Revolutionizing Caronic Anhydride Production: A Green Catalytic Route for Scalable Pharmaceutical Manufacturing
Revolutionizing Caronic Anhydride Production: A Green Catalytic Route for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, particularly for critical intermediates used in antiviral therapies. A groundbreaking patent, CN114702471A, filed in July 2022, introduces a highly efficient and environmentally benign method for synthesizing caronic anhydride, a pivotal building block for drugs like Boceprevir and Paroxetine. This innovation addresses the longstanding challenges of traditional oxidation methods by utilizing a novel gel cobalt salt catalyst coupled with ambient air as the oxidant. For R&D directors and procurement strategists, this technology represents a significant leap forward, offering a pathway to reduce hazardous waste while simultaneously boosting reaction yields to over 95 percent. The ability to replace dangerous reagents like ozone and potassium permanganate with simple air oxidation not only enhances operational safety but also streamlines the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of caronic anhydride has relied heavily on aggressive oxidation techniques that pose substantial risks to both personnel and the environment. Traditional protocols typically employ ozone (O3) or potassium permanganate (KMnO4) to cleave double bonds and oxidize side chains on cyclopropane precursors. As illustrated in the reaction schemes below, these methods are fraught with inefficiencies; ozone generation requires specialized, energy-intensive equipment and carries a high risk of explosion due to the formation of unstable ozonides. Furthermore, permanganate oxidation generates vast quantities of manganese dioxide sludge, a heavy metal waste that complicates downstream processing and imposes severe environmental disposal costs. These legacy processes often suffer from poor selectivity, leading to lower yields and necessitating complex, solvent-intensive purification steps to remove trace impurities that could compromise the safety of the final API.
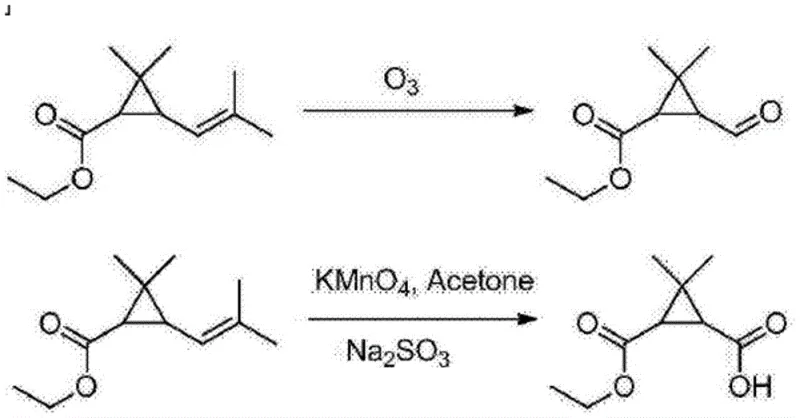
The Novel Approach
In stark contrast, the methodology disclosed in patent CN114702471A leverages a sophisticated gel cobalt salt catalyst to facilitate aerobic oxidation under mild conditions. This transformative approach utilizes molecular oxygen from the air as the sole oxidant, effectively eliminating the need for hazardous chemical oxidants. The reaction proceeds smoothly at temperatures between 20°C and 25°C, demonstrating exceptional control over the oxidation state of the substrate. By employing this catalytic system, manufacturers can achieve conversion rates exceeding 99 percent with molar yields approaching 92 percent for the acid intermediate, which is subsequently cyclized to the anhydride. This shift not only mitigates safety risks associated with high-energy oxidants but also simplifies the workflow by allowing the intermediate acid to proceed to the next step without rigorous isolation, thereby reducing solvent consumption and processing time significantly.
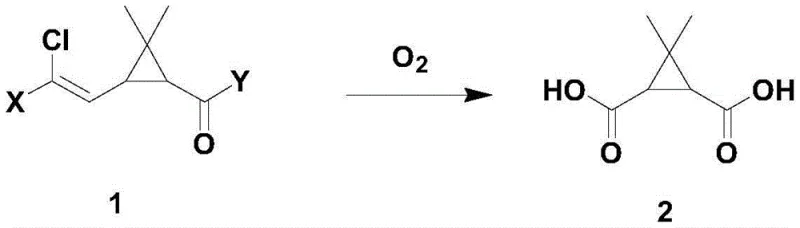
Mechanistic Insights into Gel Cobalt Salt-Catalyzed Aerobic Oxidation
The core of this technological breakthrough lies in the unique properties of the gel cobalt salt catalyst, which acts as a highly efficient mediator for electron transfer during the oxidation process. Unlike homogeneous cobalt salts that may leach or deactivate rapidly, this gel-supported variant offers a robust surface area that stabilizes the active cobalt species, allowing for sustained catalytic activity. The mechanism likely involves the activation of molecular oxygen by the cobalt center to form reactive oxygen species that selectively attack the allylic or benzylic positions of the cyclopropane derivative. This selectivity is crucial for preserving the integrity of the sensitive cyclopropane ring while oxidizing the adjacent side chain to the desired carboxylic acid functionality. The patent data highlights that the catalyst can be recycled up to 20 times without a noticeable decline in performance, suggesting a stable coordination environment that prevents metal leaching and deactivation, a common pitfall in transition metal catalysis.
