Revolutionizing Renin Inhibitor Synthesis: Advanced Metathesis Strategies for Commercial Scalability
The pharmaceutical industry continuously seeks more efficient pathways to synthesize complex active pharmaceutical ingredients (APIs), particularly for cardiovascular treatments where demand remains robust. Patent CN101679178A introduces a groundbreaking methodology for the synthesis of intermediates crucial for renin inhibitors, most notably Aliskiren. This technology leverages advanced olefin metathesis strategies to construct the challenging C-8 carbon backbone with exceptional precision. By shifting from traditional linear syntheses to a convergent approach, this patent addresses critical bottlenecks in purity and scalability. For global procurement teams and R&D directors, understanding this technological shift is vital for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory standards. The disclosed process utilizes ruthenium-based catalysts to forge carbon-carbon bonds with high stereoselectivity, offering a pathway that drastically simplifies the manufacturing landscape for high-value antihypertensive drugs.
The significance of this innovation lies in its ability to manage the four chiral centers inherent to the Aliskiren molecule. Traditional routes often struggle with the accumulation of impurities and the difficulty of separating diastereomers at late stages. In contrast, the metathesis-driven approach described in the patent establishes the core skeleton early, allowing for more effective purification and quality control. This aligns perfectly with the industry's drive towards cost reduction in API manufacturing by minimizing waste and maximizing atom economy. As we delve deeper into the technical specifics, it becomes clear that this methodology represents a substantial leap forward in process chemistry, providing a robust foundation for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of renin inhibitor intermediates has been plagued by lengthy linear sequences that suffer from low overall yields and poor stereochemical control. Conventional methods often rely on multiple protection and deprotection steps, which not only increase the material cost but also extend the production timeline significantly. The accumulation of byproducts in these linear routes creates a complex impurity profile that is difficult to purge, posing risks to the final drug safety profile. Furthermore, the reliance on stoichiometric chiral auxiliaries in older methods generates substantial chemical waste, conflicting with modern environmental compliance standards. These inefficiencies translate directly into higher costs and longer lead times, creating vulnerabilities in the commercial scale-up of complex organic molecules. Supply chain managers often face difficulties in sourcing high-purity precursors when the synthesis route is overly sensitive to minor variations in reaction conditions.
The Novel Approach
The novel approach detailed in CN101679178A circumvents these issues by employing a convergent olefin metathesis strategy. Instead of building the molecule one carbon at a time, this method couples two identical or similar C-5 fragments to instantly generate the C-8 backbone. This dimerization step is catalyzed by highly active ruthenium carbene complexes, which tolerate a wide range of functional groups without the need for extensive protection. The result is a dramatic reduction in the number of unit operations required to reach the key intermediate. By establishing the central double bond early, the process allows for precise control over the geometric isomerism (E/Z ratio), which is critical for the subsequent asymmetric hydrogenation steps. This streamlined workflow enhances process robustness, making it easier to transfer from the laboratory to commercial scale-up of complex pharmaceutical intermediates while maintaining consistent quality.
Mechanistic Insights into Ruthenium-Catalyzed Olefin Metathesis
The core of this technological advancement is the ruthenium-catalyzed cross-metathesis reaction, which serves as the pivotal step in assembling the molecular architecture. The mechanism involves the formation of a metallacyclobutane intermediate through the [2+2] cycloaddition of the ruthenium carbene species with the terminal olefins of the substrate. This highly reactive intermediate subsequently undergoes cycloreversion to release ethylene and generate a new ruthenium alkylidene species, which propagates the catalytic cycle. The patent highlights the use of second-generation Grubbs catalysts, which feature N-heterocyclic carbene (NHC) ligands that significantly enhance thermal stability and catalytic activity. These ligands prevent catalyst decomposition under the reaction conditions, ensuring high turnover numbers and minimizing the residual metal content in the final product. Such mechanistic efficiency is paramount for R&D directors focused on high-purity pharmaceutical intermediates, as it reduces the burden on downstream purification processes.
