Revolutionizing Heterocyclic Intermediate Production via Solvent-Free Cyclization Technology
Introduction to Advanced Solvent-Free Heterocyclic Synthesis
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more efficient synthetic methodologies, driven by both regulatory pressure and the economic imperative for cost reduction. A pivotal advancement in this domain is detailed in Chinese Patent CN102134224A, which discloses a robust, solvent-free preparation method for a broad spectrum of heterocyclic compounds, including thiazoles, imidazoles, and oxazoles. This technology represents a significant departure from conventional solvated protocols, offering a streamlined pathway to high-purity intermediates that are critical for the pharmaceutical and agrochemical industries. By eliminating the need for bulk organic solvents such as ethanol, DMF, or acetonitrile, this process not only mitigates environmental hazards but also fundamentally simplifies the downstream processing workflow. The core innovation lies in the utilization of alpha-halogenated compounds and nitrogen-containing reagents in a neat reaction system, where thermal energy drives the cyclization to completion with remarkable efficiency.
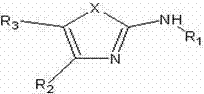
For R&D directors and process chemists, the implications of this patent are profound, as it addresses the perennial challenge of balancing reaction yield with environmental sustainability. The general formula presented in the patent encompasses a versatile array of structures where X can be oxygen, sulfur, or nitrogen, allowing for the synthesis of diverse heterocyclic cores essential for drug discovery. The method requires that at least one of the reactants be in a liquid state to facilitate mixing, while the other can be solid or liquid, ensuring flexibility in substrate selection. This adaptability makes the technology a powerful tool for a reliable pharmaceutical intermediate supplier aiming to diversify their portfolio with eco-friendly offerings. The reaction proceeds under stirring conditions at temperatures ranging from 60°C to 120°C, followed by a straightforward workup involving water washing and drying, which yields target compounds with purity often exceeding 98% without the need for further purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of heterocyclic scaffolds like thiazoles and imidazoles has relied heavily on solvent-assisted reactions, a practice that introduces significant inefficiencies into the manufacturing supply chain. Classical methods, such as the Hantzsch thiazole synthesis, typically require refluxing conditions in solvents like ethanol for extended periods, often upwards of 12 hours, to achieve acceptable conversion rates. These solvated systems necessitate complex post-treatment operations, including solvent recovery via distillation, which is both energy-intensive and capital-heavy. Furthermore, the use of polar aprotic solvents like DMF poses severe challenges regarding residual solvent limits in final API products, requiring rigorous and costly analytical testing to ensure compliance with ICH guidelines. The generation of large volumes of organic waste also creates a substantial burden on waste management infrastructure, increasing the overall carbon footprint of the chemical process. For procurement managers, these factors translate into higher raw material costs, longer lead times due to complex purification, and increased liability associated with hazardous waste disposal.
The Novel Approach
In stark contrast, the novel solvent-free approach outlined in the patent data offers a disruptive solution that bypasses these traditional bottlenecks by utilizing a neat reaction system. By heating the reactants directly to temperatures between 60°C and 120°C, the reaction mixture often undergoes a "curing" or solidification phenomenon, which serves as a visual indicator of reaction progress and product formation. This phase change effectively drives the equilibrium towards the product side, resulting in high yields (often 80-95%) within a drastically reduced timeframe of merely 10 to 40 minutes. The elimination of organic solvents means that the workup is reduced to a simple addition of water, filtration, and drying, removing the need for expensive solvent recovery units. This simplicity not only accelerates the production cycle but also enhances the safety profile of the operation by removing flammable vapors from the reactor environment. For companies seeking cost reduction in API manufacturing, this technology provides a clear pathway to lower operational expenditures through reduced utility consumption and waste treatment fees.
Mechanistic Insights into Solvent-Free Thermal Cyclization
From a mechanistic standpoint, the solvent-free cyclization of alpha-halo carbonyls with nitrogen nucleophiles operates through a concerted nucleophilic substitution and ring-closure sequence that is kinetically favored in the absence of solvation shells. In traditional solvated media, solvent molecules often stabilize the transition states and intermediates through hydrogen bonding or dipole interactions, which can sometimes retard the reaction rate or lead to competing side reactions such as hydrolysis. However, in the neat melt phase described in this patent, the high local concentration of reactants significantly increases the frequency of effective collisions, thereby accelerating the initial nucleophilic attack of the nitrogen species on the alpha-carbon. The subsequent intramolecular cyclization is facilitated by the thermal energy provided, which overcomes the activation barrier for ring closure without the damping effect of a solvent cage. The observed solidification of the reaction mass suggests that the product precipitates out of the melt as it forms, which acts as a thermodynamic sink, preventing reverse reactions and driving the conversion to near completion. This phenomenon is particularly advantageous for maintaining high selectivity, as the product is physically removed from the reactive environment once formed.
Furthermore, the impurity profile of the resulting heterocyclic compounds is exceptionally clean, primarily due to the absence of solvent-derived byproducts and the minimization of thermal degradation pathways that are often exacerbated in prolonged reflux conditions. The patent data indicates that the crude products obtained after simple water washing possess purity levels greater than 98%, as confirmed by H-NMR spectroscopy, indicating that side reactions such as polymerization or over-alkylation are effectively suppressed. For quality control teams, this means a simplified analytical workflow and a reduced risk of batch failure due to trace impurities. The ability to produce high-purity pharmaceutical intermediates directly from the reactor without chromatographic purification is a significant technical achievement that underscores the robustness of this solvent-free methodology. It demonstrates that green chemistry principles can be successfully integrated into complex heterocyclic synthesis without compromising on the stringent quality standards required for medicinal chemistry applications.
