DCC) or uronium salts (e.g., HATU, TBTU) to activate the carboxylic acid prior to nucleophilic attack by the amine. While effective, these methods suffer from poor atom economy because they generate more than one equivalent of by-product for every molecule of product formed, leading to substantial chemical waste and increased disposal costs. Furthermore, the removal of these stoichiometric by-products and the coupling agents themselves from the reaction mixture is often a tedious and expensive purification process that can significantly impact the overall yield and purity of the final active pharmaceutical ingredient. The high activation barriers of direct coupling also frequently require elevated temperatures or strong bases, which can be incompatible with sensitive functional groups commonly found in complex drug candidates and agrochemical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN112778238A utilizes a photocatalysis and metal complex catalysis concerted strategy that operates under mild room temperature conditions with visible light irradiation. This method bypasses the need for stoichiometric coupling agents entirely by employing a catalytic amount of an iridium photocatalyst and a cobalt complex to drive the reaction through a radical-mediated mechanism. The use of triphenylphosphine as a deoxidizer facilitates the reduction steps necessary for the catalytic cycle, ensuring high conversion rates without the generation of difficult-to-remove salt by-products. This shift from stoichiometric activation to catalytic turnover not only improves the environmental profile of the synthesis but also drastically simplifies the workup procedure, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing. The broad substrate scope, which includes aryl, heteroaryl, and alkyl carboxylic acids as well as various primary and secondary amines, demonstrates the versatility of this new protocol for synthesizing diverse amide libraries.
Mechanistic Insights into Ir/Co Dual-Catalyzed Photoredox Amidation
The core of this innovative synthesis lies in the sophisticated interplay between the iridium photocatalyst and the cobalt metal complex, which work in concert to activate the carboxylic acid and facilitate amide bond formation. Upon irradiation with blue LEDs, the iridium complex [Ir(dF(CF3)ppy)2(dtbbpy)]PF6 absorbs photons to reach an excited state, enabling it to participate in single-electron transfer processes that generate reactive radical species from the phosphine deoxidizer. These phosphine radicals play a crucial role in the activation of the carboxylic acid substrate, effectively lowering the energy barrier for the subsequent nucleophilic attack by the amine. Simultaneously, the cobalt complex Co(dmgH)(dmgH2)Cl2 acts as a co-catalyst that likely stabilizes intermediate species and assists in the regeneration of the active catalytic forms, ensuring the continuity of the catalytic cycle. This dual-catalytic manifold allows the reaction to proceed efficiently at room temperature, avoiding the thermal degradation pathways that often compromise sensitive substrates in traditional thermal amidation reactions.
Impurity control in this photocatalytic system is inherently superior due to the high chemoselectivity of the radical mechanism and the mild reaction conditions employed. Unlike traditional coupling methods that can lead to racemization of chiral centers or side reactions with sensitive functional groups, this light-driven process preserves the stereochemical integrity of amino acid derivatives and other chiral building blocks. The absence of strong bases or acidic activators minimizes the formation of deletion sequences or epimerization by-products, which is critical for the synthesis of high-purity peptide mimics and complex drug molecules. Furthermore, the catalytic nature of the reagents means that metal residues can be kept to trace levels, simplifying the compliance with stringent regulatory limits for heavy metals in final drug substances. The robustness of the catalytic system against moisture and oxygen, when managed under inert atmosphere, further contributes to a clean reaction profile with minimal side-product formation.
How to Synthesize Amide Intermediates Efficiently
To implement this cutting-edge amidation protocol effectively, operators must adhere to precise procedural guidelines regarding catalyst loading, light intensity, and atmospheric control to maximize yield and reproducibility. The process begins with the careful weighing of the carboxylic acid substrate, the iridium photocatalyst at 1 mol% loading, triphenylphosphine as the deoxidizer, and the cobalt catalyst at 5 mol% loading into a suitable reaction vessel. Detailed standardized synthetic steps for scaling this reaction from milligram to kilogram quantities are provided in the guide below, ensuring that technical teams can replicate the high efficiency reported in the patent examples.
- Prepare the reaction mixture by weighing the R-substituted carboxylic acid, the photocatalyst [Ir(dF(CF3)ppy)2(dtbbpy)]PF6 (1 mol%), triphenylphosphine (1.0 equiv), and the metal complex catalyst Co(dmgH)(dmgH2)Cl2 (5 mol%) into a reaction vessel.
