Advanced Synthetic Route for Aliskiren Intermediate: Commercial Scale-Up and Cost Efficiency
Introduction to the Novel Synthesis of Aliskiren Intermediate
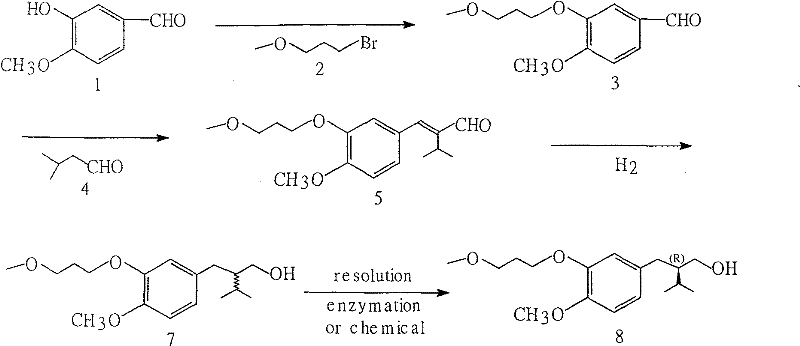
The development of efficient synthetic routes for antihypertensive agents remains a critical priority for the global pharmaceutical industry, particularly for blockbuster drugs like Aliskiren. As detailed in patent CN101284769B, a groundbreaking methodology has been established for the preparation of the main intermediate compound, (2R)-3-methyl-2-[4-methoxy-3-(3-methoxypropoxy) benzyl]-1-butanol. This innovation addresses the longstanding challenges associated with the commercial production of renin inhibitors, offering a pathway that drastically simplifies the chemical architecture required to access this vital scaffold. By shifting away from complex chiral pool strategies towards a concise racemic synthesis followed by enzymatic resolution, this technology promises to redefine the economic landscape of API manufacturing. For R&D directors and procurement specialists alike, understanding the nuances of this three-step sequence is essential for securing a competitive supply chain. The following analysis dissects the technical merits and commercial implications of this proprietary process, highlighting its potential to serve as a reliable pharmaceutical intermediate supplier solution for the next generation of cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Aliskiren intermediates was plagued by significant technical and economic hurdles that hindered efficient commercial scale-up. Existing literature describes routes relying heavily on chiral induction agents, which not only inflate raw material costs but also introduce complex purification burdens to remove residual chiral auxiliaries. Other documented pathways necessitate the use of precious metal catalysts, specifically rhodium complexes containing chiral phosphine ligands, which are notoriously difficult to prepare and often yield only moderate enantiomeric excess (ee) values ranging from 90% to 95%. Furthermore, certain legacy methods require cryogenic conditions utilizing strong bases like LDA and expensive alkylating agents such as isopropyl iodide, creating severe safety risks and operational bottlenecks in a plant setting. These conventional approaches often demand multi-step sequences just to generate the starting materials, compounding the overall yield loss and extending the production lead time significantly. Consequently, the industry has faced a persistent need for a more robust, cost-effective, and scalable alternative that does not compromise on stereochemical purity.
The Novel Approach
The methodology disclosed in the patent presents a paradigm shift by utilizing a streamlined three-step synthesis to generate the racemic intermediate, followed by a highly selective enzymatic resolution. The process initiates with the etherification of commercially abundant 3-hydroxy-4-methoxybenzaldehyde using 3-methoxypropyl bromide, a reaction that proceeds efficiently under mild basic conditions. This is followed by a condensation with isovaleraldehyde to construct the carbon skeleton, and finally, a catalytic hydrogenation step to saturate the olefinic bond. Unlike previous routes, this approach avoids expensive transition metals in the main chain construction, opting instead for recyclable catalysts like Raney Nickel or Palladium on Carbon. The final resolution step employs immobilized lipase, ensuring high optical purity without the environmental burden of heavy metal waste. This strategic simplification transforms the manufacturing profile from a complex, low-yield endeavor into a high-efficiency operation suitable for mass production.
Mechanistic Insights into the Etherification and Enzymatic Resolution
The core of this synthetic strategy lies in the precise control of reaction conditions during the initial etherification and the final biocatalytic step. The etherification of 3-hydroxy-4-methoxybenzaldehyde with 3-methoxypropyl bromide operates via a classic nucleophilic substitution mechanism, where the phenolic oxygen attacks the alkyl halide in the presence of a base such as anhydrous potassium carbonate or sodium carbonate. The patent specifies a molar ratio of 1:1 to 1.2 between the aldehyde and the bromide, optimizing reagent usage while driving the reaction to completion in solvents like acetone or acetonitrile under reflux. This step is critical as it installs the methoxypropoxy side chain, a defining structural feature of the Aliskiren molecule, with yields reported as high as 95% in optimized embodiments. The subsequent condensation with isovaleraldehyde utilizes an aldol-type mechanism in an aqueous ethanol medium, facilitated by sodium hydroxide at temperatures between 70°C and 80°C, effectively building the branched alkyl chain required for the final drug structure.
