Advanced Synthesis of Triazolinethione Derivatives for High-Efficiency Fungicide Production
Advanced Synthesis of Triazolinethione Derivatives for High-Efficiency Fungicide Production
The agrochemical industry continuously demands more efficient and sustainable pathways for producing active pharmaceutical and agricultural ingredients. Patent CN1178924C discloses a groundbreaking method for preparing triazolinethione derivatives of formula (I), which serve as critical intermediates possessing potent microbicidal and fungicidal activity. This technology represents a significant departure from traditional synthesis routes, offering a streamlined two-step process that begins with the reaction of specific hydrazine derivatives with thiocyanates. By leveraging this novel chemistry, manufacturers can achieve superior yield profiles and operational simplicity compared to legacy methods. The core innovation lies in the strategic construction of the triazole ring system through a thiosemicarbazide intermediate, avoiding the harsh conditions typically associated with sulfur incorporation. 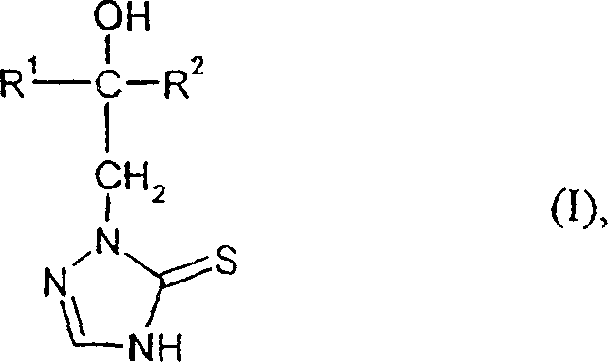
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazoline-5-thione compounds has been plagued by significant technical hurdles that impede efficient commercial manufacturing. Prior art, such as that described in WO-A96-16048, often relies on reacting triazole derivatives sequentially with strong bases and elemental sulfur, followed by hydrolysis, or directly reacting them with sulfur at elevated temperatures. These conventional approaches suffer from inherently low yields and require reaction conditions that are difficult to maintain safely and consistently on an industrial scale. Furthermore, alternative literature methods involving N-chlorothioformyl derivatives fail to address the synthesis of compounds unsubstituted at the 3-position, limiting their utility for specific high-value agrochemical targets. The reliance on elemental sulfur at high temperatures also introduces safety risks and potential impurity profiles that complicate downstream purification processes.
The Novel Approach
In stark contrast, the method disclosed in CN1178924C utilizes a mild and highly selective pathway involving thiocyanate chemistry. The process initiates by reacting a hydrazine derivative of formula (II) with a thiocyanate salt, such as ammonium thiocyanate, to form a novel thiosemicarbazide intermediate of formula (IV). This intermediate is subsequently cyclized using formic acid to yield the target triazolinethione structure. This approach eliminates the need for cryogenic conditions or hazardous sulfur powder handling at extreme temperatures. As illustrated in the specific reaction scheme below, the conversion proceeds smoothly under atmospheric pressure with standard organic solvents, demonstrating a clear advantage in process robustness and operator safety. 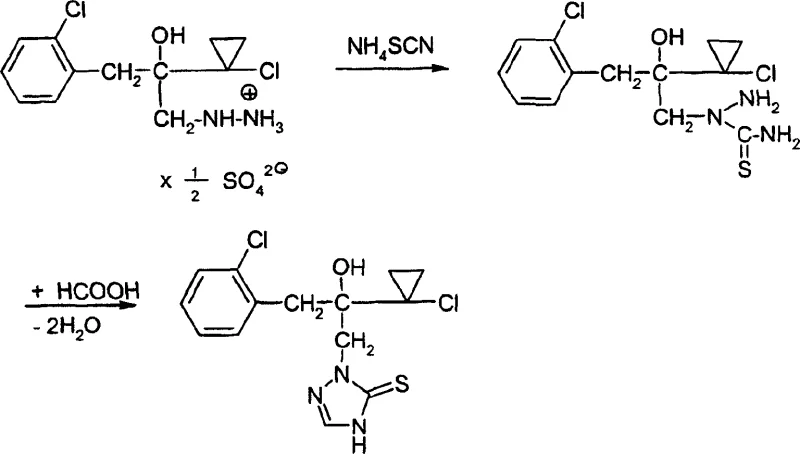
Mechanistic Insights into Thiocyanate-Mediated Cyclization
The mechanistic elegance of this synthesis lies in the unexpected regioselectivity observed during the initial nucleophilic attack. In the first step, the thiocyanate anion attacks the substituted nitrogen atom of the hydrazine derivative rather than the terminal nitrogen. This specific orientation is crucial as it directs the subsequent cyclization towards the desired 1,2,4-triazoline-5-thione scaffold with high fidelity. The formation of the thiosemicarbazide intermediate serves as a stable precursor that can be isolated or carried forward in situ, providing flexibility in process design. The use of formic acid in the second step acts as both a solvent and a dehydrating agent, facilitating the ring closure and the elimination of water to aromatize the triazole system. This dual role of formic acid simplifies the reagent list and reduces waste generation.