- Seal the vessel, evacuate the gas three times through a vacuum line, and add dichloromethane solvent under a nitrogen inert atmosphere to ensure oxygen-free conditions.
- Slowly add the R1', R2'-substituted amine to the mixture, then place the reaction under blue LED irradiation at room temperature with stirring (1200 r/min) for 3 to 18 hours until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic amidation technology offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing operations. By eliminating the requirement for expensive stoichiometric coupling reagents, the raw material costs associated with amide production are significantly reduced, while the simplified purification process lowers solvent consumption and waste disposal expenses. The mild reaction conditions, which operate at room temperature under blue light, reduce energy consumption compared to thermal methods that require heating or cooling, contributing to a more sustainable and cost-effective manufacturing footprint. Additionally, the use of stable and commercially available catalysts ensures a reliable supply chain for key reagents, mitigating the risk of production delays caused by the scarcity of specialized coupling agents.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling agents such as HATU or EDC removes a major cost driver from the bill of materials, as these reagents are often expensive and required in excess amounts. Furthermore, the reduction in chemical waste generated per kilogram of product lowers the environmental compliance costs and waste treatment fees associated with large-scale production. The simplified workup procedure, which avoids complex salt removal steps, reduces the volume of solvents and silica gel required for purification, leading to substantial operational savings. Overall, the shift to a catalytic process enhances the economic viability of producing high-value amide intermediates for the pharmaceutical and agrochemical sectors.
- Enhanced Supply Chain Reliability: The reliance on robust and stable catalysts like the iridium and cobalt complexes described in the patent ensures a consistent and dependable supply of critical reaction components. Unlike sensitive coupling reagents that may have limited shelf lives or require cold chain logistics, these metal complexes offer greater stability, simplifying inventory management and reducing the risk of spoilage. The broad substrate scope of the method allows for the flexible sourcing of various carboxylic acid and amine building blocks, enabling procurement teams to optimize their supplier base for cost and availability. This flexibility is crucial for maintaining continuous production schedules and meeting the dynamic demands of the global pharmaceutical market.
- Scalability and Environmental Compliance: The successful demonstration of this method on gram-scale reactions in the patent indicates a clear pathway for commercial scale-up to multi-kilogram and ton-level production without fundamental changes to the process chemistry. The green chemistry attributes of the method, including high atom economy and reduced hazardous waste generation, align perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The ability to synthesize complex amides under mild conditions also reduces the safety risks associated with exothermic reactions or the handling of hazardous activating agents, fostering a safer working environment. Consequently, this technology supports the long-term strategic objectives of chemical manufacturers aiming to expand capacity while minimizing their environmental impact.
Frequently Asked Questions (FAQ)
The following questions and answers address common technical and commercial inquiries regarding the implementation of this iridium-cobalt dual-catalytic amidation method in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in patent CN112778238A, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into existing manufacturing workflows for improved efficiency and product quality.
Q: What are the primary advantages of this photocatalytic amidation method over traditional coupling agents?
A: Unlike traditional methods requiring stoichiometric amounts of expensive coupling agents like EDC or HATU which generate significant waste, this method utilizes catalytic amounts of iridium and cobalt complexes with triphenylphosphine, significantly improving atom economy and simplifying downstream purification processes.
Q: Does this method support the synthesis of complex peptide structures?
A: Yes, the patent explicitly demonstrates compatibility with natural amino acids and protected amino groups, allowing for the efficient preparation of peptide products and complex drug molecule derivatives under mild room temperature conditions without racemization risks associated with harsh activation.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction proceeds under mild conditions using dichloromethane as the solvent, blue LED light irradiation, and an inert nitrogen atmosphere at room temperature, typically requiring 3 to 18 hours depending on the substrate reactivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Intermediates Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like this photocatalytic amidation method can be seamlessly transferred to large-scale manufacturing. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of amide intermediate meets the highest international standards required by top-tier pharmaceutical companies. We understand the critical importance of supply continuity and cost efficiency, and our technical team is adept at optimizing reaction parameters to maximize yield and minimize impurities for complex molecular structures. By leveraging our deep expertise in catalytic processes and green chemistry, we help our partners accelerate their drug development timelines while maintaining full regulatory compliance.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis for your specific amide synthesis projects. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how this advanced catalytic method can enhance your supply chain performance. Partner with us to unlock the full potential of this cutting-edge technology and secure a competitive advantage in the global market for high-purity pharmaceutical intermediates.