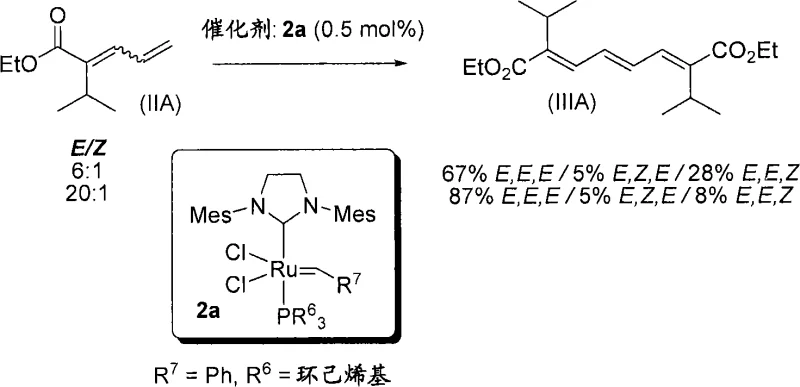
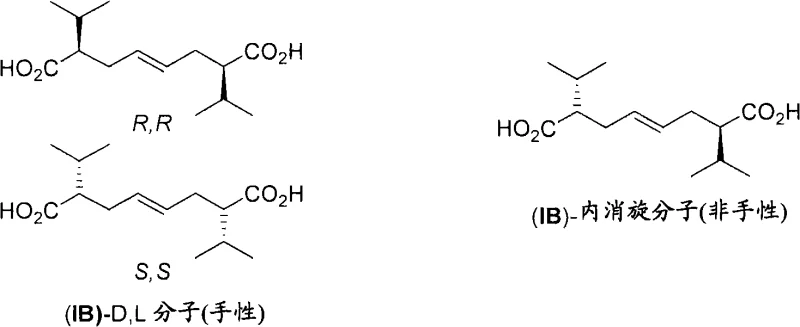
Following the construction of the triene skeleton, the process employs a sophisticated asymmetric hydrogenation step to install the necessary chiral centers. This transformation utilizes specialized ruthenium complexes bearing BoPhoz ligands, which are ferrocenyl-based diphosphines designed to induce high enantioselectivity. The catalyst differentiates between the facial biases of the olefinic bonds, selectively reducing the external double bonds while leaving the internal olefin intact or controlling its geometry. This chemoselectivity is achieved through careful tuning of the steric and electronic properties of the ligand environment around the metal center. The result is a high ratio of the desired dl-diastereomer over the meso-form, as evidenced by the NMR data provided in the patent. This level of stereocontrol eliminates the need for costly and yield-loss-inducing resolution steps later in the synthesis, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring a more sustainable manufacturing process.
How to Synthesize Aliskiren Intermediate Efficiently
Implementing this synthesis route requires a precise understanding of the reaction parameters to maximize yield and selectivity. The process begins with the preparation of the C-5 diene precursor, typically achieved through aldol condensation followed by elimination to ensure the correct olefin geometry. Once the precursor is secured, the cross-metathesis is conducted under inert atmosphere using degassed solvents to prevent catalyst oxidation. The reaction temperature and catalyst loading are optimized to balance reaction rate with selectivity, often requiring fine-tuning based on the specific scale of operation. After the metathesis step, the resulting triene is subjected to asymmetric hydrogenation under controlled pressure, where the choice of base and solvent plays a critical role in the stereochemical outcome. The detailed standardized synthesis steps see below guide.
- Prepare the C-5 diene precursor via aldol condensation of ketones with acrolein, followed by elimination to establish the terminal olefin geometry.
- Execute cross-metathesis using a second-generation Grubbs catalyst to dimerize the diene precursor, forming the central C-8 triene skeleton with high E-selectivity.
