Streamlined Manufacturing of Key PF-07321332 Intermediate Using Zinc-Mediated Cyclopropanation
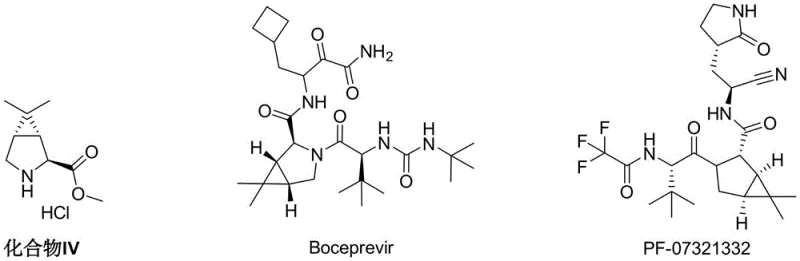
The global pharmaceutical landscape has been profoundly impacted by the emergence of potent antiviral agents, particularly those targeting the 3CL endoprotease of coronaviruses. Central to the supply chain of these life-saving medications, such as PF-07321332 (the active ingredient in Paxlovid), is the efficient production of key chiral intermediates. Patent CN114456101A discloses a groundbreaking synthesis method for N-protected (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate and its salts, which serve as the critical building blocks for both Hepatitis C therapeutics like Boceprevir and next-generation COVID-19 treatments. This technical insight report analyzes the transformative potential of this patented methodology, which shifts the paradigm from lengthy, hazardous multi-step sequences to a concise, zinc-mediated cyclopropanation strategy. By leveraging cheap and readily available double bond substrates alongside gem-dihalopropanes, this innovation addresses the urgent industry demand for reliable pharmaceutical intermediate suppliers capable of delivering high-purity materials with enhanced sustainability profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
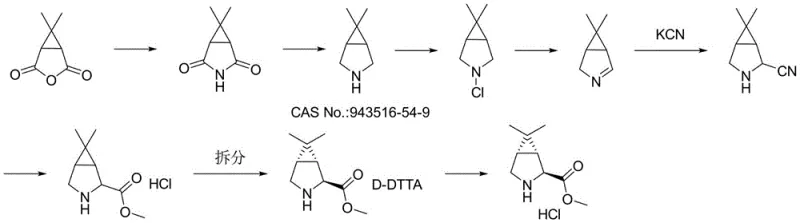
Prior to this innovation, the industrial synthesis of Compound IV relied heavily on legacy routes exemplified by WO 2012/049688A1, which presented severe bottlenecks for large-scale manufacturing. The conventional pathway is notoriously inefficient, requiring approximately eight distinct synthetic steps to construct the requisite azabicyclo[3.1.0]hexane core. A critical flaw in this traditional approach is the reliance on virulent reagents such as potassium cyanide (KCN) for ring expansion or functionalization, introducing significant safety hazards and complex waste disposal challenges that escalate operational costs. Furthermore, the stereochemical control in the old route is poor, necessitating a resolution step to isolate the biologically active (1R,2S,5S) enantiomer. This resolution inherently caps the maximum theoretical yield at 50%, leading to massive material consumption and waste generation. The starting material, caronic anhydride, is itself difficult to obtain and expensive, derived from the oxidation of chrysanthemic acid esters, further compounding the cost burden and supply chain fragility for procurement managers seeking cost reduction in API manufacturing.