Furthermore, the resulting triazolinethione derivatives exist in a tautomeric equilibrium between the thione and thiol forms, a phenomenon that is critical for understanding their biological activity and stability. The patent explicitly notes that while both forms may exist, the thione form is typically represented for simplicity. Understanding this tautomerism is essential for R&D teams optimizing formulation stability and bioavailability. The mild acidic conditions employed in the cyclization step help stabilize the desired tautomer, ensuring a consistent impurity profile. This level of control over the molecular architecture is a key differentiator for manufacturers seeking to produce high-purity agrochemical intermediates with minimal batch-to-batch variation. 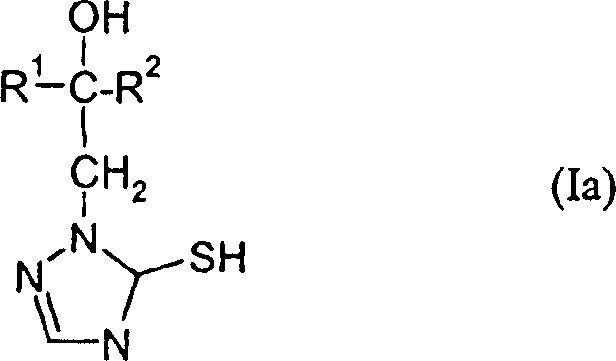
How to Synthesize Triazolinethione Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into two distinct stages: the formation of the thiosemicarbazide intermediate and the subsequent cyclization. Operators should note that the reaction temperatures are moderate, typically ranging between 70°C and 110°C, which allows for the use of standard glass-lined or stainless steel reactors without specialized cryogenic capabilities. The workup procedures involve conventional liquid-liquid extractions and drying steps, making the technology accessible for facilities with standard organic synthesis infrastructure. For detailed operational parameters and stoichiometric ratios, please refer to the standardized guide below.
- React a hydrazine derivative of formula (II) or its acid addition salt with a thiocyanate (Y-SCN) in the presence of a diluent and optionally a catalyst to form a thiosemicarbazide intermediate.
- React the resulting thiosemicarbazide derivative of formula (IV) with formic acid, optionally in the presence of a catalyst and diluent, to effect cyclization and dehydration.
- Isolate the final triazolinethione derivative of formula (I) through standard workup procedures such as washing, drying, and concentration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement managers and supply chain directors focused on cost reduction in fungicide manufacturing. The shift away from expensive and hazardous reagents like n-butyllithium or elemental sulfur towards commodity chemicals like ammonium thiocyanate and formic acid drastically simplifies the raw material sourcing strategy. This transition not only lowers the direct material costs but also mitigates the supply risk associated with specialized reagents that may have volatile market availability. The simplified process flow reduces the number of unit operations required, leading to shorter production cycles and improved asset utilization rates within the manufacturing plant.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and expensive organometallic reagents significantly lowers energy consumption and reagent costs. By avoiding the need for low-temperature reactors and complex quenching procedures associated with comparative examples, the overall operational expenditure is reduced. The use of formic acid as a cyclization agent is economically favorable compared to multi-step activation protocols, driving down the cost of goods sold for the final active ingredient.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including hydrazine derivatives and thiocyanate salts, are widely available from multiple global suppliers. This diversity in the supply base ensures continuity of supply even during market fluctuations. Additionally, the robustness of the reaction conditions means that production is less susceptible to delays caused by equipment maintenance or specialized utility requirements, thereby reducing lead time for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to sulfur-based methods, simplifying waste treatment and disposal. The absence of heavy metal catalysts or toxic sulfur residues aligns with increasingly stringent environmental regulations. This green chemistry profile facilitates easier regulatory approval and supports the sustainability goals of modern agrochemical companies, making the commercial scale-up of complex triazolinethione derivatives more feasible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a realistic view of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of this thiocyanate method over conventional sulfur powder reactions?
A: The conventional method often requires reacting triazole derivatives with sulfur powder at high temperatures or using strong bases followed by hydrolysis, which can lead to low yields and difficult industrial maintenance. The novel thiocyanate route operates under significantly milder conditions (typically 70°C-110°C) and avoids the handling of elemental sulfur at extreme temperatures, resulting in higher selectivity and easier purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is highly amenable to scale-up. It utilizes readily available raw materials such as ammonium thiocyanate and formic acid. The reaction steps involve standard organic synthesis operations like heating, washing, and concentration, without the need for complex chromatographic separations or cryogenic conditions found in comparative examples, ensuring robust supply chain continuity.
Q: How does the process control regioselectivity during the formation of the triazole ring?
A: The process exhibits surprising selectivity where the thiocyanate attack occurs primarily on the substituted nitrogen atom of the hydrazine derivative rather than the terminal nitrogen. This specific mechanistic pathway ensures the high-yield formation of the desired 1,2,4-triazoline-5-thione isomer, minimizing the generation of unwanted regioisomers that would complicate downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinethione Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the thiocyanate-mediated cyclization described in CN1178924C to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of triazolinethione derivatives meets the exacting standards required for modern fungicide formulations.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can enhance your competitive position in the global agrochemical market.