Perhaps the most sophisticated aspect of this process is the enzymatic resolution which guarantees the stringent purity specifications required for pharmaceutical intermediates. Following the catalytic hydrogenation which reduces the conjugated double bond to yield the racemic alcohol, the mixture is subjected to kinetic resolution using immobilized lipase TILM. In this biocatalytic transformation, the enzyme selectively acylates one enantiomer using allyl acetate as the acylating agent in a methyl tert-butyl ether (MTBE) solvent system. This selectivity allows for the isolation of the unreacted (R)-enantiomer with an enantiomeric excess (ee) exceeding 99%, a level of purity that is difficult to achieve through traditional chemical resolution methods. The ability to recycle the solvent and the immobilized enzyme further enhances the green chemistry profile of this route, minimizing waste generation and aligning with modern environmental compliance standards for fine chemical manufacturing.
How to Synthesize (2R)-3-methyl-2-[4-methoxy-3-(3-methoxypropoxy) benzyl]-1-butanol Efficiently
Implementing this synthetic route requires careful attention to the specific reaction parameters outlined in the patent embodiments to ensure maximum yield and purity. The process begins with the etherification step where temperature control and moisture exclusion are vital to prevent hydrolysis of the alkyl bromide. Following the isolation of the etherified aldehyde, the condensation reaction must be monitored to prevent over-reaction or polymerization, typically achieved by maintaining the pH and temperature within the narrow 70-80°C window. The hydrogenation step offers flexibility in catalyst choice, with both Raney Ni and Pd/C demonstrating high efficacy, though pressure and temperature conditions vary slightly between embodiments (e.g., 10-15 kg pressure). Finally, the enzymatic resolution demands precise stoichiometry of the acylating agent and sufficient reaction time (approximately 48 hours) to reach equilibrium and maximize the recovery of the desired chiral alcohol. For a detailed breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- Etherify 3-hydroxy-4-methoxybenzaldehyde with 3-methoxypropyl bromide using a base like potassium carbonate in acetone or acetonitrile.
- Condense the resulting aldehyde with isovaleraldehyde in an aqueous ethanol solution with sodium hydroxide at 70-80°C.
- Perform catalytic hydrogenation using Pd/C or Raney Ni, followed by enzymatic resolution with immobilized lipase to obtain the chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this novel synthetic route offers transformative benefits that directly address the pain points of cost volatility and material availability in API manufacturing. By relying on commodity chemicals such as 3-hydroxy-4-methoxybenzaldehyde and isovaleraldehyde, the process decouples production from the fluctuating markets of specialized chiral reagents or precious metal catalysts. This shift to readily available feedstocks ensures a more stable supply chain, reducing the risk of production stoppages due to raw material shortages. Furthermore, the reduction in synthetic steps from complex multi-stage sequences to a concise three-step racemic synthesis significantly lowers the operational overhead, including labor, energy consumption, and equipment occupancy time. These factors collectively contribute to a substantial reduction in the cost of goods sold (COGS), making the final API more competitive in the global marketplace without sacrificing quality.
- Cost Reduction in Manufacturing: The elimination of expensive rhodium catalysts and chiral induction agents removes a major cost driver from the production budget. Additionally, the use of recyclable catalysts like Raney Nickel and the ability to recover solvents like MTBE and ethanol further drive down variable costs. The high yields reported in the patent embodiments (up to 96% for hydrogenation) minimize material waste, ensuring that every kilogram of raw material contributes effectively to the final output. This efficiency translates into significant margin improvements for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Sourcing simplicity is a key advantage, as the primary starting materials are bulk chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency, a critical factor for supply chain heads managing long-term contracts. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures or ultra-high pressures, also means that the process can be executed in a wider range of manufacturing facilities, increasing overall production capacity and flexibility.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor types and separation techniques familiar to chemical engineers. The avoidance of toxic heavy metals in the main synthetic line simplifies waste treatment and regulatory compliance, reducing the environmental footprint of the manufacturing site. The enzymatic resolution step operates under mild conditions, further lowering energy requirements and aligning with sustainability goals that are increasingly important for corporate social responsibility reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or process optimization. We encourage stakeholders to review these points carefully to appreciate the full scope of the innovation.
Q: What are the key advantages of this new synthetic route compared to prior art?
A: The novel route eliminates the need for expensive chiral induction agents or rhodium catalysts found in previous methods. It utilizes readily available raw materials like 3-hydroxy-4-methoxybenzaldehyde and reduces the synthesis of the racemate to just three robust steps, significantly lowering production costs and complexity.
Q: How is high optical purity achieved in the final step?
A: High optical purity (ee > 99%) is achieved through a biological resolution method using immobilized lipase (Lipase TILM). This enzymatic process selectively acylates one enantiomer, allowing for the isolation of the desired (2R)-isomer with exceptional stereochemical control without requiring complex chromatographic separations.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It relies on standard unit operations such as reflux, filtration, and catalytic hydrogenation in common solvents like ethanol and acetone. The catalysts used, such as Raney Ni or Pd/C, are recoverable and recyclable, making the process economically viable for metric ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aliskiren Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and high-quality supply of critical pharmaceutical intermediates like the Aliskiren precursor. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for FDA-approved drug synthesis. Our expertise in enzymatic resolution and catalytic hydrogenation positions us as a leader in delivering complex chiral intermediates with consistent quality and reliability.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and reduce your time to market.