How to Synthesize 2-Aminothiazole Derivatives Efficiently
The practical implementation of this solvent-free technology requires precise control over reaction parameters to ensure reproducibility and safety on a larger scale. The process begins with the selection of appropriate alpha-halogenated precursors and nitrogen sources, ensuring that at least one component is liquid at the reaction temperature to facilitate effective mixing. The reaction is typically conducted in a stirred vessel equipped with heating capabilities, where the temperature is ramped to the optimal range of 60°C to 120°C depending on the specific reactivity of the substrates. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities are critical for maintaining the high yields and purity observed in the patent examples. The following guide outlines the generalized steps for executing this synthesis, though specific conditions may vary based on the substituents involved.
- Mix alpha-halogenated compounds and nitrogen-containing reagents (such as thiourea or urea) in a reactor under stirring conditions.
- Heat the mixture to a temperature range of 60°C to 120°C and maintain stirring until the reaction solidifies or completes (typically 10-40 minutes).
- Add water to the reaction mass to wash the product, filter the solid, and dry to obtain the target heterocyclic compound with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free synthesis technology offers tangible strategic advantages that extend beyond mere technical novelty. The primary benefit lies in the drastic simplification of the manufacturing process, which directly correlates to reduced production costs and enhanced supply chain reliability. By eliminating the need for bulk organic solvents, manufacturers can significantly reduce their exposure to volatile raw material markets and the logistical complexities associated with solvent storage and handling. This reduction in material complexity also translates to a smaller physical footprint for production facilities, as large solvent storage tanks and recovery distillation columns are no longer required. Consequently, the capital expenditure for setting up new production lines is lowered, allowing for more agile responses to market demand fluctuations. The simplified workup procedure, involving only water washing and filtration, further reduces the labor and utility costs associated with downstream processing, making the overall cost structure of these intermediates highly competitive.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes a major cost center from the production budget, including the purchase of solvents, the energy costs for solvent recovery, and the fees for hazardous waste disposal. Without the need for complex distillation setups, the energy consumption per kilogram of product is significantly lowered, contributing to a leaner manufacturing model. Additionally, the high purity of the crude product reduces or eliminates the need for expensive recrystallization or chromatography steps, further driving down the cost of goods sold. This economic efficiency allows suppliers to offer more competitive pricing to their clients while maintaining healthy margins, creating a win-win scenario for the entire value chain.
- Enhanced Supply Chain Reliability: Relying on fewer raw materials and simpler processing steps inherently reduces the risk of supply chain disruptions. Solvent shortages or quality issues, which can halt production in traditional facilities, are rendered irrelevant in this solvent-free context. The robustness of the reaction conditions, which tolerate a range of temperatures and mixing speeds, ensures consistent batch-to-batch quality, minimizing the risk of production delays due to out-of-specification results. This reliability is crucial for pharmaceutical customers who require uninterrupted supply of key intermediates to maintain their own API production schedules. The ability to rapidly scale up production without the bottleneck of solvent handling capacity provides a distinct competitive advantage in meeting urgent market demands.
- Scalability and Environmental Compliance: The solvent-free nature of this process aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. Facilities adopting this technology can easily meet环保 compliance standards without investing in expensive scrubbing or abatement systems. The scalability of the process is excellent, as the heat transfer and mixing requirements are straightforward to engineer for large-scale reactors. The generation of primarily aqueous waste streams simplifies wastewater treatment, reducing the environmental burden and associated compliance costs. This sustainability profile enhances the brand reputation of the manufacturer as a responsible partner in the green chemistry movement, appealing to environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free heterocyclic synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or for sourcing these intermediates from external partners. The clarity provided here aims to bridge the gap between academic innovation and industrial application.
Q: How does the solvent-free method impact the purity profile of the final heterocyclic intermediate?
A: The solvent-free methodology described in patent CN102134224A eliminates the risk of residual organic solvent contamination, a common regulatory hurdle in API manufacturing. By relying on thermal cyclization followed by a simple aqueous wash, the process consistently yields products with purity levels exceeding 98%, often negating the need for further recrystallization or chromatographic purification.
Q: Is this solvent-free cyclization technology scalable for commercial production?
A: Yes, the technology is highly scalable. The reaction relies on the physical state of the reactants (at least one being liquid) and thermal energy, which translates well from laboratory glassware to industrial reactors. The absence of volatile organic solvents significantly reduces explosion risks and simplifies the engineering controls required for large-scale manufacturing.
Q: What are the environmental benefits compared to traditional Hantzsch synthesis?
A: Traditional methods often utilize large volumes of ethanol or DMF, generating significant hazardous waste. This novel approach replaces organic solvents with a neat reaction system, drastically reducing the E-factor (mass of waste per mass of product). The only waste stream generated is aqueous wash water, which is far easier and cheaper to treat than organic solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of solvent-free synthesis technologies in reshaping the future of fine chemical manufacturing. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying that every batch meets the highest international standards. We understand that the transition to greener chemistry requires not just technical capability but also a deep understanding of regulatory requirements and supply chain dynamics, areas where our team excels.
We invite you to collaborate with us to leverage this advanced solvent-free technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget. We encourage you to contact us to request specific COA data for our heterocyclic intermediates and to discuss route feasibility assessments for your target molecules. Together, we can drive efficiency, sustainability, and profitability in your chemical supply chain.
