Advanced Manufacturing of Triazolinethione Derivatives for Fungicide Production
Advanced Manufacturing of Triazolinethione Derivatives for Fungicide Production
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of heterocyclic intermediates, particularly those based on the triazolinethione scaffold. A pivotal advancement in this domain is detailed in Chinese Patent CN1121397C, which discloses a novel and robust method for preparing triazolinethione derivatives of formula (I). This technology represents a significant leap forward for manufacturers seeking to optimize their production lines for agrochemical active ingredients. By leveraging a unique two-step sequence involving hydrazine derivatives, carbonyl compounds, and thiocyanates, followed by a formic acid-mediated cyclization, this process overcomes the longstanding limitations of traditional synthetic routes. For R&D directors and procurement specialists alike, understanding the nuances of this patented methodology is crucial for securing a competitive edge in the supply of high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the technology outlined in CN1121397C, the synthesis of triazolinethione derivatives was fraught with significant technical and economic challenges that hindered large-scale commercial viability. Conventional methods, such as those described in WO-A96-16048, typically relied on the reaction of triazole derivatives with strong bases and elemental sulfur, followed by hydrolysis, or direct reaction with sulfur at elevated temperatures. These processes were inherently problematic; they often resulted in relatively low yields of the desired product, necessitating extensive purification steps that drove up costs and waste generation. Furthermore, the requirement for high-temperature conditions with elemental sulfur posed safety risks and equipment corrosion issues, making industrial scale-up difficult to maintain consistently. Another historical approach, documented in Bull.Chem.Soc.Japan, involved reacting phenylhydrazine with sodium thiocyanate and ketones in the presence of hydrochloric acid, but this method suffered from excessively long reaction times and was incapable of producing unsubstituted triazolinethione compounds at the 3-position, severely limiting its utility for diverse chemical libraries.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach presented in the patent introduces a streamlined, high-yielding pathway that utilizes mild reagents and accessible reaction conditions. The core innovation lies in the initial condensation of a hydrazine derivative of formula (II) with a carbonyl compound of formula (III), such as acetone, in the presence of a thiocyanate salt like potassium thiocyanate. This step efficiently constructs the triazolidinethione ring system under moderate conditions, avoiding the need for hazardous elemental sulfur or cryogenic temperatures. The subsequent treatment of this intermediate with formic acid facilitates a selective dehydration and aromatization to yield the final triazolinethione structure with exceptional purity. As illustrated in the reaction scheme below, this methodology not only simplifies the operational workflow but also dramatically improves the overall mass balance of the process.
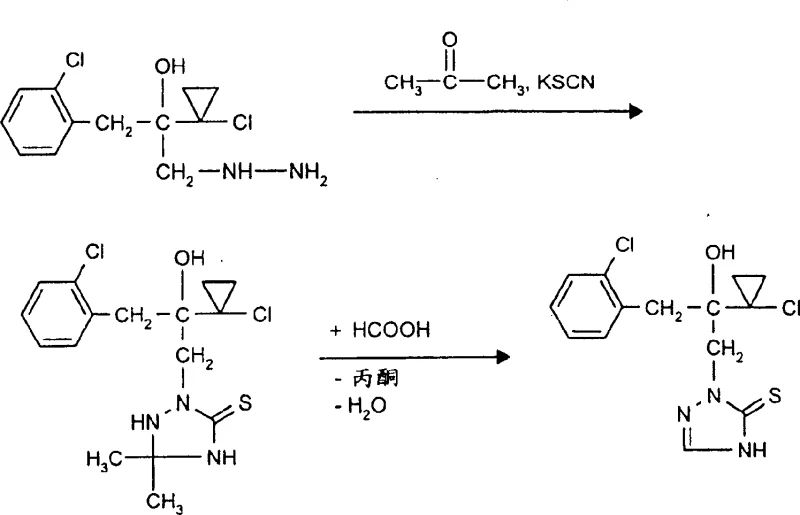
The versatility of this new route is further evidenced by its compatibility with a broad spectrum of substrates. The patent explicitly details that R1 and R2 groups can vary widely, encompassing straight-chain or branched alkyls, alkenyls, cycloalkyls, and various aryl or heteroaryl moieties. This structural flexibility allows manufacturers to produce a diverse array of fungicidal intermediates using a single, standardized platform technology. Moreover, the use of commodity chemicals such as acetone, potassium thiocyanate, and formic acid ensures that the raw material supply chain remains stable and cost-effective, addressing a primary concern for procurement managers looking to mitigate volatility in input costs.
Mechanistic Insights into Thiocyanate-Mediated Cyclization
From a mechanistic perspective, the success of this synthesis hinges on the precise orchestration of nucleophilic attacks and cyclization events driven by the thiocyanate ion. In the first stage, the hydrazine derivative acts as a nucleophile, attacking the carbonyl carbon of the ketone or aldehyde to form a hydrazone intermediate in situ. The thiocyanate ion then participates in a cyclization reaction, likely attacking the electrophilic center generated by the protonated hydrazone or iminium species, leading to the formation of the five-membered triazolidinethione ring. This step is critical as it establishes the core heterocyclic framework while incorporating the sulfur atom directly from the thiocyanate source, thereby bypassing the need for separate thionation steps that often require toxic reagents like Lawesson's reagent or phosphorus pentasulfide. The reaction is typically conducted in the presence of an acid catalyst, such as hydrochloric acid or p-toluenesulfonic acid, which activates the carbonyl group and facilitates the elimination of water during the condensation phase.
