Advanced Manufacturing of Triazolinethione Derivatives for High-Efficiency Fungicide Production
Introduction to Patent CN1137103C and Technological Breakthroughs
The global demand for high-efficacy fungicidal agents continues to drive innovation in the synthesis of heterocyclic intermediates, particularly within the triazole class. Patent CN1137103C introduces a transformative methodology for the preparation of triazolinethione derivatives, which serve as critical active compounds possessing potent microbicidal and specifically fungicidal activity. This intellectual property represents a significant leap forward from prior art, addressing long-standing inefficiencies in heterocyclic ring construction. By leveraging a novel two-step sequence involving the condensation of hydrazine derivatives with formaldehyde and thiocyanates, followed by a controlled oxidative dehydrogenation, this process achieves superior purity profiles and operational simplicity. For R&D directors and procurement strategists, understanding the nuances of this patented route is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality at scale. The technology effectively bypasses the limitations of traditional sulfurization methods, offering a streamlined pathway that aligns with modern green chemistry principles while maintaining the structural integrity required for high-performance crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazolinethione derivatives has been plagued by significant technical hurdles that impede efficient commercial scale-up of complex heterocyclic intermediates. Prior art, such as that described in WO-A96-16048, typically relies on reacting triazole derivatives sequentially with strong bases and elemental sulfur, followed by hydrolysis, or direct reaction with sulfur at elevated temperatures. These conventional approaches suffer from inherently low yields and require reaction conditions that are difficult to maintain safely on an industrial scale, often necessitating specialized equipment to handle corrosive bases and high-temperature sulfur melts. Furthermore, alternative literature methods involving N-chlorothioformyl-N-(1-chloro-1-alkenyl)-amines or phenylhydrazine reactions with sodium thiocyanate often result in excessively long reaction times or fail to produce the desired unsubstituted 3-position derivatives entirely. The reliance on cryogenic conditions for lithiation steps in some legacy processes further exacerbates cost and safety concerns, creating a bottleneck for manufacturers seeking cost reduction in fungicide manufacturing. These inefficiencies translate directly into higher production costs, increased waste generation, and supply chain vulnerabilities that modern agrochemical enterprises can ill afford.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the process disclosed in CN1137103C offers a remarkably elegant and efficient solution. The novel approach initiates with the reaction of a hydrazine derivative of formula (II) with formaldehyde and a thiocyanate salt, such as ammonium thiocyanate, in the presence of a diluent and optionally an acid. This first step constructs the triazolidinethione ring system under mild conditions, typically ranging from -20°C to +100°C, avoiding the extreme thermal stresses of previous methods. The subsequent second step involves the conversion of this intermediate into the final triazolinethione derivative via oxidation using agents like oxygen, sulfur, or potassium peroxide, or alternatively through treatment with formic acid. This two-stage protocol not only simplifies the synthetic route but also dramatically improves the overall yield and purity of the final product. As illustrated in the reaction scheme below, the transformation proceeds smoothly without the need for hazardous strong bases or cryogenic reagents, marking a paradigm shift in how these valuable intermediates are produced.
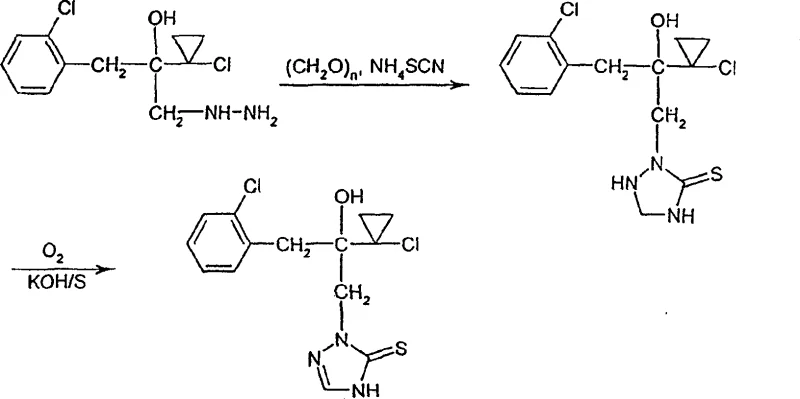
Mechanistic Insights into Hydrazine-Thiocyanate Cyclization and Oxidation
To fully appreciate the technical superiority of this method, one must delve into the mechanistic details of the cyclization and subsequent aromatization steps. The process begins with the nucleophilic attack of the hydrazine moiety in the starting material, depicted structurally in the image below, upon the electrophilic carbon of the formaldehyde-thiocyanate complex. This interaction facilitates the formation of the five-membered triazolidine ring, a saturated precursor that is stable enough to be isolated or carried forward in situ. The versatility of this mechanism is underscored by the broad tolerance for various R1 and R2 substituents, ranging from simple alkyl chains to complex aryl and heteroaryl groups, ensuring that the process is applicable to a wide library of potential agrochemical candidates. The use of diluents such as methyl tert-butyl ether or alcohols provides a homogeneous reaction medium that enhances mass transfer and kinetic control, crucial for minimizing side reactions and maximizing the formation of the desired regioisomer.