The Novel Approach
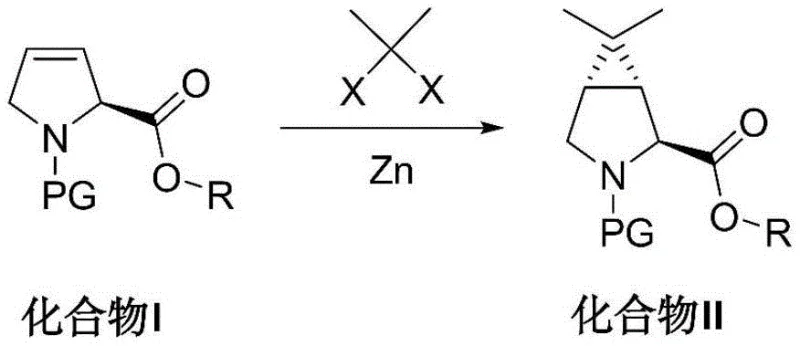
In stark contrast, the methodology disclosed in CN114456101A offers a radical simplification of the synthetic landscape by constructing the three-membered ring key intermediate in a single, highly efficient transformation. This novel approach utilizes a Simmons-Smith cyclopropanation reaction between a cheap, easily obtained double bond substrate (Compound I) and an accessible gem-dihalopropane. By directly forging the cyclopropane ring onto the pyrroline scaffold, the synthesis is condensed from eight steps down to merely three, representing a dramatic improvement in atom economy and process mass intensity. The use of zinc-mediated chemistry avoids the need for toxic cyanide reagents entirely, aligning with modern green chemistry principles. Moreover, the reaction exhibits excellent stereoselectivity, directly yielding the desired (1R,2S,5S) configuration without the need for wasteful resolution steps. This streamlined route not only lowers the barrier to entry for production but also significantly enhances the robustness of the supply chain for high-purity pharmaceutical intermediates.
Mechanistic Insights into Zinc-Mediated Simmons-Smith Cyclopropanation
The core of this technological breakthrough lies in the precise execution of the Simmons-Smith cyclopropanation, facilitated by a zinc-copper couple system. Mechanistically, the reaction involves the generation of an organozinc carbenoid species from the interaction of zinc powder and the gem-dihalopropane (such as 2,2-dibromopropane or 2,2-diiodopropane). The addition of a copper salt, such as cuprous chloride or copper sulfate, acts as a crucial activator, enhancing the reactivity of the zinc surface and facilitating the formation of the reactive zinc-carbenoid intermediate. This carbenoid then undergoes a concerted addition across the electron-rich double bond of the N-protected pyrroline substrate (Compound I). The stereochemical outcome is governed by the existing chirality at the C2 position of the pyrroline ring and the steric environment created by the N-protecting group (PG), which can range from Boc and Cbz to Fmoc and Bn. The reaction proceeds with high diastereoselectivity to install the dimethyl-substituted cyclopropane ring on the convex face of the molecule, establishing the critical (1R,2S,5S) stereocenters required for biological activity.
Impurity control in this process is inherently superior due to the direct nature of the bond formation. Unlike the prior art, where multiple functional group interconversions offered numerous opportunities for side reactions and racemization, this one-step ring closure minimizes the formation of structural analogs. The choice of solvent plays a pivotal role in managing the reaction kinetics and solubility of the organometallic species; ethers such as isopropyl ether, methyl tert-butyl ether, or tetrahydrofuran are preferred for their ability to stabilize the zinc carbenoid while remaining inert to the reaction conditions. The inclusion of catalysts like titanium tetrachloride further refines the reaction profile, likely by coordinating with the ester carbonyl or the nitrogen protecting group to direct the approach of the carbenoid, ensuring that the impurity profile remains clean and manageable for downstream processing, a key concern for R&D directors focused on purity specifications.
How to Synthesize N-Protected (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate Efficiently
Implementing this synthesis requires careful attention to the activation of the zinc reagent and the control of reaction temperature to maximize yield and stereoselectivity. The patent outlines a robust protocol where the reactants are combined under an inert nitrogen atmosphere to prevent oxidation of the sensitive organozinc intermediates. The process is versatile regarding the protecting groups and ester substituents, allowing manufacturers to tailor the intermediate to specific downstream coupling requirements. While the general chemistry is straightforward, the precise stoichiometry of the zinc-to-substrate ratio and the timing of catalyst addition are critical process parameters that determine the success of the scale-up. For a detailed breakdown of the standardized operating procedures, including specific reagent grades and workup protocols, please refer to the technical guide below.
- Prepare the reaction mixture by combining an organic solvent (such as isopropyl ether), zinc powder, a copper salt activator, 2,2-dihalopropane, and the protected pyrrole substrate (Compound I) under a nitrogen atmosphere.
- Heat the mixture to reflux temperatures (approximately 65°C) and introduce a catalyst such as titanium tetrachloride to initiate the Simmons-Smith cyclopropanation, maintaining reaction for 3 to 24 hours.