The second stage involves the conversion of the saturated triazolidinethione intermediate into the unsaturated triazolinethione final product. This transformation is achieved through treatment with formic acid, which serves both as a solvent and a dehydrating agent. The mechanism likely involves the protonation of the nitrogen atom followed by the elimination of a molecule corresponding to the R3 and R4 groups (often as a ketone or aldehyde byproduct, such as acetone), resulting in the formation of the double bond within the ring system. This step proceeds with high selectivity, ensuring that the sensitive functional groups on the R1 and R2 side chains remain intact. The ability to perform this dehydration under relatively mild thermal conditions (80°C-150°C) prevents the degradation of thermally labile substituents, a common issue in high-temperature sulfurization methods. This mechanistic elegance translates directly into a cleaner impurity profile, reducing the burden on downstream purification units and enhancing the overall quality of the final API intermediate.
How to Synthesize Triazolinethione Derivatives Efficiently
Implementing this synthesis route in a commercial setting requires careful attention to reaction parameters and stoichiometry to maximize yield and minimize waste. The patent outlines a preferred embodiment where the hydrazine derivative is reacted with a slight excess of the carbonyl compound and thiocyanate salt in a suitable diluent such as toluene or an alcohol. Following the formation of the intermediate, the reaction mixture can be worked up to isolate the solid triazolidinethione, which is then subjected to the formic acid treatment. Alternatively, for even greater efficiency, the patent describes a "one-pot" variant where the hydrazine precursor is generated in situ from chlorohydrins or epoxides and reacted directly without isolation. This telescoped approach reduces solvent consumption and handling time, offering substantial operational savings. The visual representation of this continuous process highlights the seamless transition between the hydrazine formation and the subsequent cyclization steps.
- React a hydrazine derivative of formula (II) with a carbonyl compound (such as acetone) and potassium thiocyanate in the presence of an acid like hydrochloric acid to form a triazolidinethione intermediate.
- Treat the resulting triazolidinethione derivative with formic acid, optionally with a catalyst, at elevated temperatures (80°C-150°C) to effect cyclization and dehydration.
- Isolate the final triazolinethione product through standard workup procedures including washing, drying, and recrystallization.

Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1121397C offers compelling strategic benefits that extend beyond mere chemical curiosity. The shift away from specialized, hazardous reagents towards commodity chemicals fundamentally alters the cost structure and risk profile of manufacturing these valuable intermediates. By eliminating the dependence on elemental sulfur and strong bases, facilities can reduce their exposure to volatile raw material markets and stringent environmental regulations associated with sulfur waste disposal. Furthermore, the improved yield profile means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS) without compromising on quality standards. This efficiency gain is critical for maintaining margins in the highly competitive agrochemical sector.
- Cost Reduction in Manufacturing: The replacement of expensive or hazardous thionation agents with inexpensive potassium thiocyanate and formic acid leads to a drastic simplification of the bill of materials. Since acetone and formic acid are produced on a massive global scale, their pricing is generally stable and predictable, allowing for more accurate long-term budgeting. Additionally, the avoidance of cryogenic conditions (such as the -70°C required in some comparative examples) significantly reduces energy consumption for cooling, contributing to lower utility costs per kilogram of product. The high selectivity of the reaction also minimizes the formation of byproducts, which reduces the load on waste treatment facilities and lowers the cost of solvent recovery and recycling operations.
- Enhanced Supply Chain Reliability: Relying on widely available building blocks enhances the resilience of the supply chain against disruptions. Unlike specialized reagents that may have single-source suppliers or long lead times, the key inputs for this process—hydrazines, ketones, and thiocyanates—are stocked by numerous chemical distributors worldwide. This redundancy ensures that production schedules can be maintained even if one supplier faces logistical challenges. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch failures and ensuring a consistent flow of material to downstream customers who rely on just-in-time delivery models for their own formulation plants.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates easier scale-up from pilot plant to commercial production volumes. The absence of highly exothermic steps or the generation of toxic gases like hydrogen sulfide simplifies the engineering requirements for large-scale reactors, lowering capital expenditure for new capacity. From an environmental standpoint, the process generates fewer hazardous byproducts, aligning with increasingly strict global regulations on industrial emissions and effluent discharge. The ability to recycle solvents like toluene and the use of aqueous workups further supports sustainability goals, making this technology attractive for companies aiming to reduce their carbon footprint and achieve green chemistry certifications.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis route for their specific applications, we have compiled answers to common questions regarding the process parameters and scope. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for process development decisions. Understanding these details is essential for assessing the compatibility of this method with existing infrastructure and quality control protocols.
Q: What are the key advantages of this new synthesis method over prior art?
A: The method described in CN1121397C offers significantly higher yields (e.g., 82.1% in the first step) compared to conventional methods which often suffer from low yields or require harsh conditions like high temperatures with elemental sulfur. It also utilizes readily available starting materials like acetone and potassium thiocyanate.
Q: Can this process be performed as a one-pot reaction?
A: Yes, the patent describes a variant where the hydrazine derivative is generated in situ from chlorohydrin or epoxide precursors and reacted directly without isolation, streamlining the production workflow and reducing solvent usage.
Q: What types of substituents are compatible with this reaction?
A: The process is highly versatile, accommodating a wide range of substituents (R1, R2) including alkyl, alkenyl, cycloalkyl, aralkyl, and various heteroaryl groups, allowing for the synthesis of diverse fungicidal active ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinethione Derivatives Supplier
As the agrochemical industry continues to evolve, the ability to access high-quality intermediates through efficient and sustainable manufacturing routes becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced technologies like the one described in CN1121397C to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of triazolinethione derivatives meets the exacting standards required for modern fungicide formulations.
We invite you to explore how our optimized synthesis capabilities can enhance your supply chain efficiency and product performance. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to drive innovation and reliability in your agrochemical manufacturing operations, ensuring a secure and prosperous future for your business.
