Advanced Organocatalytic Route for High-Purity Aprepitant Intermediates Manufacturing
Advanced Organocatalytic Route for High-Purity Aprepitant Intermediates Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for synthesizing complex active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in Chinese Patent CN112174909B, which discloses a novel preparation method for a critical chiral intermediate used in the synthesis of Aprepitant. Aprepitant is a highly selective Neurokinin-1 (NK1) receptor antagonist, widely utilized clinically to prevent acute and delayed nausea and vomiting associated with highly emetogenic cancer chemotherapy. The molecular architecture of Aprepitant is intricate, featuring three distinct chiral centers that must be precisely controlled to ensure therapeutic efficacy and safety. 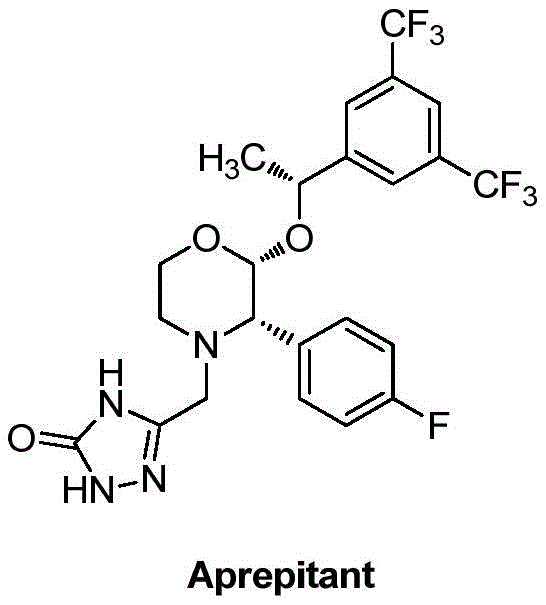 . The patent introduces a groundbreaking organocatalytic transfer hydrogenation strategy that replaces traditional, hazardous high-pressure hydrogenation methods with a mild, biomimetic approach, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
. The patent introduces a groundbreaking organocatalytic transfer hydrogenation strategy that replaces traditional, hazardous high-pressure hydrogenation methods with a mild, biomimetic approach, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the chiral morpholine core found in Aprepitant has relied heavily on asymmetric hydrogenation techniques. These conventional processes typically necessitate the use of transition metal catalysts, such as rhodium or ruthenium complexes, coupled with high-pressure hydrogen gas as the reducing agent. While effective in laboratory settings, these methods present substantial challenges for industrial scale-up. The requirement for specialized high-pressure reactors increases capital expenditure and operational complexity, introducing significant safety hazards related to hydrogen handling. Furthermore, the use of transition metals raises critical concerns regarding residual metal contamination in the final drug substance, necessitating expensive and time-consuming purification steps to meet stringent regulatory limits. The harsh reaction conditions often associated with these metal-catalyzed processes can also lead to side reactions, compromising the overall yield and optical purity of the sensitive chiral intermediate.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN112174909B leverages a sophisticated biomimetic system to achieve the same transformation under remarkably mild conditions. The core innovation lies in the use of a chiral organocatalyst, specifically a 1,4-dihydropyridine derivative functionalized with a chiral α-hydroxyphenylacetamide moiety. This catalyst facilitates a transfer hydrogenation reaction, effectively delivering hydride equivalents to the substrate without the need for external hydrogen gas. 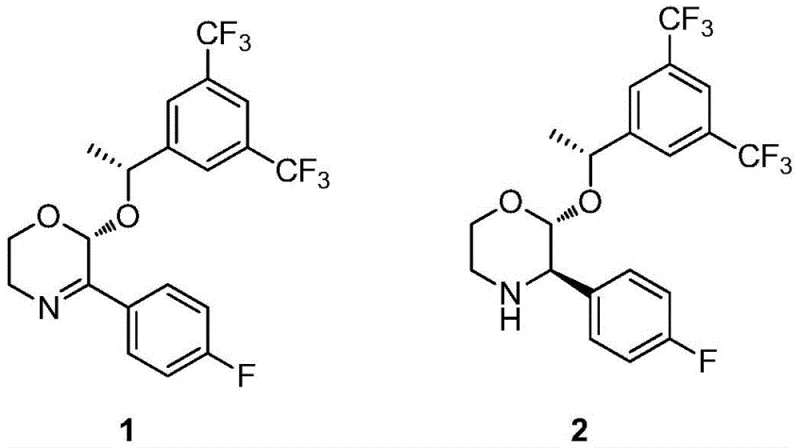 . The reaction proceeds smoothly in common organic solvents like tetrahydrofuran at temperatures around 45°C, eliminating the need for complex high-pressure equipment. This shift from metal-catalyzed high-pressure hydrogenation to organocatalytic transfer hydrogenation represents a paradigm shift in process chemistry, significantly lowering the barrier to entry for manufacturing this high-value intermediate while enhancing the environmental profile of the synthesis.
. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran at temperatures around 45°C, eliminating the need for complex high-pressure equipment. This shift from metal-catalyzed high-pressure hydrogenation to organocatalytic transfer hydrogenation represents a paradigm shift in process chemistry, significantly lowering the barrier to entry for manufacturing this high-value intermediate while enhancing the environmental profile of the synthesis.
Mechanistic Insights into Biomimetic Transfer Hydrogenation
The efficacy of this novel synthetic route is rooted in the unique structural features of the chiral catalyst, designated as Formula (I) in the patent documentation. 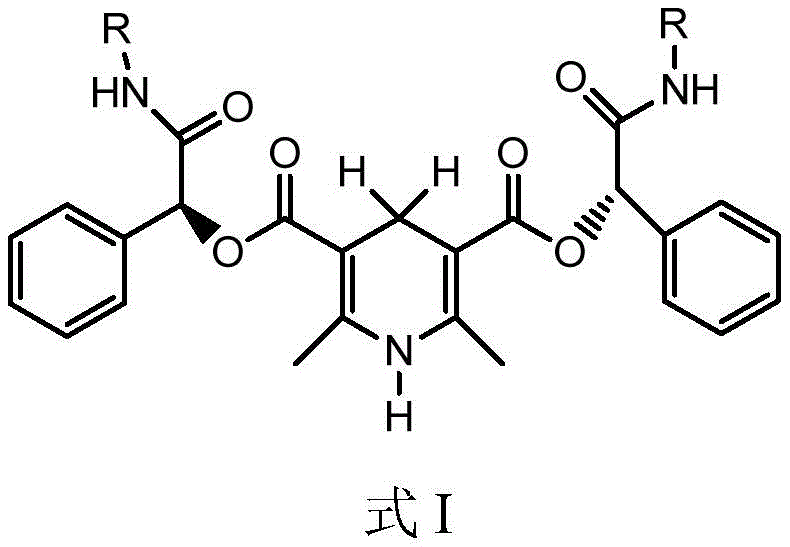 . This catalyst operates through a mechanism that mimics biological redox systems, specifically the function of NADH/NADPH coenzymes. The 1,4-dihydropyridine core serves as the hydride donor, transferring a hydride ion to the electrophilic carbon of the imine substrate (Formula 1). Crucially, the chiral information is encoded in the α-hydroxyphenylacetamide side chain attached to the dihydropyridine ring. This chiral pocket creates a sterically defined environment that directs the approach of the substrate, ensuring that the hydride transfer occurs selectively to one face of the planar imine bond. This precise stereocontrol is what allows the process to generate the desired (S)-configuration at the new chiral center with exceptional fidelity, as evidenced by the high enantiomeric excess values reported in the examples.
. This catalyst operates through a mechanism that mimics biological redox systems, specifically the function of NADH/NADPH coenzymes. The 1,4-dihydropyridine core serves as the hydride donor, transferring a hydride ion to the electrophilic carbon of the imine substrate (Formula 1). Crucially, the chiral information is encoded in the α-hydroxyphenylacetamide side chain attached to the dihydropyridine ring. This chiral pocket creates a sterically defined environment that directs the approach of the substrate, ensuring that the hydride transfer occurs selectively to one face of the planar imine bond. This precise stereocontrol is what allows the process to generate the desired (S)-configuration at the new chiral center with exceptional fidelity, as evidenced by the high enantiomeric excess values reported in the examples.
Furthermore, the reaction system incorporates a protic acid additive, such as hydrochloric acid or trifluoroacetic acid, which plays a pivotal role in activating the imine substrate. Protonation of the imine nitrogen increases the electrophilicity of the carbon atom, facilitating the nucleophilic attack by the hydride from the catalyst. The synergy between the chiral organocatalyst and the acid promoter allows the reaction to proceed rapidly and selectively at moderate temperatures. This mechanistic elegance not only ensures high conversion rates but also minimizes the formation of byproducts and racemic impurities. The ability to fine-tune the steric and electronic properties of the catalyst by varying the 'R' group on the amide nitrogen provides additional flexibility for optimizing the process for specific substrates, making this a versatile platform technology for the synthesis of complex chiral amines beyond just the Aprepitant intermediate.