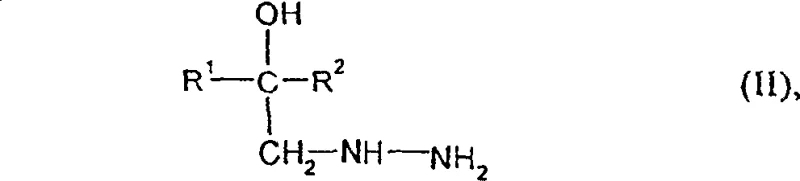
Following the initial cyclization, the critical transformation involves the dehydrogenation of the triazolidinethione intermediate to form the unsaturated triazolinethione ring. This oxidative step is mechanistically distinct from the harsh sulfurization of the past; instead, it utilizes mild oxidants like atmospheric oxygen in the presence of a basic catalyst such as potassium hydroxide and sulfur powder. This catalytic system promotes the removal of hydrogen atoms from the ring system, establishing the conjugated double bond necessary for the compound's biological activity. Alternatively, the use of formic acid serves as both a reactant and a solvent, driving the elimination of water and facilitating aromatization through an acid-catalyzed mechanism. This dual-pathway flexibility allows manufacturers to optimize the process based on available infrastructure and specific substrate requirements, ensuring robust impurity control mechanisms are in place to meet stringent purity specifications required for downstream formulation.
How to Synthesize Triazolinethione Derivatives Efficiently
Implementing this synthesis route in a production environment requires careful attention to reaction parameters and workup procedures to ensure optimal recovery and quality. The process is designed to be operationally simple, utilizing common industrial solvents and reagents that are readily accessible in the global chemical supply chain. The initial condensation step can be performed in a standard stirred tank reactor, where temperature control is maintained within the specified range to prevent decomposition of the sensitive hydrazine intermediates. Following the reaction, standard aqueous workup techniques involving washing with brine or mild base solutions effectively remove inorganic salts and unreacted thiocyanates. The subsequent oxidation step can be conducted in the same vessel or a dedicated reactor, with the choice of oxidant dictating the specific safety protocols required. For a detailed breakdown of the standardized operating procedures, including specific molar ratios, addition rates, and purification techniques, please refer to the technical guide below.
- React a hydrazine derivative of formula (II) with formaldehyde and a thiocyanate salt (such as ammonium thiocyanate) in a diluent like methyl tert-butyl ether, optionally with an acid catalyst, to form the triazolidinethione intermediate.
- Subject the resulting triazolidinethione compound of formula (IV) to an oxidative dehydrogenation step using oxygen (air), sulfur, or potassium peroxide in the presence of a catalyst like potassium hydroxide.
- Alternatively, treat the intermediate with formic acid to effect cyclization and aromatization, followed by standard workup procedures including solvent extraction and recrystallization to isolate the high-purity final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages for procurement managers and supply chain heads focused on cost reduction in fungicide manufacturing and supply reliability. The elimination of cryogenic reagents like n-butyllithium and the avoidance of high-temperature sulfur melts significantly lowers the barrier to entry for production, reducing both capital expenditure on specialized equipment and operational expenditure on energy and safety measures. By utilizing commodity chemicals such as formaldehyde and ammonium thiocyanate, the process insulates manufacturers from the volatility associated with niche reagent markets, thereby enhancing supply chain reliability. Furthermore, the simplified workup procedures, which often involve straightforward filtration and crystallization rather than complex chromatographic separations, lead to substantial cost savings in downstream processing and waste management. This efficiency translates directly into a more competitive pricing structure for the final active ingredient, providing a strategic advantage in the highly price-sensitive agrochemical sector.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and cryogenic cooling systems, which are capital-intensive and energy-demanding. By operating at near-ambient pressures and moderate temperatures, the facility utility load is drastically reduced, leading to lower overhead costs per kilogram of product. Additionally, the high atom economy of the cyclization step minimizes raw material waste, further contributing to substantial cost savings without compromising on the quality of the output.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals like formaldehyde and thiocyanates ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to reagent shortages. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demands. This resilience is critical for maintaining the continuity of supply for key agrochemical intermediates, ensuring that downstream formulation partners receive their orders on time.
- Scalability and Environmental Compliance: The absence of hazardous strong bases and the ability to use air as an oxidant significantly improve the environmental footprint of the manufacturing process. This alignment with green chemistry principles simplifies regulatory compliance and reduces the costs associated with waste disposal and emissions control. The process is inherently scalable from pilot plant to multi-ton commercial production, allowing for seamless technology transfer and rapid capacity expansion to meet growing global demand for high-performance fungicides.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the flexibility and robustness of the method, reassuring stakeholders about its viability for large-scale production.
Q: What are the primary advantages of this new synthesis method over conventional triazole thione production?
A: Unlike conventional methods that require harsh conditions such as strong bases, elemental sulfur at high temperatures, or cryogenic lithiation, this novel process operates under mild conditions (-20°C to 100°C) and utilizes readily available reagents like formaldehyde and thiocyanates, resulting in significantly higher yields and easier industrial scalability.
Q: Can this process accommodate diverse substituent groups on the hydrazine starting material?
A: Yes, the method demonstrates exceptional substrate scope, tolerating a wide variety of R1 and R2 groups including substituted alkyl, alkenyl, cycloalkyl, aralkyl, and heteroaryl moieties, allowing for the flexible production of diverse agrochemical intermediates without compromising reaction efficiency.
Q: What oxidizing agents are suitable for the second step of the reaction?
A: The patent specifies several effective oxidizing options for converting the triazolidinethione intermediate to the final triazolinethione, including oxygen (preferably as air), sulfur powder, potassium peroxide, or alternatively, formic acid can be used to drive the reaction through a different mechanistic pathway.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinethione Derivatives Supplier
As the agrochemical industry evolves, the need for partners who can bridge the gap between innovative patent chemistry and commercial reality has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of the CN1137103C process, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the successful deployment of this technology requires not just chemical expertise but also a deep commitment to quality assurance and regulatory compliance, values that are embedded in our corporate culture. By partnering with us, you gain access to a supply chain that is both resilient and responsive, capable of delivering the high-purity triazolinethione derivatives necessary for next-generation crop protection solutions.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new product development, our experts are ready to assist. Let us demonstrate how our manufacturing capabilities can enhance your competitive position in the global market, ensuring a steady flow of critical intermediates for your fungicide formulations.
