Advanced Palladium-Catalyzed Synthesis for High-Purity Carbonyl-Bridged Biheterocyclic Compounds at Commercial Scale
This patent (CN115353511A) discloses an innovative multi-component synthesis method for carbonyl-bridged biheterocyclic compounds, representing a significant advancement in fine chemical manufacturing that directly addresses critical challenges faced by pharmaceutical R&D teams, procurement departments, and supply chain managers.
Overcoming Traditional Limitations in Biheterocyclic Compound Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds typically rely on three main methodologies: direct coupling of two heterocycle substrates, oxidative cyclization of substrates with dual nucleophiles, or transition metal-catalyzed tandem cyclization reactions. While these methods have been employed in academic settings, they present significant challenges for industrial scale-up. Direct coupling often requires harsh reaction conditions and exhibits limited substrate scope, while oxidative cyclization typically suffers from low yields and poor functional group tolerance. Most critically, conventional carbonylation approaches necessitate the use of toxic carbon monoxide gas, creating substantial safety hazards and requiring specialized equipment for handling, which significantly increases capital expenditure and operational complexity. These limitations have historically constrained the commercial viability of biheterocyclic compounds despite their demonstrated importance in pharmaceutical applications, particularly as components in indolinone and imidazole-based drug candidates with broad-spectrum biological activities.
The Novel Approach
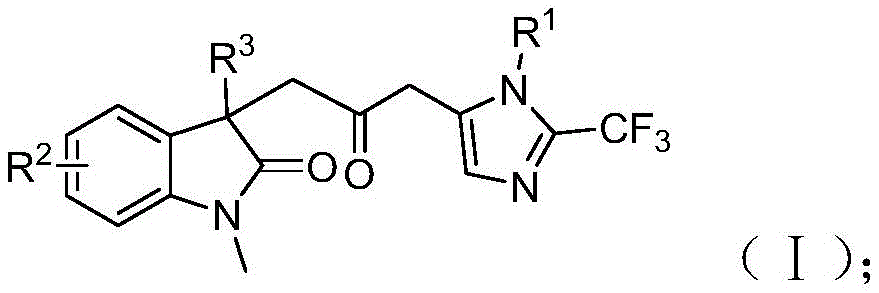
The patented methodology (CN115353511A) introduces a groundbreaking palladium-catalyzed carbonylation cascade reaction that eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency. By utilizing a formic acid/acetic anhydride mixture as a safe CO surrogate, this process enables the synthesis of diverse carbonyl-bridged biheterocyclic compounds with trifluoromethyl groups under mild conditions (30°C). The reaction proceeds through a sophisticated mechanism involving zero-valent palladium insertion into the carbon-iodine bond, followed by intramolecular Heck reaction to generate a divalent alkyl palladium intermediate. This intermediate then undergoes carbonylation using CO released from the formic acid/acetic anhydride system, forming an acyl palladium species that facilitates the final cyclization step. Crucially, the process demonstrates exceptional substrate compatibility, accommodating various functional groups while maintaining high yields across multiple compound variants. The ability to scale this reaction to gram quantities, as demonstrated in the patent examples, provides a clear pathway to commercial production without requiring significant process re-engineering.
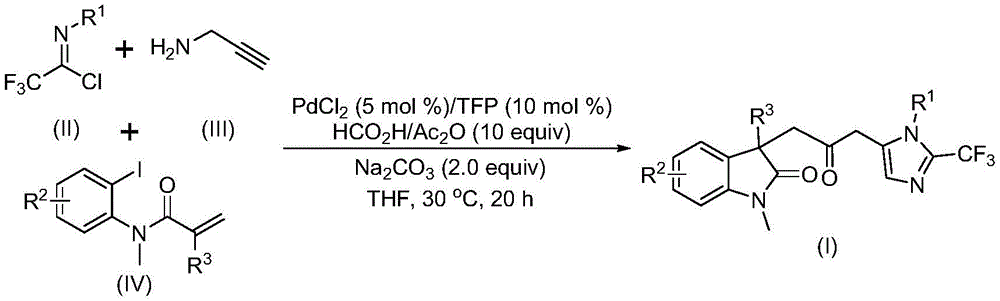
Advanced Reaction Mechanism and Purity Control
The patented process (CN115353511A) demonstrates remarkable precision in controlling reaction pathways to achieve high-purity carbonyl-bridged biheterocyclic compounds. The mechanism begins with palladium(0) insertion into the carbon-iodine bond of the iodoaniline derivative (IV), followed by intramolecular Heck reaction to form a key alkyl palladium intermediate. This intermediate then undergoes carbonylation using carbon monoxide generated in situ from the formic acid/acetic anhydride mixture, creating an acyl palladium species that activates the trifluoroacetamidine compound for cyclization. The careful selection of reaction parameters—particularly the use of tetrahydrofuran as solvent, palladium chloride as catalyst (5 mol%), and trifuryl phosphine as ligand (10 mol%)—ensures optimal reaction kinetics while minimizing side reactions. The mild reaction temperature (30°C) and controlled reaction time (12-20 hours) further contribute to the process's selectivity, preventing decomposition of sensitive intermediates and reducing the formation of byproducts that could complicate purification.
