Revolutionizing Caronic Anhydride Production: A Safe, Scalable Route for Global Antiviral Supply Chains
Revolutionizing Caronic Anhydride Production: A Safe, Scalable Route for Global Antiviral Supply Chains
The global demand for high-purity antiviral intermediates has intensified, particularly for compounds serving as the backbone of protease inhibitors like Boceprevir and the core raw materials for Paxlovid. Patent CN115417767A introduces a transformative preparation method for Caronic Anhydride and its intermediates, addressing critical bottlenecks in existing industrial synthesis. This technical disclosure outlines a novel five-step sequence that bypasses the dangerous oxidation steps characteristic of legacy routes, utilizing a sulfur ylide-mediated cyclopropanation strategy. By shifting from hazardous permanganate oxidations to mild, room-temperature quaternization and coupling reactions, this methodology offers a robust framework for cost reduction in antiviral intermediate manufacturing. For procurement leaders and R&D directors, this patent represents a pivotal shift towards safer, more environmentally compliant production capabilities that ensure supply continuity for essential pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Caronic Anhydride has been dominated by two primary pathways, both of which present substantial hurdles for large-scale manufacturing. The first conventional route, often referred to as Route 1, initiates with the oxidation of ethyl chrysanthemate using potent oxidizing agents such as potassium permanganate (KMnO4). While chemically feasible, this approach is fraught with industrial dangers, including the handling of strong oxidants and the generation of massive quantities of manganese-containing wastewater that requires complex and costly treatment protocols. Furthermore, the starting material, ethyl chrysanthemate, is subject to market volatility and limited availability, creating a fragile supply chain foundation. The second conventional pathway involves a Corey-Chaykovsky epoxidation which, although effective, relies on foul-smelling dimethyl sulfide and necessitates subsequent oxidation steps that complicate the process flow and increase the environmental footprint.
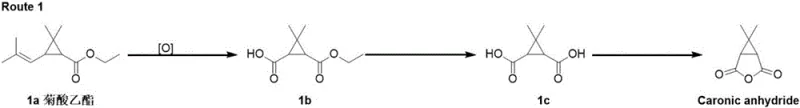
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN115417767A proposes a streamlined, oxidation-free trajectory that fundamentally reimagines the construction of the cyclopropane ring. This novel approach leverages the reactivity of tetrahydrothiophene to generate a sulfur ylide in situ, which then undergoes a highly selective cyclopropanation with ethyl prenylate. The process operates predominantly at room temperature, eliminating the need for energy-intensive heating or cryogenic cooling in the critical bond-forming steps. By avoiding strong oxidants entirely in the early stages and utilizing readily available reagents like ethyl bromoacetate, this route significantly de-risks the manufacturing process. The ability to recover and recycle tetrahydrothiophene further enhances the economic viability, making this a superior choice for establishing a reliable Caronic Anhydride supplier network capable of meeting surging global demand without the baggage of hazardous waste streams.
Mechanistic Insights into Sulfur Ylide Mediated Cyclopropanation
The core chemical innovation lies in the generation and utilization of the sulfur ylide intermediate (7d-1), which serves as the active species for constructing the strained cyclopropane motif. The mechanism initiates with a nucleophilic attack by tetrahydrothiophene on ethyl bromoacetate, forming a stable sulfonium salt (7c-1) in a polar aprotic solvent like acetone. This quaternization is exothermic but manageable, proceeding efficiently at ambient temperatures. Subsequent treatment with a base, such as aqueous sodium hydroxide in a biphasic system, deprotonates the alpha-carbon adjacent to the sulfur, generating the reactive ylide. This ylide then attacks the electron-deficient double bond of ethyl prenylate in a concerted [2+1] cycloaddition. The stereoelectronic properties of the ylide ensure high regioselectivity, minimizing the formation of unwanted isomers and simplifying downstream purification. This mechanistic elegance allows for the precise assembly of the 6,6-dimethyl-3-oxabicyclo[3.1.0]hexane skeleton with minimal by-product formation.
From an impurity control perspective, the mild reaction conditions are paramount. Traditional oxidation routes often lead to over-oxidation by-products or degradation of the sensitive ester functionalities, requiring rigorous chromatographic purification that is impractical at scale. In this new protocol, the room temperature cyclopropanation preserves the integrity of the ester groups, and the subsequent hydrolysis and cyclization steps are performed under controlled pH and thermal conditions. The use of acetic anhydride for the final dehydration ensures a clean conversion to the anhydride without introducing halogenated impurities or heavy metal residues. This results in a final product profile that consistently meets stringent purity specifications, often exceeding 98.5% purity by HPLC, which is critical for API applications where impurity thresholds are tightly regulated by health authorities.