- Perform chemoselective asymmetric hydrogenation using BoPhoz-Ruthenium complexes to reduce external double bonds while establishing critical chiral centers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metathesis-based technology offers tangible strategic benefits beyond mere chemical elegance. The convergent nature of the synthesis significantly reduces the dependency on long chains of specialized raw materials, thereby mitigating supply chain risks associated with single-source vendors. By consolidating multiple synthetic steps into fewer, high-yielding transformations, manufacturers can achieve substantial cost savings in terms of labor, energy, and solvent consumption. The robustness of the ruthenium catalysts also means that the process is less susceptible to batch-to-batch variability, ensuring a consistent supply of quality material. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturing. Furthermore, the improved impurity profile simplifies quality assurance protocols, reducing the time and resources spent on analytical testing and release procedures.
- Cost Reduction in Manufacturing: The elimination of multiple protection-deprotection sequences and the reduction in total step count directly translate to lower operational expenditures. By utilizing a convergent strategy, the process minimizes the loss of valuable chiral materials that often occurs in linear syntheses. The high efficiency of the metathesis catalyst allows for lower loading levels, reducing the cost associated with precious metal recovery and disposal. Additionally, the simplified workup procedures decrease the volume of waste generated, leading to lower environmental compliance costs. These factors collectively contribute to a more economically viable production model without compromising on the quality of the high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as simple ketones and acrolein derivatives, are commodity chemicals available from multiple global suppliers. This diversification of the raw material base reduces the risk of supply disruptions caused by geopolitical issues or manufacturer-specific problems. The robustness of the catalytic system ensures that production can be scaled up rapidly to meet surges in demand without the need for extensive process re-validation. This flexibility allows supply chain managers to respond more agilely to market dynamics, ensuring that the reliable pharmaceutical intermediates supplier can meet delivery commitments consistently. The stability of the intermediates also facilitates easier storage and transportation, further enhancing logistical efficiency.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plants to commercial reactors. The use of catalytic amounts of reagents rather than stoichiometric quantities aligns with green chemistry principles, reducing the overall environmental footprint of the manufacturing process. The reduction in solvent usage and waste generation simplifies the management of effluent treatment systems, ensuring compliance with increasingly stringent environmental regulations. This sustainability aspect is becoming a key differentiator for procurement teams evaluating potential partners, as it future-proofs the supply chain against regulatory changes. The ability to produce high-purity pharmaceutical intermediates with minimal environmental impact adds significant value to the end product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific pain points identified in the background section of the patent and the beneficial effects demonstrated in the examples. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers provide clarity on the practical implications of the chemistry, focusing on scalability, purity, and regulatory alignment. This information is essential for making informed decisions about technology transfer and vendor qualification.
Q: What is the primary advantage of using olefin metathesis for Aliskiren intermediates?
A: Olefin metathesis allows for a convergent synthesis strategy, significantly reducing the number of linear steps compared to traditional methods. This approach utilizes readily available starting materials and robust ruthenium catalysts to construct the complex C-8 backbone efficiently, enhancing overall process scalability and yield consistency.
Q: How is stereochemical control achieved in this synthesis route?
A: Stereochemical integrity is maintained through a combination of E-selective cross-metathesis and subsequent asymmetric hydrogenation. The use of specialized BoPhoz-Ruthenium catalysts ensures high diastereoselectivity during the reduction of the triene intermediate, effectively distinguishing between dl and meso forms to produce the desired enantiomerically pure acid.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent describes conditions compatible with commercial scale-up, including the use of stable catalysts that function at moderate temperatures and pressures. The ability to recycle catalysts and the robustness of the metathesis step against functional group interference make it highly viable for manufacturing high-purity pharmaceutical intermediates at multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aliskiren Intermediate Supplier
The technological potential of the olefin metathesis route for renin inhibitor intermediates is immense, offering a clear path to more efficient and sustainable manufacturing. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the highest industry standards. Our commitment to quality and process optimization makes us an ideal partner for companies seeking to secure their supply chain for critical cardiovascular medications.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner. By leveraging our expertise in complex organic synthesis, we can help you achieve your goals for cost efficiency and supply security. Let us work together to bring high-quality renin inhibitor intermediates to the market faster and more effectively.