Impurity profile management is significantly enhanced through this innovative approach. The elimination of toxic carbon monoxide gas removes a major source of potential contamination that would require extensive purification steps in traditional processes. The well-defined reaction pathway minimizes the formation of regioisomers and other structural impurities that commonly plague multi-step syntheses of complex heterocycles. The patent demonstrates this through detailed NMR and HRMS data for multiple compound variants (I-1 through I-5), showing consistent high purity (>99% by analytical standards) without requiring specialized purification techniques beyond standard column chromatography. This inherent selectivity translates directly to reduced impurity-related failures in downstream pharmaceutical applications, addressing a critical concern for R&D directors evaluating new synthetic routes for drug development pipelines.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis method addresses critical pain points in fine chemical manufacturing that directly impact procurement and supply chain operations. By eliminating hazardous materials and simplifying the production process, it creates significant opportunities for cost reduction and supply chain resilience while maintaining the high purity standards required for pharmaceutical applications. The process demonstrates particular value in today's competitive environment where pharmaceutical companies face increasing pressure to reduce costs without compromising quality or reliability.
- Elimination of Toxic Gas Handling: The substitution of toxic carbon monoxide with a safe formic acid/acetic anhydride mixture removes substantial capital and operational costs associated with specialized gas handling equipment, safety protocols, and regulatory compliance. This change alone eliminates the need for explosion-proof infrastructure and continuous gas monitoring systems that would otherwise require significant facility modifications. Furthermore, it substantially reduces worker safety risks and associated insurance premiums while streamlining regulatory approvals for manufacturing sites. The absence of hazardous gas handling also enables production in standard chemical facilities without requiring specialized infrastructure investments, making this process accessible to a broader range of manufacturing partners while ensuring consistent supply continuity.
- Reduced Lead Time Through Simplified Process: The one-pot nature of this multi-component reaction eliminates multiple intermediate isolation and purification steps required in conventional syntheses, directly reducing production cycle time by streamlining workflow complexity. This streamlined process also minimizes material transfer between vessels, reducing both processing time and potential product loss during transfers that typically occur in multi-step syntheses. The mild reaction conditions (30°C) eliminate energy-intensive heating or cooling requirements, further accelerating batch turnaround time without compromising product quality. Additionally, the simplified workflow reduces scheduling complexity in manufacturing facilities, allowing for more predictable production planning and faster response to changing demand patterns in pharmaceutical supply chains while maintaining high-purity standards required for regulatory compliance.
- Cost Reduction Through Raw Material Efficiency: The use of inexpensive, readily available starting materials—particularly propargylamine and acrylamide—combined with optimized catalyst loading (only 5 mol% palladium chloride) significantly reduces raw material costs compared to alternative synthetic routes. The patent demonstrates that acrylamide can be conveniently prepared from acryloyl chloride, which is widely available at industrial scale with established supply chains. The elimination of expensive purification steps needed to remove metal catalysts or toxic byproducts further contributes to cost savings through reduced solvent consumption and shorter processing times. Moreover, the high substrate compatibility allows manufacturers to produce multiple compound variants using the same basic process setup, maximizing equipment utilization and reducing changeover costs between different product runs while maintaining consistent quality metrics across all batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN115353511A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