How to Synthesize Caronic Anhydride Efficiently
The practical execution of this synthesis involves a sequential five-step protocol that balances reaction efficiency with operational simplicity. The process begins with the formation of the sulfonium salt, followed by ylide generation and cyclopropanation to build the core ring system. Subsequent hydrolysis converts the diester into the corresponding dicarboxylic acid, which is finally dehydrated to the target anhydride. Each step has been optimized to maximize yield while minimizing solvent usage and energy consumption. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures derived from the patent examples, are outlined below to guide process engineers in replicating this high-efficiency route.
- Quaternization of tetrahydrothiophene with ethyl bromoacetate in acetone at room temperature to form sulfonium salt intermediate 7c-1.
- Generation of sulfur ylide intermediate 7d-1 by treating the sulfonium salt with aqueous sodium hydroxide in a biphasic DCM/water system.
- Cyclopropanation reaction between the sulfur ylide and ethyl prenylate in dichloromethane to yield the diester intermediate 7e-1.
- Hydrolysis of the diester intermediate using sodium hydroxide followed by acidification to isolate the dicarboxylic acid intermediate 7f.
- Dehydrative cyclization of the dicarboxylic acid in acetic anhydride under reflux to afford the final Caronic Anhydride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into enhanced operational resilience and cost efficiency. By eliminating the dependency on expensive and volatile raw materials like ethyl chrysanthemate, manufacturers can stabilize their input costs and secure a more predictable supply baseline. The removal of hazardous oxidation steps not only reduces the capital expenditure required for specialized safety equipment and wastewater treatment facilities but also accelerates the regulatory approval process for new manufacturing sites. This streamlined workflow supports the commercial scale-up of complex pharmaceutical intermediates by reducing the number of unit operations and simplifying the overall process flow, thereby decreasing the potential for batch failures and production delays.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the substitution of high-cost oxidants and rare starting materials with commodity chemicals like ethyl bromoacetate and tetrahydrothiophene. The ability to recover and recycle the tetrahydrothiophene solvent significantly lowers the net consumption of reagents, driving down the variable cost per kilogram. Furthermore, the avoidance of cryogenic conditions and high-pressure oxidation reactors reduces utility costs associated with heating, cooling, and pressure management. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available, non-restricted raw materials that are not subject to the same geopolitical or seasonal constraints as natural product-derived precursors. The robustness of the room-temperature reactions means that production is less susceptible to disruptions caused by utility fluctuations or equipment maintenance issues associated with extreme condition processing. This reliability ensures that downstream API manufacturers can maintain consistent inventory levels, reducing lead time for high-purity Caronic Anhydride deliveries and mitigating the risk of stockouts during periods of heightened demand for antiviral therapeutics.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles, facilitating easier permitting and compliance with increasingly strict environmental regulations. The reduction in hazardous waste generation, particularly the elimination of heavy metal sludge from permanganate oxidation, simplifies waste disposal logistics and lowers associated fees. The process is inherently scalable, as the exothermic nature of the reactions is mild and easily managed in large reactors, allowing for a seamless transition from pilot plant to multi-ton commercial production without the need for significant process re-engineering or safety overrides.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Caronic Anhydride synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and scalability factors. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Traditional Route 1 relies on hazardous potassium permanganate oxidation of ethyl chrysanthemate, posing significant industrial safety risks and wastewater treatment challenges. The novel method described in CN115417767A eliminates the need for strong oxidants in the early stages, utilizing mild room temperature conditions and recyclable tetrahydrothiophene, drastically reducing operational hazards and environmental burden.
Q: What are the yield and purity benchmarks for this Caronic Anhydride process?
A: According to the patent data, the optimized embodiment achieves a final isolated yield of approximately 80.6% for Caronic Anhydride with an HPLC purity of 98.5%. Key intermediates like the sulfonium salt and sulfur ylide are generated in high crude yields (up to 100%), ensuring efficient material throughput and minimizing waste generation throughout the multi-step sequence.
Q: Is the tetrahydrothiophene reagent recoverable in this process?
A: Yes, a critical economic advantage of this sulfur ylide mediated pathway is the recoverability of tetrahydrothiophene. During the workup of the cyclopropanation step, the solvent and the released tetrahydrothiophene can be distilled off and recycled, significantly lowering the raw material costs and enhancing the overall sustainability profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caronic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of critical antiviral intermediates like Caronic Anhydride. Our technical team has thoroughly analyzed the pathway described in CN115417767A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to life. We are equipped with rigorous QC labs and advanced process engineering capabilities to ensure that every batch meets stringent purity specifications, delivering a product that is ready for immediate integration into your API synthesis workflows without the need for additional purification.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this oxidation-free synthesis can drive value and security for your antiviral drug production programs.