- Quench the reaction with aqueous ammonium chloride, filter off residual zinc, extract the organic phase, and purify the resulting N-protected bicyclic ester (Compound II) via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic advantages that extend beyond simple chemistry. The elimination of the resolution step alone represents a doubling of theoretical yield from the chiral pool, which fundamentally alters the cost structure of the intermediate. By removing the need to discard half of the produced material, the process dramatically reduces the raw material burden per kilogram of final product. Furthermore, the substitution of hazardous potassium cyanide with benign zinc and copper salts simplifies the regulatory compliance landscape, reducing the costs associated with hazardous waste disposal and worker safety monitoring. This shift allows for more flexible manufacturing site selection, as facilities do not require specialized containment for cyanide handling, thereby enhancing supply chain resilience and continuity.
- Cost Reduction in Manufacturing: The streamlined three-step sequence significantly lowers the operational expenditure compared to the legacy eight-step route. By utilizing inexpensive, commodity-grade starting materials like 2,2-dibromopropane and avoiding costly chiral resolving agents, the overall cost of goods sold (COGS) is substantially decreased. The high yields reported in the examples (up to 78% for the key cyclopropanation step and 97% for the deprotection) indicate a highly efficient process that minimizes material loss. Additionally, the simplified workup, which involves basic filtration and extraction rather than complex chromatographic separations, reduces solvent consumption and energy usage, contributing to further economic efficiency in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-restricted reagents ensures a stable supply chain不受 geopolitical or regulatory disruptions that often affect specialized chiral reagents or toxic chemicals. The robustness of the zinc-mediated reaction means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures. This reliability is crucial for maintaining the continuous production schedules required for global antiviral drug supplies. The ability to source starting materials from multiple vendors without compromising the reaction outcome provides procurement teams with greater negotiating power and security against supply shortages.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard reactor equipment and avoiding extreme conditions such as cryogenic temperatures or high pressures. The use of isopropyl ether and other common solvents facilitates easy recovery and recycling, aligning with sustainability goals. The absence of heavy metal catalysts (other than trace copper which is easily removed) and toxic cyanide wastes simplifies the environmental permitting process and reduces the ecological footprint of the manufacturing site. This environmental compatibility is increasingly becoming a prerequisite for partnerships with major pharmaceutical companies committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve upon the conventional method for PF-07321332 intermediates?
A: The conventional route described in WO 2012/049688A1 requires approximately 8 steps and utilizes hazardous reagents like potassium cyanide, necessitating a resolution step that wastes 50% of the material. The novel method disclosed in CN114456101A reduces the synthesis to just 3 steps, eliminates the need for toxic cyanide, and achieves high stereoselectivity directly, thereby drastically reducing material costs and environmental hazards.
Q: What are the critical reaction conditions for the zinc-mediated cyclopropanation?
A: The reaction typically employs zinc powder (4 equivalents) and a copper salt activator (such as cuprous chloride) in an ether solvent like isopropyl ether or THF. The process operates effectively at reflux temperatures around 65°C. A catalyst, such as titanium tetrachloride (0.02 equivalents), is injected to activate the organozinc species, ensuring high yield (up to 78%) and correct stereochemistry (1R,2S,5S).
Q: Is this process scalable for commercial production of antiviral intermediates?
A: Yes, the process is highly scalable. It utilizes inexpensive, commercially available starting materials like 2,2-dibromopropane and avoids cryogenic conditions often associated with traditional Simmons-Smith reactions. The workup involves simple filtration of zinc residues and standard extraction, making it robust for multi-kilogram to ton-scale manufacturing without requiring specialized hazardous waste handling for cyanide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PF-07321332 Intermediate Supplier
The synthesis method disclosed in CN114456101A represents a significant leap forward in the production of antiviral intermediates, yet translating patent chemistry into commercial reality requires deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to bridge this gap, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling zinc-mediated chemistries safely and efficiently, ensuring that the high yields and purity demonstrated in the lab are maintained at scale. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-protected (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate meets the exacting standards required for GMP API synthesis.
We invite pharmaceutical partners to collaborate with us to fully realize the cost and efficiency benefits of this innovative route. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this streamlined synthesis can optimize your supply chain. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation, ensuring a secure and economical supply of this critical pandemic-response intermediate.