How to Synthesize Aprepitant Intermediate Efficiently
The practical implementation of this technology involves a streamlined two-stage process: first, the preparation of the specialized chiral catalyst, and second, the catalytic reduction of the imine substrate. The catalyst synthesis itself is straightforward, involving the condensation of a chiral α-hydroxyphenylacetamide with a dioxinone derivative, followed by cyclization with hexamethylenetetramine and ammonium acetate. Once the catalyst is secured, the key transformation involves mixing the imine substrate, the catalyst, and an acid source in a solvent like THF. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures derived from the patent examples, are provided in the guide below to assist process chemists in replicating this high-efficiency route.
- Prepare the chiral catalyst by reacting chiral α-hydroxyphenylacetamide with 2,2,6-trimethyl-1,3-dioxin-4-one in toluene, followed by cyclization with hexamethylenetetramine and ammonium acetate.
- Mix the imine substrate (Formula 1), the prepared chiral catalyst (Formula I), and a protic acid additive in a solvent such as tetrahydrofuran.
- Stir the reaction mixture at mild temperatures (around 45°C) for approximately 8 hours, then quench, extract, and crystallize to obtain the high-purity chiral intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The transition away from transition metal catalysts and high-pressure infrastructure directly addresses several pain points associated with the sourcing and manufacturing of complex pharmaceutical intermediates. By simplifying the reaction conditions and eliminating the need for specialized high-pressure vessels, manufacturers can significantly reduce capital investment and operational overhead. This process intensification allows for more flexible production scheduling and the utilization of standard multipurpose reactors, thereby enhancing the overall agility of the supply chain. Moreover, the removal of heavy metals from the process stream simplifies downstream purification, reducing the consumption of scavengers and chromatography media, which translates into substantial cost savings in raw materials and waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, such as rhodium or ruthenium complexes, removes a significant cost driver from the bill of materials. Additionally, the avoidance of high-pressure hydrogenation equipment reduces both the initial capital expenditure for plant setup and the ongoing maintenance costs associated with safety compliance for high-pressure systems. The mild reaction conditions also imply lower energy consumption for heating and cooling compared to more vigorous traditional processes. These factors combine to create a more economically efficient manufacturing process, allowing for competitive pricing strategies in the global market for pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Reliance on precious metal catalysts often introduces supply chain vulnerabilities due to geopolitical factors and market volatility affecting metal prices. By switching to an organocatalytic system based on readily available organic building blocks, manufacturers can secure a more stable and predictable supply of critical reagents. The robustness of the reaction, which tolerates mild variations in conditions while maintaining high selectivity, further ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream API manufacturers and pharmaceutical companies.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process align perfectly with modern environmental regulations and corporate sustainability goals. The absence of toxic heavy metals simplifies waste treatment and reduces the environmental footprint of the manufacturing facility. The use of common solvents and the ability to perform the reaction at near-ambient pressures make the scale-up from laboratory to commercial production significantly safer and more straightforward. This ease of scale-up ensures that supply can be rapidly ramped up to meet surges in demand, providing a secure source of high-purity intermediates for the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational parameters and expected outcomes of the process. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their existing manufacturing workflows.
Q: How does this organocatalytic method improve upon traditional hydrogenation?
A: Unlike conventional methods requiring high-pressure hydrogen gas and transition metal catalysts, this patented process utilizes a biomimetic organic catalyst under atmospheric pressure and mild temperatures, eliminating heavy metal residues and reducing safety risks.
Q: What level of enantiomeric excess (ee) can be achieved?
A: The patent data demonstrates that this method consistently achieves excellent optical purity, with enantiomeric excess (ee) values reaching up to 99.3%, which is critical for the efficacy of the final NK1 receptor antagonist API.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids complex high-pressure equipment, uses readily available solvents like THF, and involves simple workup procedures like freezing crystallization, making it highly adaptable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aprepitant Intermediate Supplier
The technological advancements described in Patent CN112174909B represent a significant leap forward in the synthesis of chiral pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we recognize the immense potential of this organocatalytic approach to redefine the manufacturing landscape for high-value compounds like the Aprepitant intermediate. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that ensure every batch meets the highest international standards. We are committed to leveraging such innovative green chemistry solutions to deliver superior products to our global clientele.
We invite pharmaceutical companies and contract manufacturers to explore the possibilities of this advanced synthesis route with us. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By partnering with us, you gain access to not just a product, but a comprehensive solution that optimizes your supply chain efficiency. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and to discuss how we can support your long-term strategic goals in the competitive pharmaceutical marketplace.