Furthermore, the process design incorporates a seamless transition from oxidation to cyclization, minimizing the exposure of reactive intermediates to potential degradation pathways. Following the aerobic oxidation, the reaction mixture undergoes alkaline hydrolysis and acidification to yield 3,3-dimethyl-1,2-cyclopropane dicarboxylic acid. This crude acid is then subjected to thermal dehydration using acetic anhydride and sodium acetate at elevated temperatures (170°C) to form the final anhydride ring. The high purity of the final product, reported at 99.8 percent, indicates that the catalytic system effectively suppresses side reactions such as over-oxidation or ring-opening, which are prevalent in harsher chemical environments. This level of impurity control is vital for pharmaceutical applications, where strict regulatory limits on genotoxic impurities and heavy metals must be met.
How to Synthesize Caronic Anhydride Efficiently
The synthesis of caronic anhydride via this green route involves a streamlined two-step sequence that begins with the catalytic oxidation of readily available cyclopropane precursors such as cyhalothric acid or its esters. The process is designed for scalability, utilizing standard reactor equipment capable of handling gas-liquid reactions, making it accessible for commercial scale-up of complex pharmaceutical intermediates. The elimination of cryogenic conditions required for ozonolysis and the avoidance of heavy metal sludge from permanganate oxidation significantly lowers the barrier to entry for manufacturers seeking to optimize their production lines. While the specific stoichiometric details and workup procedures are critical for reproducibility, the general workflow emphasizes simplicity and safety, aligning with modern green chemistry principles.
- Oxidize the starting cyclopropane derivative (e.g., cyhalothric acid) using a gel cobalt salt catalyst (3-8% wt) under an air atmosphere at 20-25°C for 5-8 hours.
- Perform alkaline hydrolysis and acidification to isolate 3,3-dimethyl-1,2-cyclopropane dicarboxylic acid without intermediate purification.
- Cyclize the dicarboxylic acid with acetic anhydride and sodium acetate at 170°C, followed by recrystallization to obtain pure caronic anhydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic aerobic oxidation technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The transition from stoichiometric oxidants to a catalytic air-oxidation model fundamentally alters the cost structure of manufacturing this key intermediate. By removing the dependency on expensive and hazardous reagents like ozone generators or large quantities of potassium permanganate, the process significantly reduces raw material costs and the associated logistics of handling dangerous goods. Moreover, the simplified workup procedure, which avoids complex extraction and purification of intermediates, leads to a drastic reduction in solvent usage and waste treatment expenses, contributing to substantial cost savings in overall manufacturing operations.
- Cost Reduction in Manufacturing: The implementation of a recyclable gel cobalt salt catalyst eliminates the recurring cost of purchasing stoichiometric oxidants for every batch. Since the catalyst retains its activity over multiple cycles, the effective cost per kilogram of product is lowered considerably. Additionally, the reduction in waste generation means lower fees for hazardous waste disposal and reduced consumption of water and energy for purification processes, further enhancing the economic viability of the production line.
- Enhanced Supply Chain Reliability: Relying on air as the primary oxidant removes the supply chain vulnerabilities associated with specialized chemical reagents that may face availability fluctuations or shipping restrictions. The robustness of the catalyst, demonstrated by its stability over 20 cycles, ensures consistent production throughput without frequent interruptions for catalyst replenishment. This reliability is crucial for maintaining continuous supply of critical antiviral intermediates, especially in times of heightened global demand.
- Scalability and Environmental Compliance: The process operates under mild conditions (20-25°C) and atmospheric pressure, which simplifies the engineering requirements for scaling up from pilot to commercial production. The absence of explosive ozonides and heavy metal waste streams makes regulatory compliance significantly easier, reducing the administrative burden and risk of environmental fines. This green profile aligns perfectly with the sustainability goals of major pharmaceutical companies, facilitating smoother audits and partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the safety advantages of the new gel cobalt catalyst method over traditional ozone oxidation?
A: The new method replaces hazardous ozone and potassium permanganate with ambient air oxidation. This eliminates the risk of explosive ozonide formation and removes the generation of heavy metal manganese waste, significantly improving process safety and environmental compliance.
Q: How does the catalyst stability impact long-term production costs?
A: The patent data indicates that the novel gel cobalt salt catalyst maintains its activity without significant reduction even after 20 recycling cycles. This durability drastically reduces catalyst consumption costs and minimizes downtime associated with catalyst replacement.
Q: What is the purity profile of the caronic anhydride produced via this route?
A: The optimized synthetic route yields caronic anhydride with a purity exceeding 99.8%. The process simplifies purification by avoiding complex intermediate separations, ensuring the final product meets stringent pharmaceutical specifications for antiviral drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caronic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of caronic anhydride in the synthesis of next-generation antiviral therapeutics and are committed to delivering this intermediate with unmatched quality and consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical supply chains. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of caronic anhydride meets the highest standards required for API synthesis, leveraging advanced catalytic technologies to minimize impurities and maximize yield.
We invite you to collaborate with us to optimize your supply chain for antiviral drug production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our green manufacturing processes can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and efficient supply of this vital pharmaceutical intermediate.
